Enhance Reliability of Parts and Components in Medical Devices
Introduction The reliability of parts and components in medical devices is essential, as...

In the competitive landscape of manufacturing, the integration of various components into a finished product presents significant challenges. Each part, from hardware to software, plays a pivotal role in determining not only the product's functionality but also its market appeal and regulatory compliance.
Manufacturers face significant challenges in integrating diverse components effectively. Failure to address these challenges can lead to non-compliance and diminished market competitiveness. Addressing these challenges is crucial for manufacturers aiming to enhance product performance and ensure safety compliance.
The various components and materials that collectively form a finished product, what do you call the parts you need to construct a product, each play a vital role in its development. These include tangible elements such as hardware and software, as well as what do you call the parts you need to construct a product, including packaging and design specifications.
In product development, what do you call the parts you need to construct a product? Each component plays a critical role, directly impacting functionality, usability, and marketability.
In the realm of medical equipment, for example, the integration of sensors, circuit boards, and software algorithms is essential. This seamless collaboration guarantees not only the safety and efficacy of the product but also adherence to stringent regulatory standards.
The integration of hardware and software is paramount for developing effective medical devices. Notable instances of this integration are found in wearable health technologies that employ sophisticated sensors and software for real-time patient monitoring, demonstrating how integrated systems can enhance patient care and operational efficiency.
As the medical device sector increasingly depends on software to determine clinical value, this effective integration provides a competitive edge, enabling manufacturers to innovate while ensuring compliance.
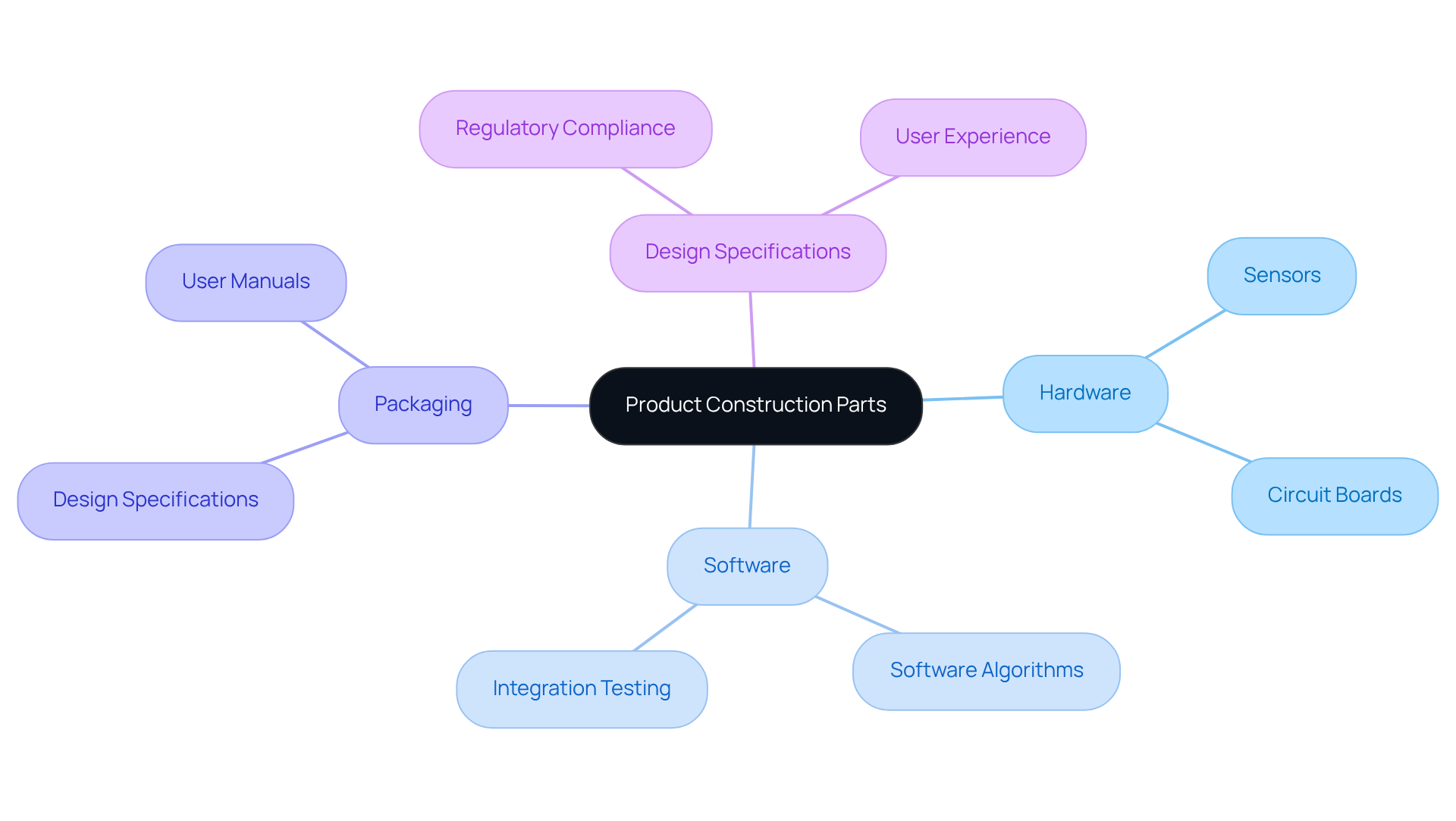
In the development of wearable technology, strategic decisions play a critical role beyond mere assembly. Each element must fulfill its functional role while aligning with market demands. Essential components such as battery life, sensor accuracy, and user interface design significantly influence consumer expectations and satisfaction. These components impact performance and regulatory compliance, especially in healthcare, where safety is non-negotiable.
As the market evolves, manufacturers must prioritize these elements to ensure their products meet user needs while adhering to stringent industry regulations. Common mistakes in manufacturing tests can lead to significant delays, making the application of best practices essential for quality and efficiency in electronic product development.
Leveraging innovative embedded systems design, such as FPGA technology and AI, can enhance battery life and overall performance in wireless medical devices. Therefore, it is crucial for manufacturers to stay ahead of technological advancements while navigating the EU's new requirements for MDR/IVDR devices. Manufacturers who fail to adapt risk not only their market position but also the safety and satisfaction of their users.
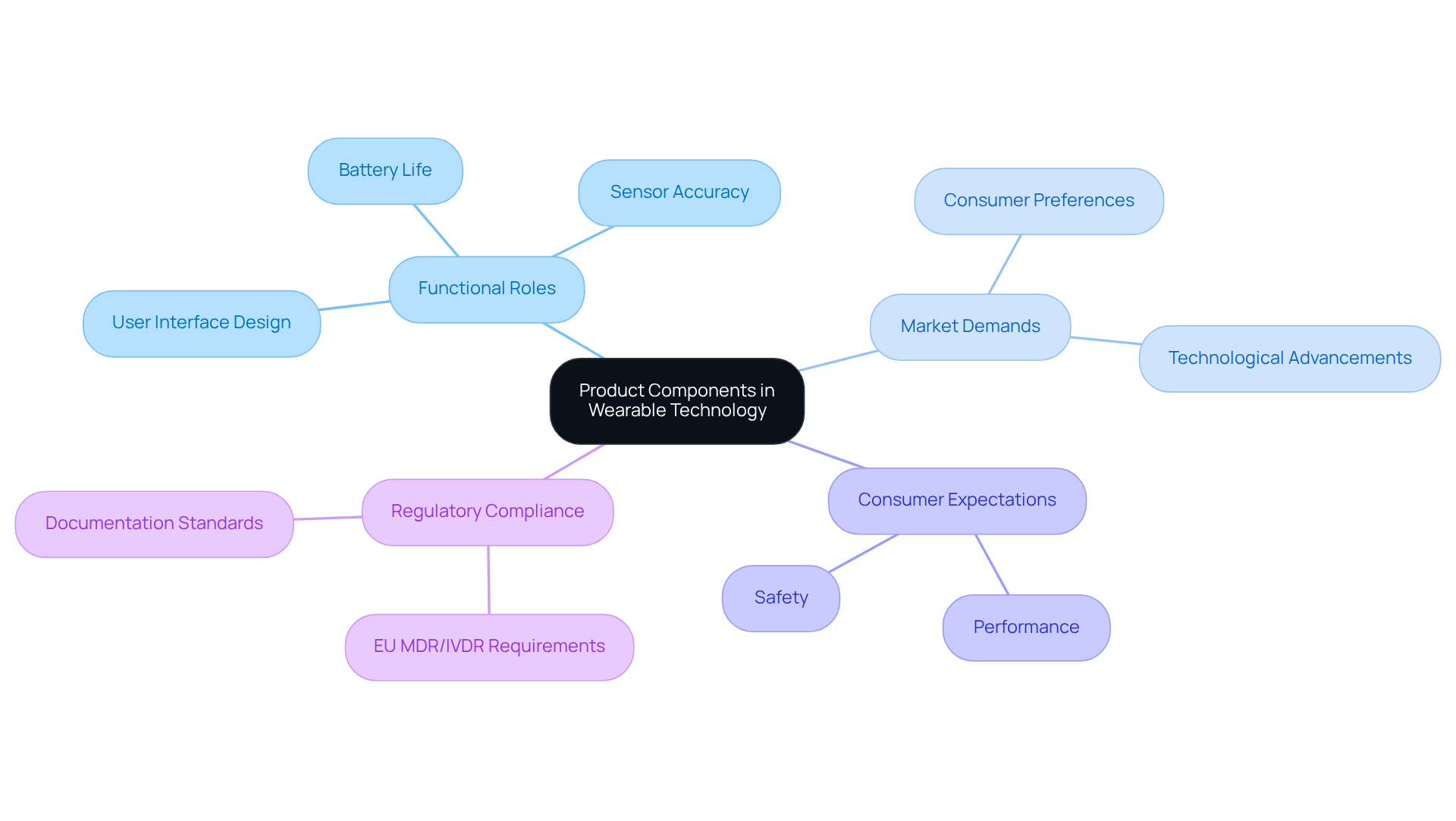
The evolution of product development, particularly in the medical device sector, can be traced back to the transformative changes initiated by the Industrial Revolution. This period marked a significant shift towards mass manufacturing and the use of interchangeable parts, enabling manufacturers to produce components that were not only easily replaceable but also more efficient and cost-effective.
As technology advanced, the incorporation of electronics and software into products revolutionized traditional components, enhancing their functionality and efficiency. For instance, the evolution of medical instruments has transitioned from simple mechanical components to sophisticated electronic systems that greatly improve functionality and guarantee patient safety.
Voler Systems is at the forefront of this transformation, leveraging its expertise in AI-driven engineering and design development to create compliant and reliable electronic products. Additionally, Voler Systems is particularly experienced in designing wearable and IoT devices, which are essential in modern healthcare.
However, this shift also introduces complexities in ensuring product reliability and patient safety. Consequently, companies must innovate continuously to meet the evolving demands of the healthcare sector. Understanding this historical context is essential for navigating the complexities of modern medical device innovation and ensuring compliance with industry standards.
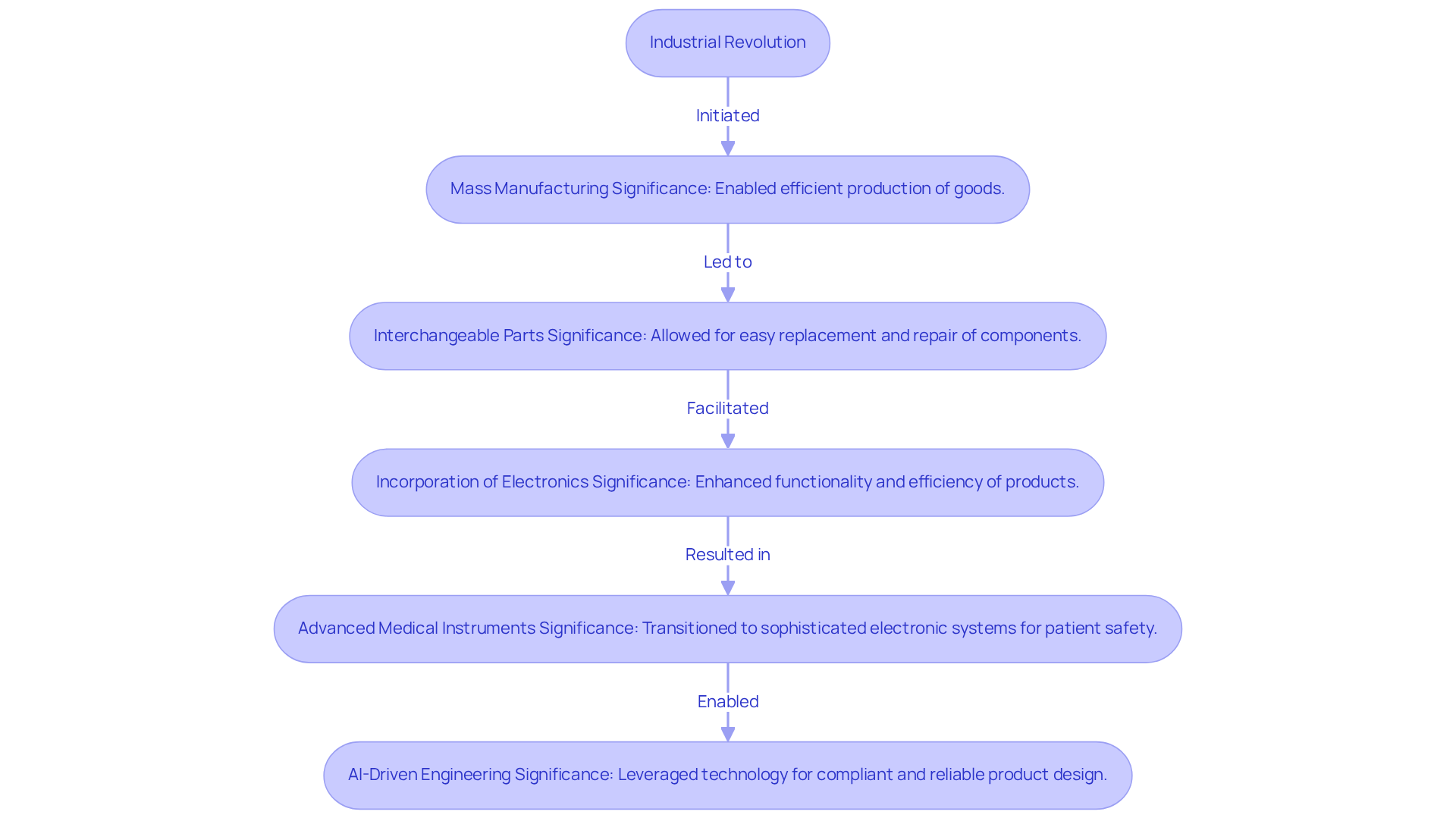
In the competitive landscape of medical equipment, understanding what do you call the parts you need to construct a product is essential for success. Key characteristics of what do you call the parts you need to construct a product include functionality, durability, compatibility, and cost-effectiveness. The characteristics of a product can vary significantly based on what do you call the parts you need to construct a product, the manufacturing processes employed, and the design choices made. In this field, what do you call the parts you need to construct a product that must not only meet stringent regulatory standards but also be designed for ease of use and patient comfort?
Voler Systems emphasizes the importance of compliance documentation, offering guidance on ISO 13485 standards for quality management systems in medical instruments. This support is crucial for ensuring product quality and safety. Furthermore, the incorporation of Artificial Intelligence (AI) is transforming medical equipment design. AI enables improved functionality and customized solutions that enhance performance. For instance, AI-driven analytics can optimize device performance and patient outcomes by providing real-time data insights.
By understanding these characteristics, product developers can better align what do you call the parts you need to construct a product with specific healthcare applications, leading to enhanced product success and improved patient outcomes. This shift towards AI integration not only streamlines design processes but also significantly improves patient care outcomes.
A comprehensive understanding of a product's essential components is critical for successful construction and market relevance. Each part, from hardware to software, plays a crucial role in shaping the product's functionality, usability, and market appeal. Without a clear grasp of these components, manufacturers risk falling behind in innovation and market relevance. Recognizing the significance of these components is essential for manufacturers who wish to innovate and comply with industry standards, especially in the medical device sector where safety is crucial.
This article has highlighted critical insights into the integration of various components, such as the importance of battery life and sensor accuracy in wearable technology. The historical evolution of product parts underscores the transition from mechanical to electronic systems, emphasizing the need for continuous innovation. Additionally, the role of AI in enhancing product design and patient outcomes showcases the dynamic nature of product development in today's competitive landscape.
Ultimately, understanding the essential components of product construction goes beyond assembly; it requires strategic alignment with market demands and regulatory standards. Manufacturers are encouraged to stay informed about technological advancements and best practices to ensure their products not only meet user needs but also contribute positively to the industry. Neglecting these elements may result in missed opportunities for innovation and market leadership. By prioritizing these elements, companies can enhance their market position and ensure the safety and satisfaction of their users, paving the way for future innovations in product development.
What are product construction parts?
Product construction parts refer to the various components and materials that collectively form a finished product, including hardware, software, packaging, and design specifications.
Why are product construction parts important?
Each component plays a critical role in product development, directly impacting functionality, usability, and marketability.
Can you give an example of product construction parts in medical equipment?
In medical equipment, product construction parts include sensors, circuit boards, and software algorithms, which are essential for ensuring safety, efficacy, and compliance with regulatory standards.
How does the integration of hardware and software affect medical device development?
The integration of hardware and software is crucial for developing effective medical devices, as seen in wearable health technologies that use sophisticated sensors and software for real-time patient monitoring.
What advantage does effective integration of components provide to manufacturers in the medical device sector?
Effective integration provides manufacturers with a competitive edge by enabling innovation while ensuring compliance with regulatory standards.
