Introduction
The integration of application-specific integrated circuits (ASICs) is fundamentally transforming the healthcare landscape, providing customized solutions that significantly improve the performance and dependability of medical devices. As the demand for sophisticated healthcare instruments rises, understanding the strategic integration of ASICs becomes crucial for engineers and manufacturers alike.
Engineers and manufacturers often face hurdles in ASIC design and compliance, which can impede progress. Failure to address these challenges may hinder advancements in patient care and device efficacy. Addressing these challenges is essential for fostering innovation and ensuring that medical devices continue to enhance patient outcomes.
Define Application-Specific Integrated Circuits (ASICs) and Their Role in Medical Devices
Application specific integrated circuits (ASICs) are essential components in the healthcare equipment industry, designed specifically for targeted applications rather than general use. For example, ASICs are commonly found in wearable health trackers, heart pumps, and diagnostic instruments. They enable precise data processing and real-time monitoring.
Producers face challenges in meeting the growing demand for specialized healthcare instruments. This necessity positions the application specific integrated circuit as vital for developing innovative solutions that improve patient outcomes and ensure equipment reliability.
Voler Systems leverages its expertise in ASIC design to improve battery life in wireless medical devices using ultra-low power techniques. Their ability to integrate AI-driven engineering ensures these products meet current healthcare demands while being designed for future advancements in intelligent healthcare.
Recent advancements in application specific integrated circuit technology, including ultra-low-power designs and improved integration capabilities, highlight their significance in advancing healthcare technology.
Industry leaders emphasize that the growing trend toward customized application specific integrated circuit solutions is fostering innovation, making these chips essential to modern healthcare technology development.
Implement Design Strategies for ASIC Integration in Compliance with Medical Standards
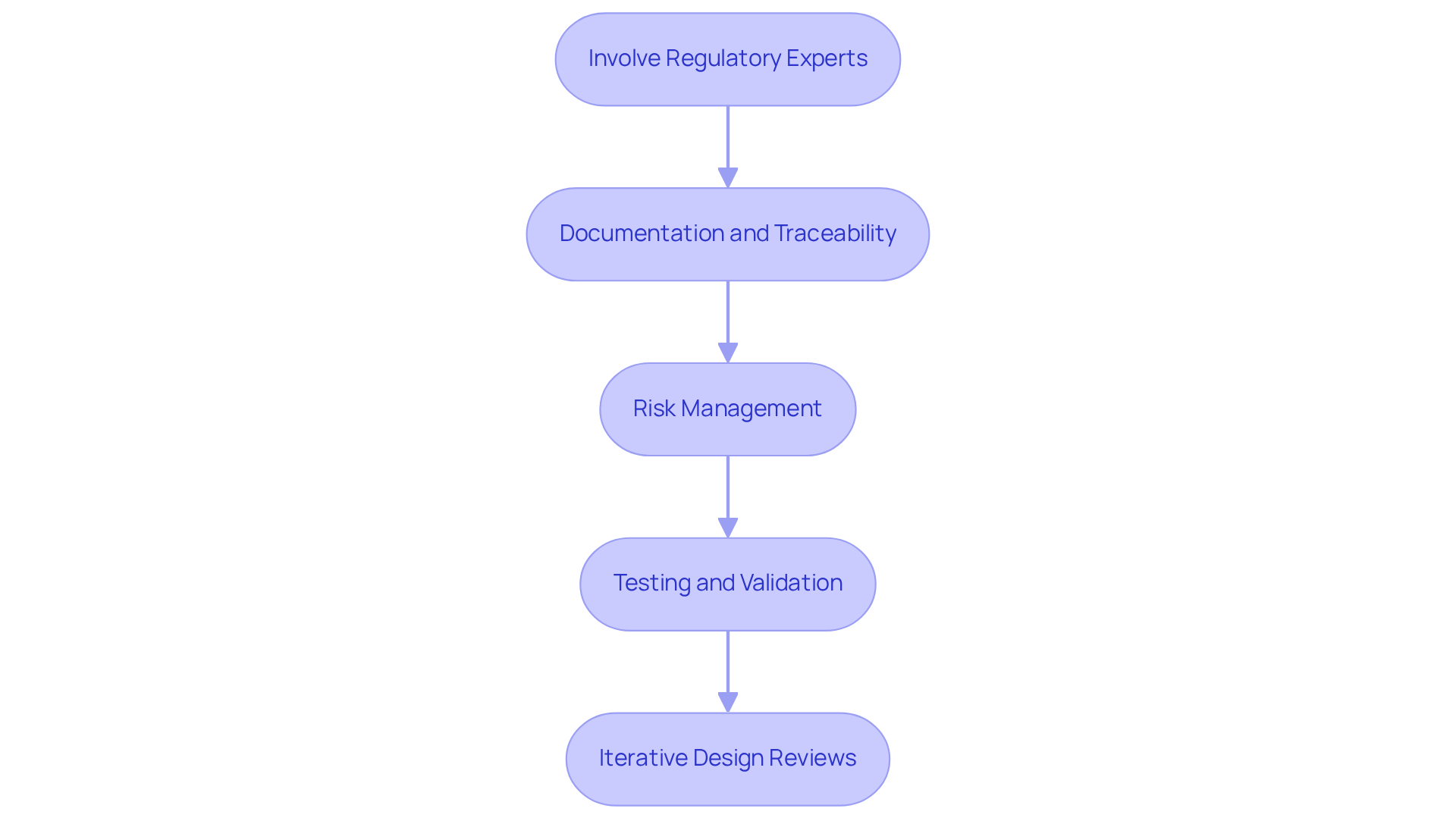
Incorporating application specific integrated circuits into medical devices presents significant challenges that require strategic planning and adherence to medical standards. Key strategies include:
- Early involvement of regulatory experts is crucial to ensure that all aspects of the application specific integrated circuit adhere to necessary standards, facilitating smoother approvals during the planning phase.
- Documentation and Traceability: Keeping thorough records of the development process-including specifications, testing protocols, and compliance checks-is essential for facilitating audits and regulatory reviews.
- The application specific integrated circuit is essential for efficient processing. Risk Management: Implementing a robust risk management framework helps identify potential hazards associated with application specific integrated circuit functionality and outlines effective mitigation strategies, aligning with evolving regulatory expectations.
- Application specific integrated circuit. Testing and Validation: Conducting rigorous testing under various conditions is vital to validate the performance of the application specific integrated circuit, ensuring it meets stringent safety and efficacy requirements.
- Iterative Design Reviews: Consistently assessing design iterations with multidisciplinary teams facilitates prompt detection and resolution of compliance concerns, improving the overall dependability and safety of the healthcare instruments.
Neglecting these strategies could compromise the reliability and safety of healthcare instruments, ultimately impacting patient outcomes.
Evaluate Benefits and Trade-offs of ASICs in Medical Device Development
When considering ASICs for medical device development, evaluating their benefits and trade-offs is essential for informed decision-making:
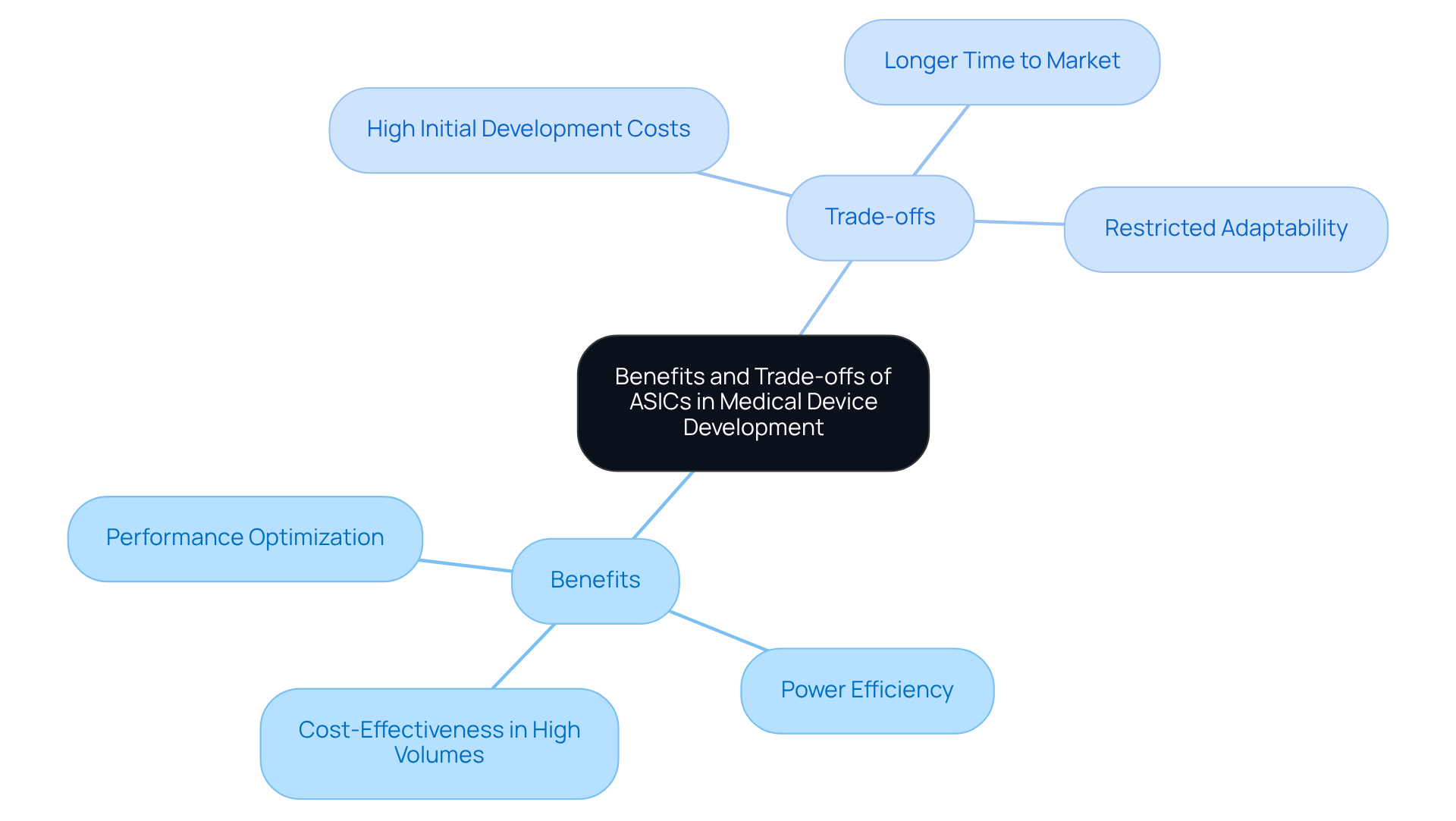
Benefits:
Trade-offs:
- High Initial Development Costs: The design and fabrication of ASICs involve considerable upfront investment, which may not be practical for low-volume applications. This financial barrier can deter smaller companies from pursuing ASIC solutions. Lengthy development cycles for ASIC chips can delay time-to-market for specialized solutions, a challenge that Voler Systems addresses through innovative processes.
- Longer Time to Market: The ASIC development process is often lengthy, potentially delaying product launches compared to off-the-shelf components. This extended timeline poses a significant disadvantage in fast-paced medical markets, particularly for companies seeking rapid FDA approval.
- Restricted Adaptability: Once produced, the application specific integrated circuit cannot be reprogrammed or altered, which poses difficulties if specifications change after manufacturing. This inflexibility requires careful planning and validation in the design phase. As noted by Voler Systems, this characteristic is especially beneficial for healthcare instruments that need to evolve to meet new functionalities or regulatory standards.
By thoughtfully assessing these advantages and compromises, engineers can make educated choices about incorporating application specific integrated circuits into their healthcare instruments, ensuring that they satisfy both performance and regulatory criteria.
Adopt Proven Implementation Techniques for Successful ASIC Integration
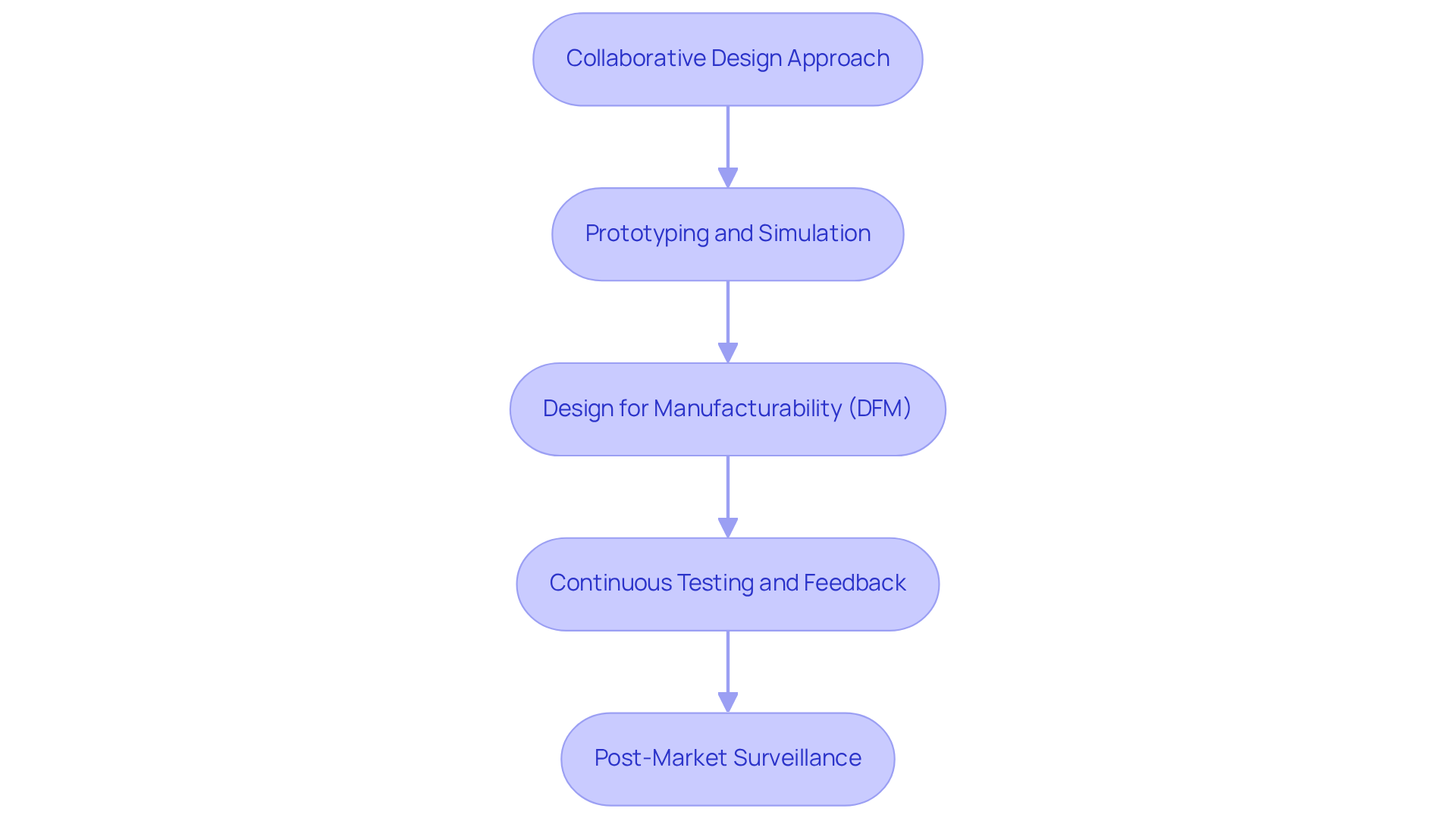
To achieve successful ASIC integration in medical devices, engineers must adopt a structured approach that emphasizes collaboration and precision:
- Collaborative Design Approach: Encourage collaboration between hardware and software teams to ensure seamless integration of ASICs with other system components. This approach fosters innovation while reducing the chances of misalignment among components.
- Prototyping and Simulation: Utilize prototyping tools and simulation software to assess ASIC models prior to full-scale production. This practice allows for the early detection of issues, thereby improving overall project outcomes. Effective prototyping has been shown to reduce development time by as much as 30%, thereby significantly improving project efficiency.
- Design for Manufacturability (DFM): Integrate DFM principles to streamline the manufacturing process, minimize costs, and improve product reliability. By concentrating on manufacturability during the development phase, teams can prevent expensive redesigns later in the process.
- Continuous Testing and Feedback: Establish a continuous testing regime throughout the design and production phases. This ongoing feedback loop enables teams to make real-time adjustments, ensuring that the final product meets quality and performance standards.
- Post-Market Surveillance: Create a comprehensive post-market monitoring strategy to observe the performance of ASIC-integrated products in real-world environments. This strategy is crucial for maintaining compliance with regulatory standards and ensuring patient safety.
By implementing these techniques, engineers can ensure that their application specific integrated circuit products not only meet but exceed industry standards for quality and reliability. Ultimately, these strategies not only enhance product quality but also ensure compliance with the rigorous demands of the healthcare sector.
Conclusion
In an era where patient outcomes hinge on technological advancements, the role of application-specific integrated circuits (ASICs) in the medical device sector is more critical than ever. Their ability to enable precise data processing and real-time monitoring is essential, as achieving optimal patient outcomes becomes increasingly challenging in today's technology-driven healthcare landscape. As the demand for specialized medical instruments continues to rise, the strategic integration of ASICs becomes vital for both manufacturers and healthcare providers.
Throughout the article, key strategies for successful ASIC integration have been outlined, including the importance of early regulatory involvement, thorough documentation, and a robust risk management framework. The benefits of ASICs, such as performance optimization, power efficiency, and cost-effectiveness in large-scale production, are balanced against the trade-offs of high initial costs and longer development cycles. Engineers can effectively navigate these challenges by employing proven implementation techniques such as collaborative design and continuous testing, which ensure compliance and enhance product reliability.
The future of healthcare technology hinges on the strategic adoption of ASICs, shaping the landscape of medical devices to better serve patients and providers alike. Engaging with current trends in ASIC technology and continuously refining design practices will be essential for those looking to lead in this dynamic field.
Frequently Asked Questions
What are Application-Specific Integrated Circuits (ASICs)?
ASICs are specialized components designed for specific applications rather than general use, commonly utilized in the healthcare equipment industry.
Where are ASICs commonly used in medical devices?
ASICs are often found in wearable health trackers, heart pumps, and diagnostic instruments, enabling precise data processing and real-time monitoring.
What challenges do producers face regarding ASICs in healthcare?
Producers face challenges in meeting the growing demand for specialized healthcare instruments, which positions ASICs as vital for developing innovative solutions that improve patient outcomes and ensure equipment reliability.
How does Voler Systems utilize ASIC technology?
Voler Systems leverages its expertise in ASIC design to enhance battery life in wireless medical devices through ultra-low power techniques and integrates AI-driven engineering for future advancements in intelligent healthcare.
What recent advancements have been made in ASIC technology?
Recent advancements include ultra-low-power designs and improved integration capabilities, underscoring the significance of ASICs in advancing healthcare technology.
Why are customized ASIC solutions important in the healthcare industry?
The growing trend toward customized ASIC solutions fosters innovation, making these chips essential to the development of modern healthcare technology.
List of Sources
- Define Application-Specific Integrated Circuits (ASICs) and Their Role in Medical Devices
- Application Specific Integrated Circuit Market Report 2026 (https://researchandmarkets.com/reports/5792982/application-specific-integrated-circuit-market?srsltid=AfmBOoq_G-6qL26mHcyAw-HWCaJkxESSf1N8BvsvMD7giQMPkKoVH_BU)
- Custom ASICs designed for advanced medical devices (https://csem.ch/en/technical-focus/asics-for-the-edge/custom-asics-optimized-medical-devices)
- Application Specific Integrated Circuit Market Size Report, 2034 (https://fortunebusinessinsights.com/application-specific-integrated-circuit-market-104779)
- NANS 2026 - ASICs for neuromodulation and recording - ICsense (https://icsense.com/nans-2026-asics-for-neuromodulation-and-recording)
- ASIC Chip Market Size, Trends & YoY Growth Rate, 2026-2033 (https://coherentmarketinsights.com/industry-reports/asic-chip-market)
- Implement Design Strategies for ASIC Integration in Compliance with Medical Standards
- Regulatory Innovation as a Catalyst: How New Compliance Strategies Are Speeding Up Healthcare Technology Adoption - MedCity News (https://medcitynews.com/2026/02/regulatory-innovation-as-a-catalyst-how-new-compliance-strategies-are-speeding-up-healthcare-technology-adoption)
- FDA’s AI Device Guidelines Evolve: How Medtech Can Adapt (https://mddionline.com/artificial-intelligence/fda-s-ai-device-regulations-key-updates-compliance-strategies-for-medtech-success)
- 2026 Commences with a Look Back, a Look Forward and New Legislation and Guidance (https://emergobyul.com/news/2026-commences-look-back-look-forward-and-new-legislation-and-guidance)
- Top Medical Device Regulations to Watch in 2026 (https://xtalks.com/top-medical-device-regulations-to-watch-in-2026-4549)
- Six Strategies for Medical Device Compliance in 2026 | White Paper (https://intertek.com/resources/white-papers/2026/medical-six-strategies-for-device-compliance)
- Evaluate Benefits and Trade-offs of ASICs in Medical Device Development
- FPGA Companies vs. ASICs: Key Insights for Medical Device Makers (https://volersystems.com/blog/fpga-companies-vs-asics-key-insights-for-medical-device-makers)
- When does an ASIC make sense? | imec (https://imec-int.com/en/articles/when-does-asic-make-sense-your-product)
- Electrocardiography ADC architectures, trade-offs and use cases (https://medicaldesignandoutsourcing.com/ensilica-electrocardiography-ecg-adc-architecture-asics)
- ASIC Chip Market Size, Trends & YoY Growth Rate, 2026-2033 (https://coherentmarketinsights.com/industry-reports/asic-chip-market)
- Adopt Proven Implementation Techniques for Successful ASIC Integration
- MedTech Trends Shaping 2026 (https://iqvia.com/blogs/2026/02/medtech-trends-shaping-2026)
- AI/ML Medical Devices: Navigating FDA's Evolving Regulatory Framework in 2026 (https://proximacro.com/news/ai-ml-medical-devices-navigating-fdas-evolving-regulatory-framework-in-2026)
- 4 Medical Device Manufacturing Trends for 2026 (https://mastercontrol.com/gxp-lifeline/2026-medical-device-manufacturing-trends-life-sciences)
- Medtech 2026: Complexity, Bottlenecks And Cybersecurity Among Key Regulatory Concerns (https://insights.citeline.com/medtech-insight/perspectives/medtech-2026-complexity-bottlenecks-and-cybersecurity-among-key-regulatory-concerns-PIVHDSC6BBGRPAPEAUHGMJGIPE)
- The Next Frontier: Medical Device Innovation Unveiled (https://medtechbusinessrevieweurope.com/news/the-next-frontier-medical-device-innovation-unveiled-nwid-586.html)





