4 Best Practices for Effective Medical Product Development
Introduction Navigating the complexities of medical product development necessitates a...

The landscape of medical device engineering is experiencing a significant shift, driven by the demand for innovative solutions that prioritize patient safety and regulatory compliance.
As healthcare technology evolves, it is crucial for manufacturers to understand best practices in hardware product engineering to effectively navigate the complexities of product development.
With the rapid integration of AI and the emergence of new regulatory standards, companies face the challenge of ensuring their products not only meet market demands but also comply with stringent safety protocols.
This article explores the essential stages of hardware product engineering, providing insights into effective strategies that can improve design efficiency, compliance, and project management within the dynamic medical device sector.
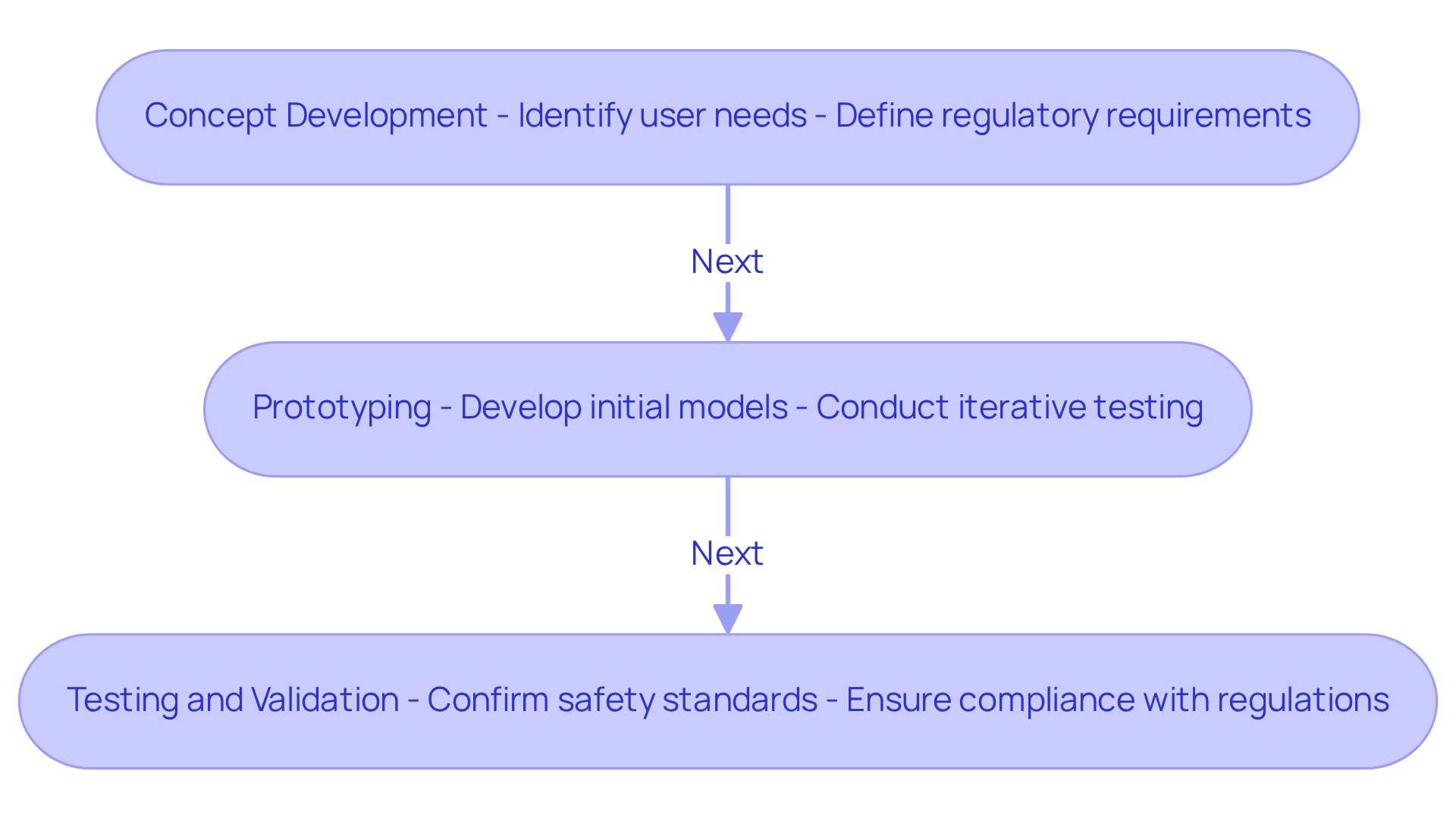
Hardware engineering in medical equipment entails a meticulous process of design, creation, and testing specifically tailored for medical applications. This multifaceted approach encompasses several key stages:
Staying abreast of the is essential. The integration of AI and machine learning is revolutionizing how products are designed and tested, enhancing their adaptability and performance. Voler Systems utilizes of intelligent healthcare. According to industry forecasts, the and is projected to expand at a CAGR of 10.1% from 2024 to 2029, reaching US$14.7 billion by 2029. By adhering to , including a , companies can navigate the complexities of health-related product development, ensuring timely market entry and high-quality outcomes.
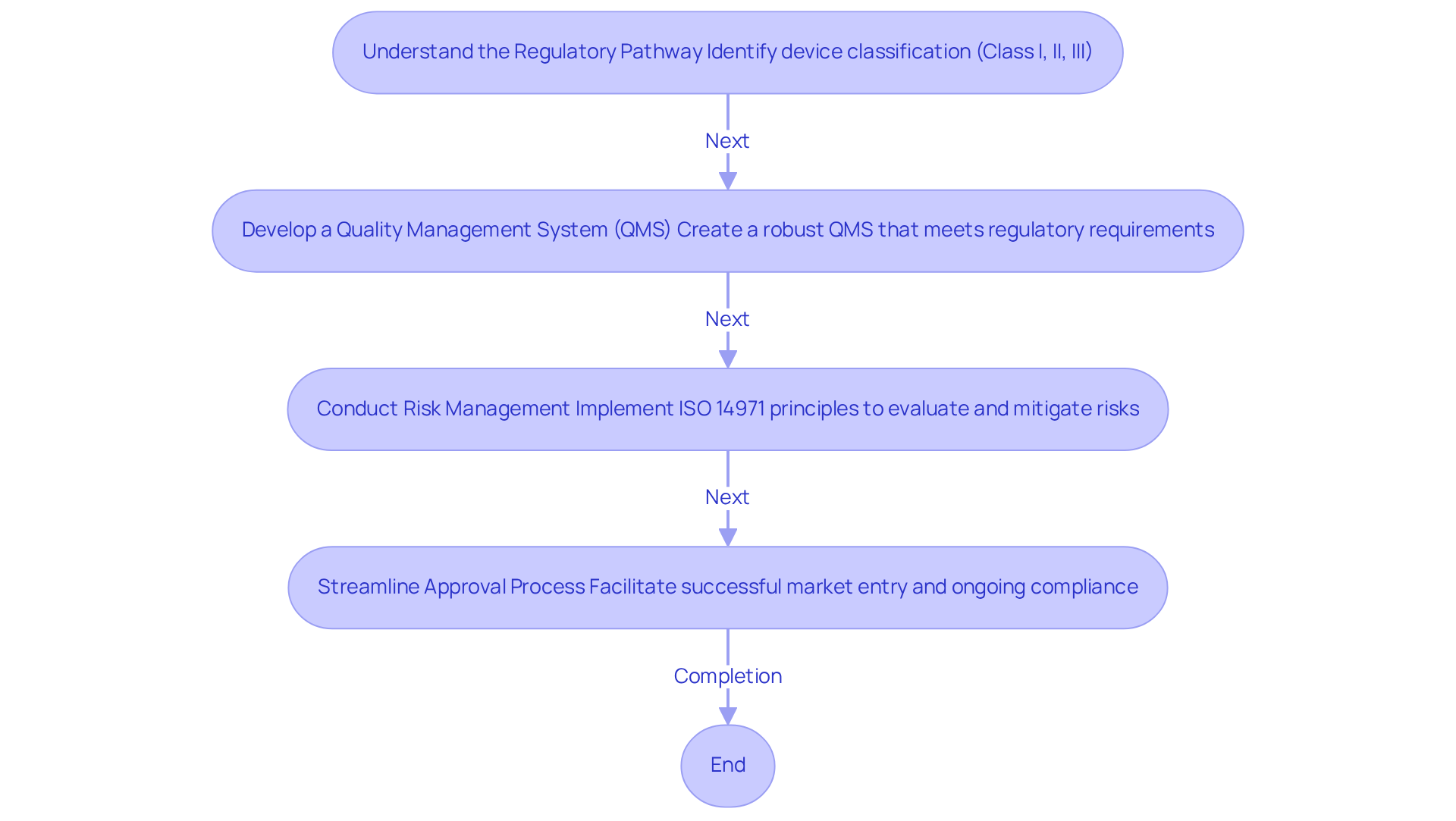
Adherence to regulatory standards is essential in the healthcare equipment industry, particularly regarding the FDA's , which governs the Quality System Regulation (QSR). Effective February 2, 2026, the FDA will implement a new Regulation, highlighting the necessity for manufacturers to align their practices with . To ensure compliance, manufacturers should concentrate on the following key practices:
By adhering to these , companies can streamline the approval process, improve product safety, and reduce the risk of costly delays, ultimately facilitating successful market entry and ongoing compliance. is dedicated to providing to manufacturers of healthcare equipment, assisting them in navigating to ensure that their innovative technologies meet compliance standards and support reliable electronic equipment design. For instance, has effectively aided clients in achieving compliance through tailored QMS design and strategies, demonstrating a commitment to excellence in the healthcare sector.
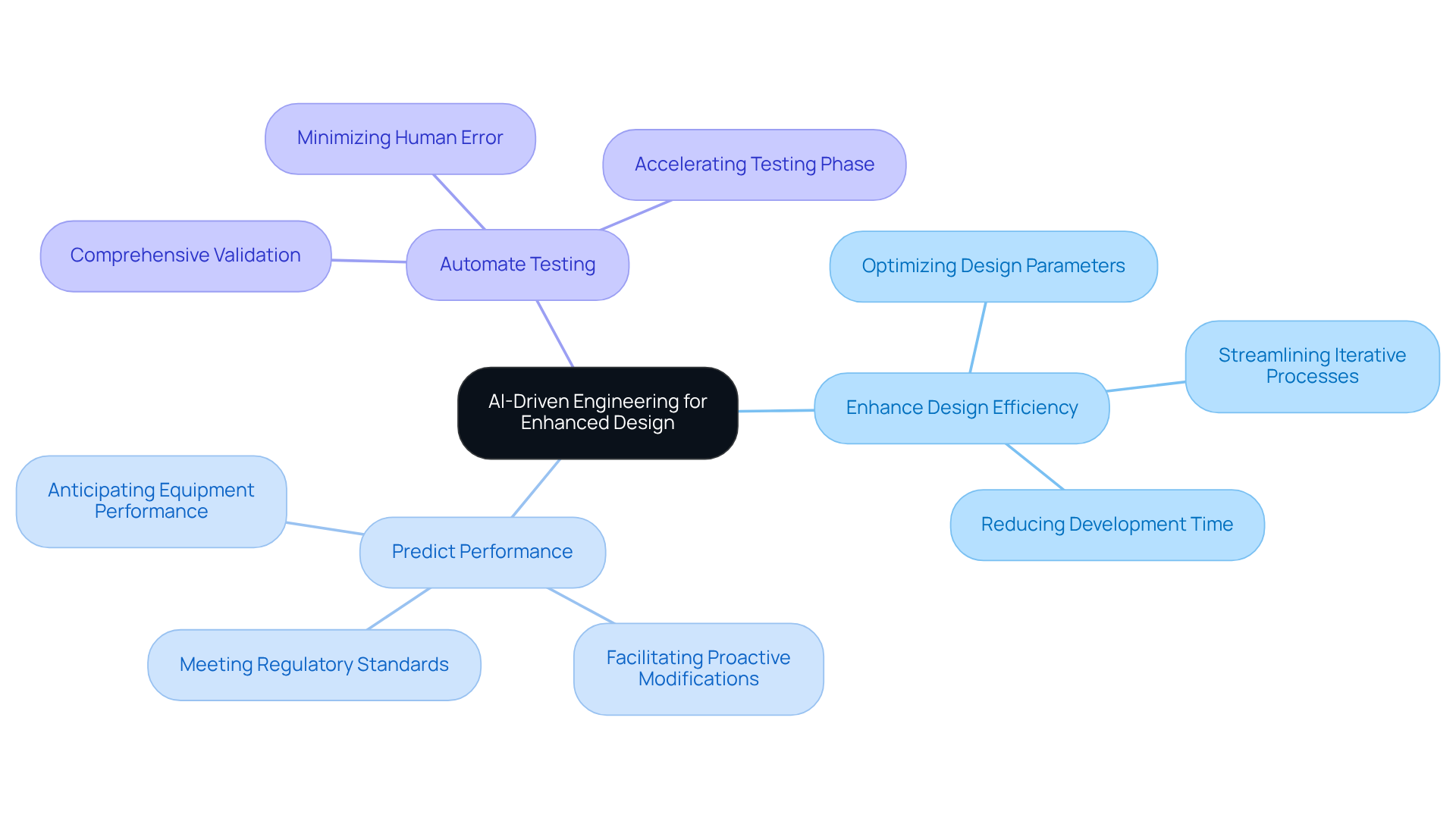
is revolutionizing the design of by markedly enhancing efficiency and reliability. By leveraging AI technologies, engineers can achieve several key advancements:
For instance, AI tools can analyze extensive datasets from previous designs to identify patterns that correlate with successful outcomes. This data-driven approach not only shortens timelines but also enhances product reliability, ultimately leading to . As the healthcare sector continues to evolve, the integration of AI in engineering practices will be pivotal in fostering innovation and efficiency.
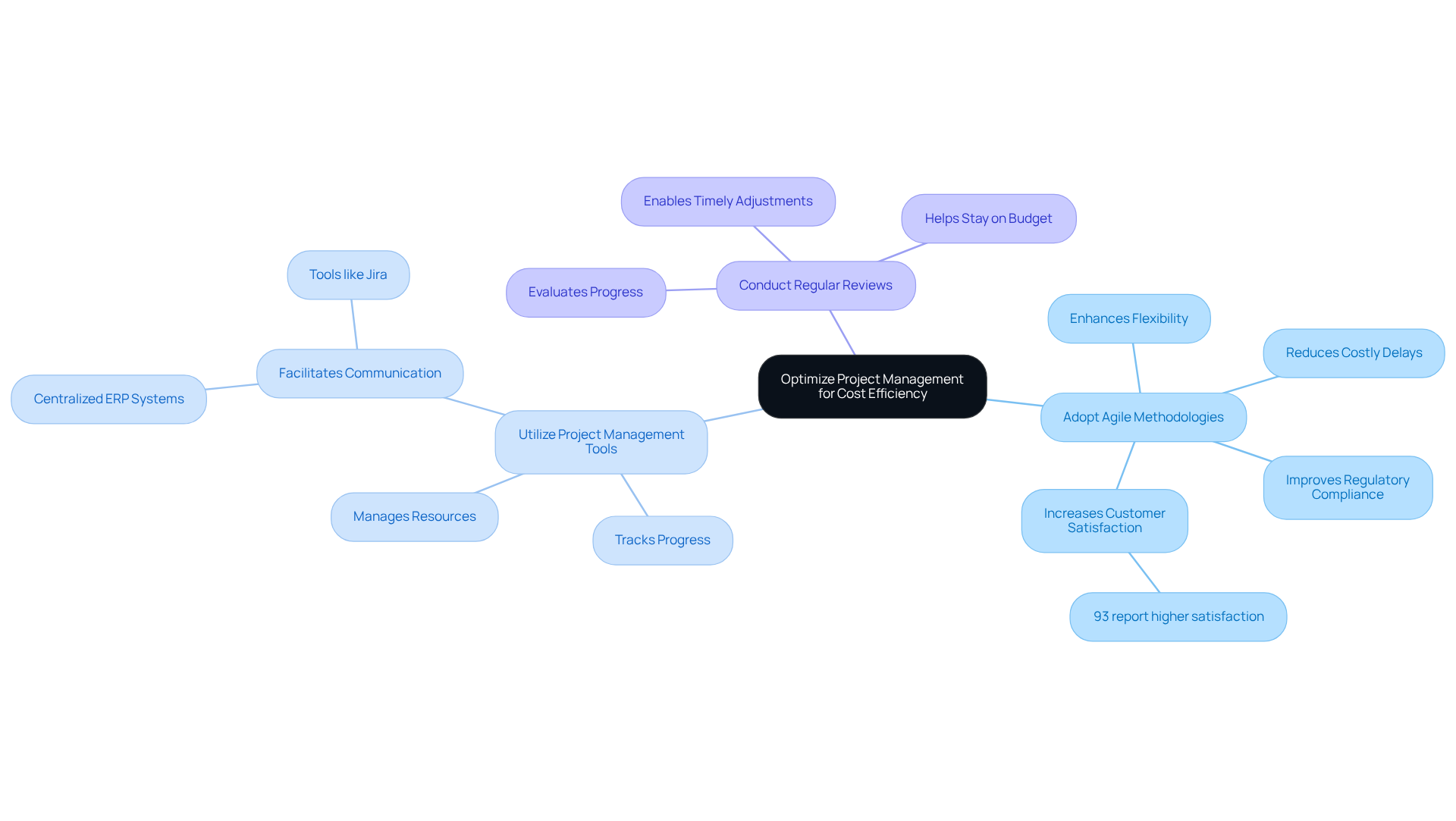
Efficient is crucial for the success of . To optimize for , consider the following strategies:
By focusing on these strategies, teams can enhance collaboration, minimize waste, and ensure that projects are delivered on time and within financial constraints. This ultimately leads to in the competitive .
In the field of hardware product engineering for medical devices, a comprehensive approach is essential, integrating design, prototyping, testing, and compliance. By prioritizing an understanding of user needs and regulatory requirements from the outset, organizations can establish a robust foundation for successful product development. The careful stages of prototyping and validation ensure that devices not only fulfill technical specifications but also comply with critical safety standards, ultimately protecting patient health.
Key insights from this discussion underscore the necessity of adopting best practices in hardware engineering. Leveraging AI-driven technologies can enhance design efficiency and predictive performance. Furthermore, effective project management strategies, including Agile methodologies and advanced project management tools, are vital for optimizing cost efficiency and ensuring timely market entry. Compliance with regulatory standards, such as the forthcoming FDA Quality Management System Regulation, further highlights the importance of adherence in facilitating successful product launches.
As the medical device landscape evolves, embracing these best practices is imperative for manufacturers seeking to innovate while ensuring safety and compliance. The integration of AI and robust project management techniques not only streamlines the development process but also positions companies to excel in a competitive market. By prioritizing these strategies, stakeholders can significantly advance healthcare technologies that improve patient outcomes and propel the industry forward.
What is hardware product engineering in medical devices?
Hardware product engineering in medical devices involves a meticulous process of design, creation, and testing specifically tailored for medical applications, encompassing several key stages.
What are the key stages of hardware product engineering?
The key stages include concept development, prototyping, testing and validation, and staying updated on the latest trends in medical equipment development.
What happens during the concept development phase?
The concept development phase focuses on identifying user needs and regulatory requirements, which are crucial for informing the design process and establishing a foundation for successful product creation.
How does prototyping contribute to hardware product engineering?
Prototyping allows engineers to develop initial models to evaluate functionality and design, facilitating iterative testing and refinement to ensure the device meets user expectations and technical specifications.
Can you provide an example of effective prototyping in medical devices?
The development of the HemeChip for rapid blood testing is an example where effective prototyping yielded innovative solutions that address real-world needs.
What is the importance of testing and validation in medical device engineering?
Testing and validation are essential to confirm that the apparatus meets safety and efficacy standards, ensuring compliance with industry regulations and safeguarding patient health.
How does Voler Systems support compliance in medical device development?
Voler Systems manages compliance from prototype to production, which is particularly vital for manufacturers of wearable and IoT solutions.
What trends are influencing the development of medical equipment?
The integration of AI and machine learning is revolutionizing product design and testing, enhancing adaptability and performance in medical devices.
What is the projected growth of the healthcare equipment engineering market?
The global healthcare equipment engineering market was valued at US$8.2 billion in 2023 and is projected to expand at a CAGR of 10.1% from 2024 to 2029, reaching US$14.7 billion by 2029.
What best practices should companies follow in hardware product engineering?
Companies should adhere to best practices that include delivering products within budget and on schedule to navigate the complexities of health-related product development, ensuring timely market entry and high-quality outcomes.
