4 Best Practices for Effective PCB Layout Design in Medical Devices
Introduction The design of medical devices requires not only innovation but also a...

The demand for high-quality printed circuit boards (PCBs) in medical devices has surged to unprecedented levels. This increase is driven by the critical need for safety, reliability, and compliance with stringent regulatory standards.
Implementing essential PCB quality control methods is vital, as it not only ensures adherence to guidelines such as IPC standards but also enhances the overall performance of healthcare technologies.
As the landscape of medical device manufacturing continues to evolve, manufacturers must effectively navigate the complexities of quality control. This navigation is crucial for minimizing defects and maximizing reliability.
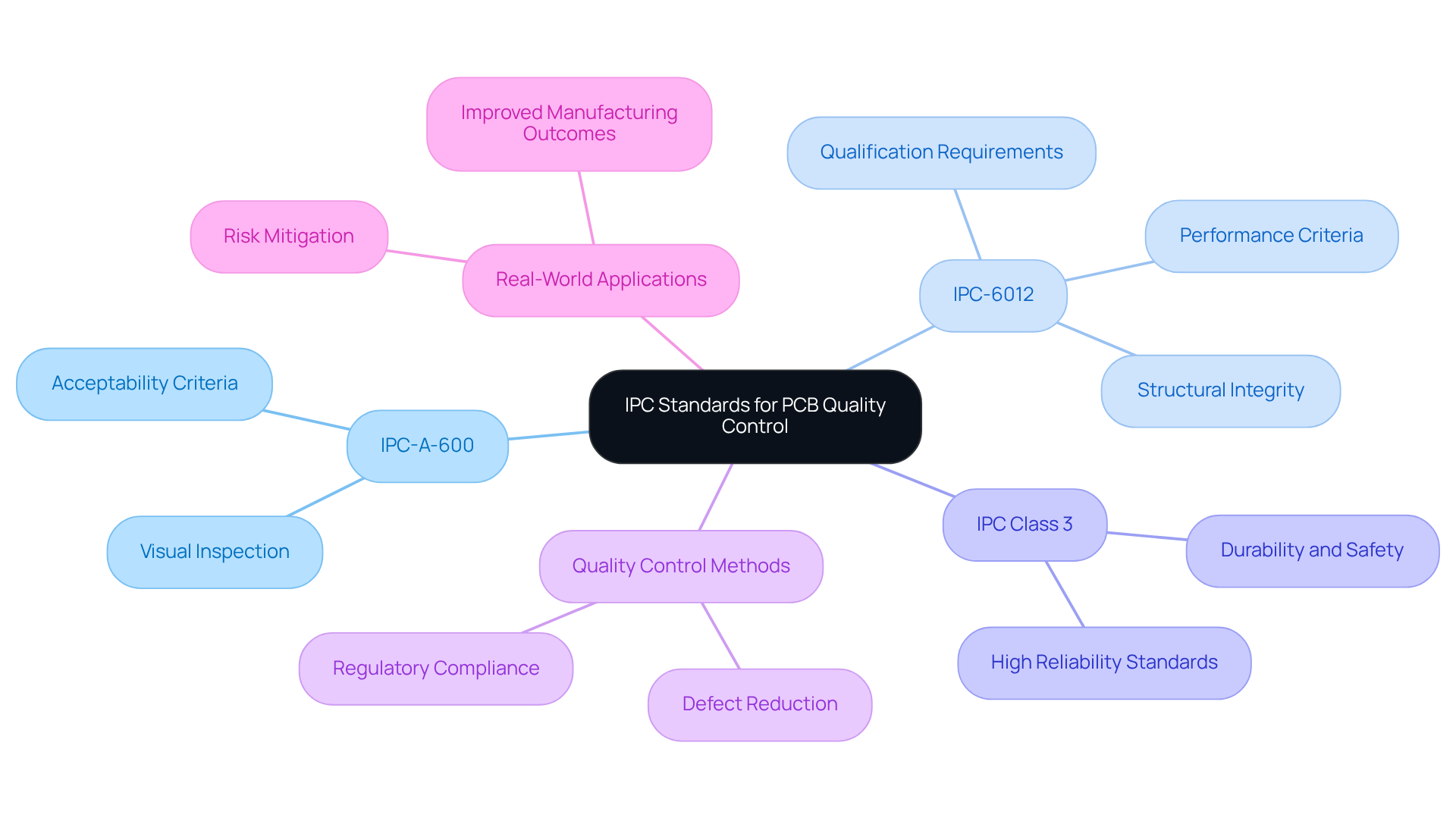
IPC (Institute for Printed Circuits) criteria serve as essential guidelines that establish quality and reliability benchmarks for printed circuit boards (PCBs) utilized in healthcare devices. Key criteria, such as IPC-A-600, which emphasizes visual inspection, and IPC-6012, detailing qualification requirements, are vital for engineers engaged in PCB design and manufacturing. These guidelines ensure that PCB quality control methods are in place to meet the stringent demands of healthcare applications, including durability, safety, and performance across various conditions.
Adhering to IPC guidelines and utilizing PCB quality control methods allows manufacturers to significantly reduce defects, enhance product reliability, and maintain compliance with regulatory standards. For example, IPC Class 3 criteria are frequently applied to healthcare instruments, necessitating the highest level of reliability. A thorough understanding of these classifications empowers engineers to make informed decisions regarding materials, design, and testing protocols, ultimately leading to the development of safer and more effective medical equipment.
Real-world applications of IPC guidelines illustrate their effectiveness in ensuring PCB quality control methods. Organizations that implement adherence to IPC-A-600 and IPC-6012 have reported improved manufacturing outcomes and reduced risks associated with equipment failures. Furthermore, common errors in manufacturing tests, such as insufficient testing protocols or failure to meet qualification requirements, can result in significant delays in product development. At Voler Systems, we leverage our expertise in electronic design services to develop a range of health-related products, including wearable technology and heart pumps, while ensuring compliance with IPC guidelines. As the healthcare equipment market continues to evolve, the importance of these standards in guaranteeing high-quality production processes cannot be overstated.
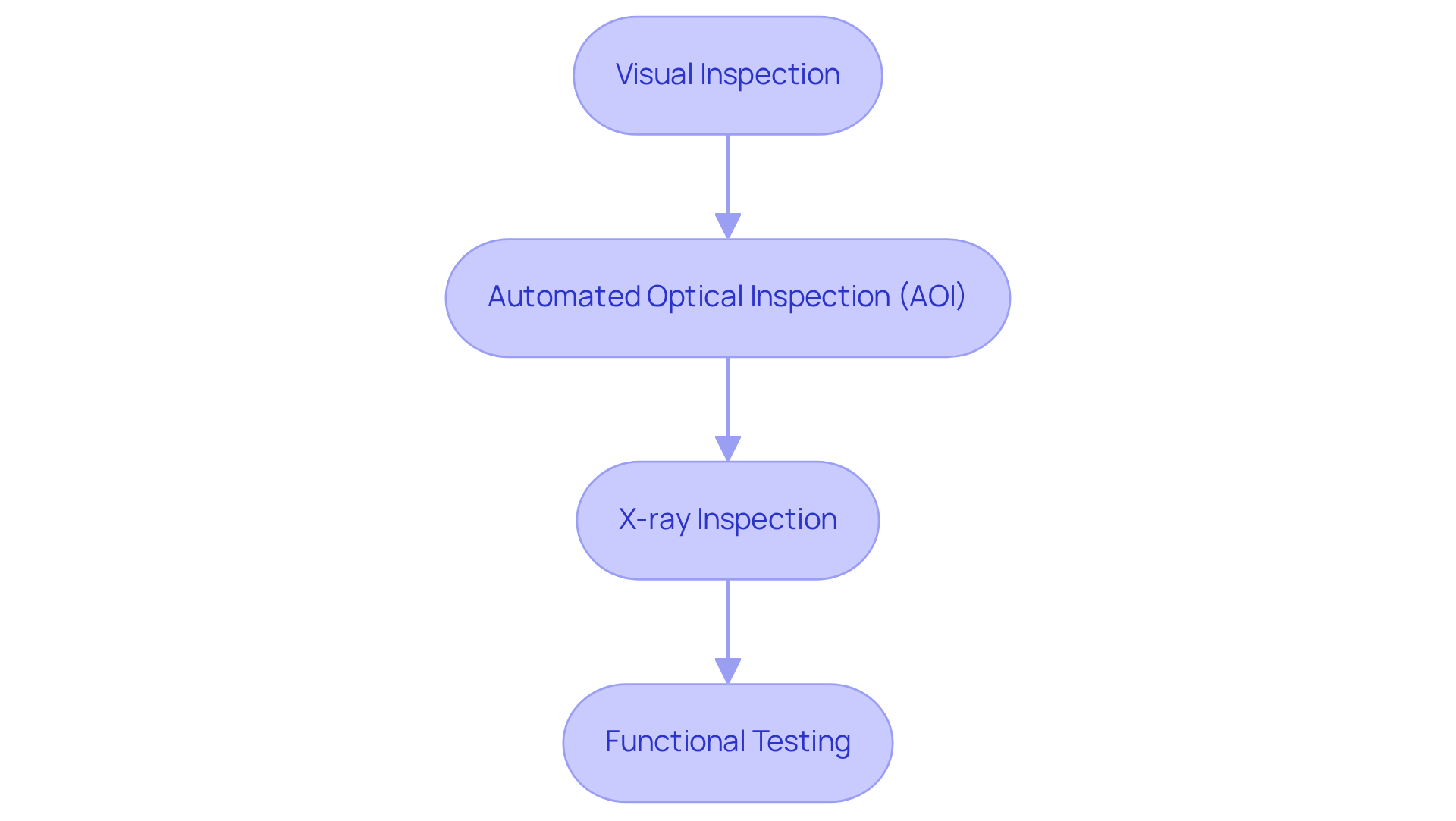
To achieve the highest standards in PCB manufacturing for medical devices, implementing robust PCB quality control methods is essential. This protocol should include a combination of visual inspections, automated optical inspection (AOI), X-ray inspection, and functional testing, each serving a distinct purpose:
Incorporating PCB quality control methods into the production process not only minimizes the chance of flaws but also enhances the reliability and safety of healthcare products, ensuring compliance with the strict regulatory standards of the sector.
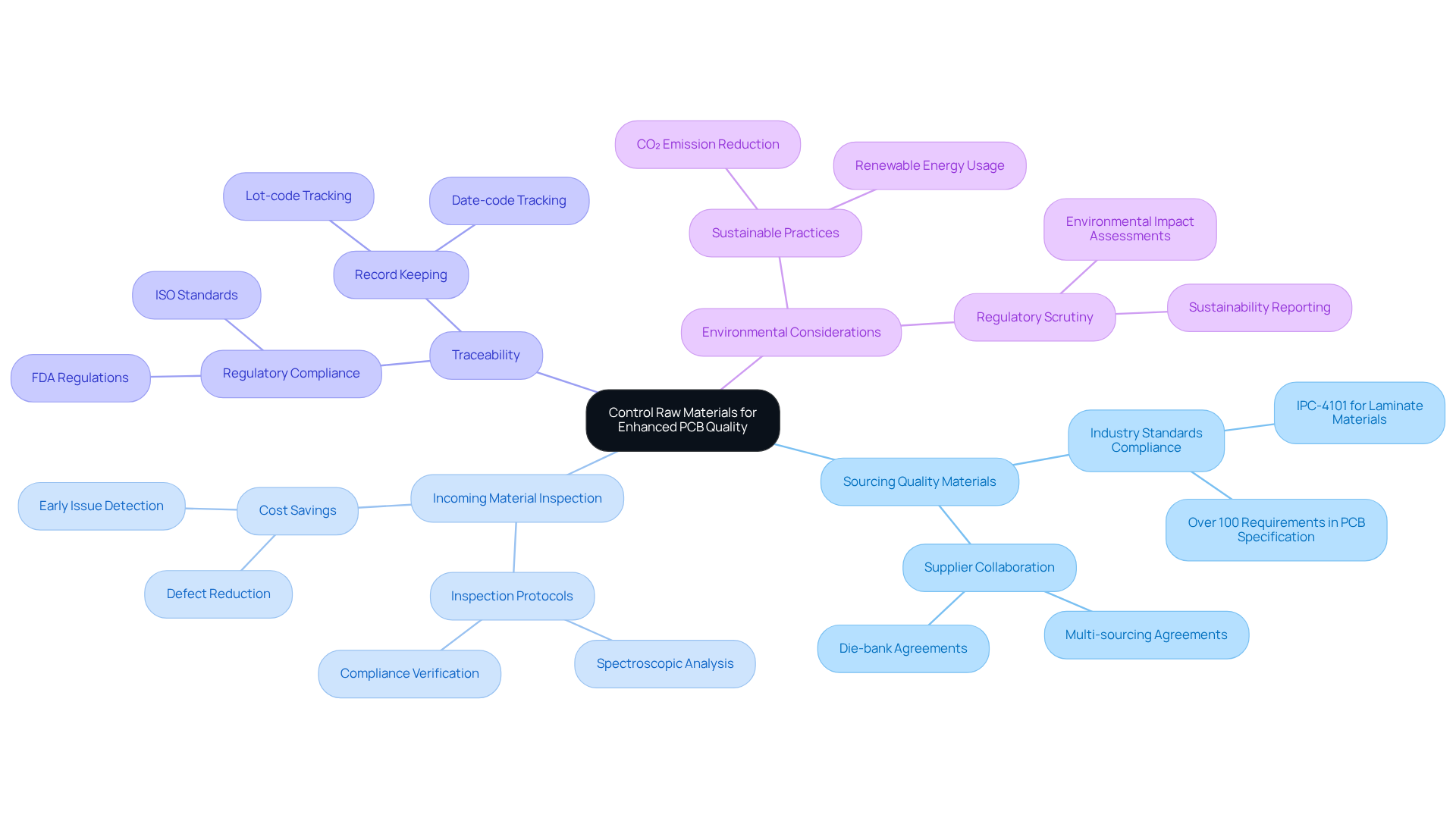
Effective control of raw materials is essential for maintaining high standards through PCB quality control methods in medical devices. This involves several key practices:
By managing raw materials efficiently, manufacturers can improve the overall standard and reliability of their PCBs, resulting in safer medical devices.
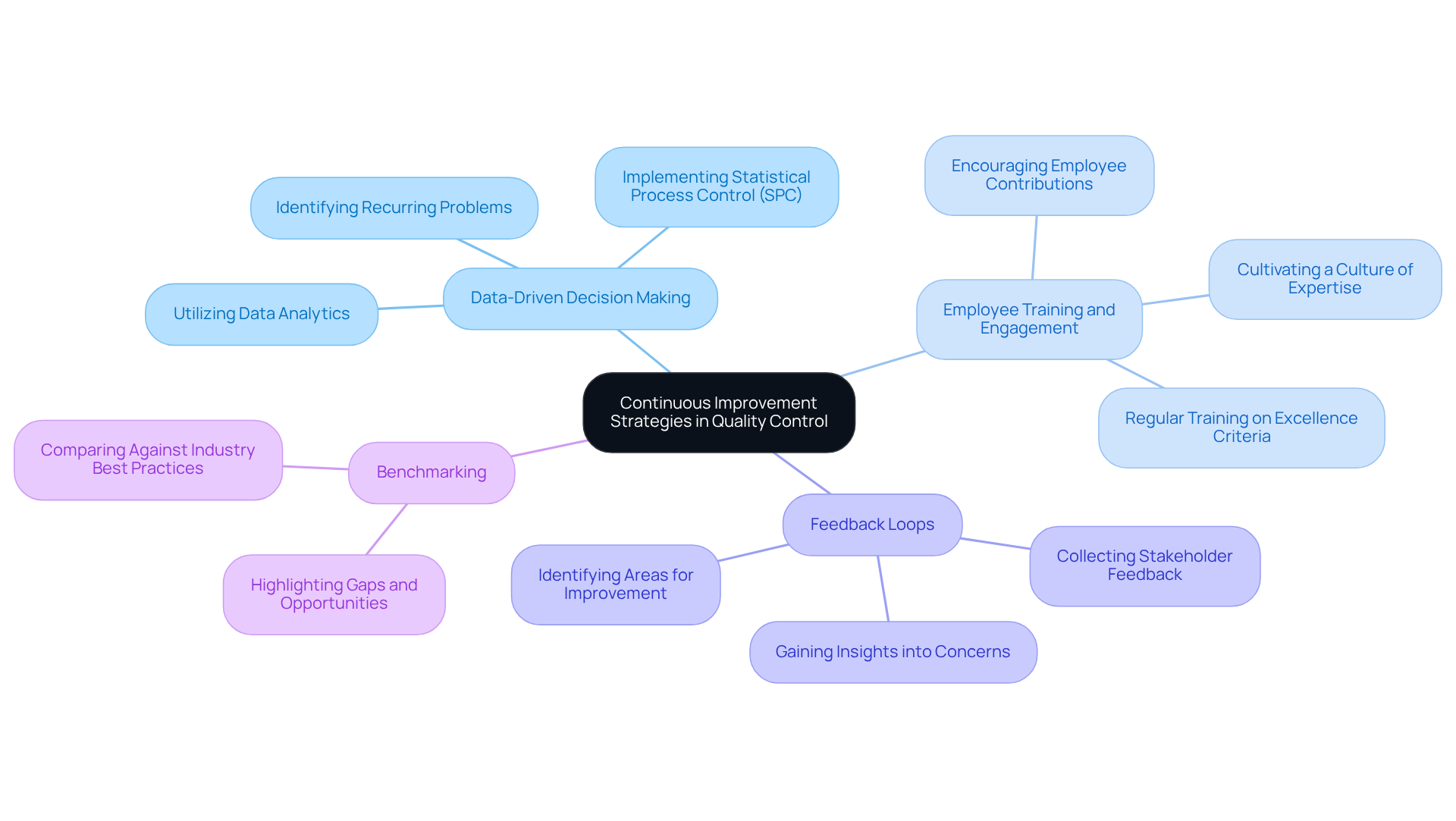
To maintain and enhance PCB control, it is essential to adopt continuous improvement strategies. Several effective approaches include:
With the enforceability of the Management System Regulation (QMSR) commencing on February 2, 2026, it is imperative for companies to ensure their management systems are audit-ready at all times. By fostering a culture of continuous improvement, companies can enhance their PCB quality control methods, leading to better outcomes in medical device manufacturing.
Implementing effective PCB quality control methods is essential for the success of medical devices, ensuring that products adhere to the highest standards of safety, reliability, and performance. By following IPC standards, manufacturers can significantly reduce defects and enhance product quality, ultimately leading to improved health outcomes for patients. The focus on rigorous testing, inspection protocols, and the control of raw materials is vital in achieving these objectives, underscoring the importance of quality assurance in the manufacturing process.
This article has discussed key strategies such as:
Each of these elements contributes to establishing a robust quality control framework that not only meets regulatory requirements but also fosters innovation and efficiency in medical device production. By prioritizing these best practices, manufacturers can confidently navigate the complexities of the healthcare market.
The significance of PCB quality control in medical devices is paramount. As the industry continues to evolve, embracing these quality management strategies will empower manufacturers to deliver safer and more effective healthcare solutions. It is imperative for organizations to commit to continuous improvement and invest in the necessary resources to uphold the highest standards in PCB quality. This dedication not only enhances product reliability but also ultimately serves the greater good of patient care and safety.
What are IPC standards and why are they important for PCBs in healthcare devices?
IPC standards are guidelines established by the Institute for Printed Circuits that set quality and reliability benchmarks for printed circuit boards (PCBs) used in healthcare devices. They are important because they ensure PCB quality control methods meet the stringent demands of healthcare applications, including durability, safety, and performance.
What specific IPC criteria are crucial for PCB quality control?
Key IPC criteria include IPC-A-600, which focuses on visual inspection, and IPC-6012, which outlines qualification requirements. These criteria are vital for engineers involved in PCB design and manufacturing.
How do IPC guidelines impact defect reduction and product reliability?
Adhering to IPC guidelines and utilizing PCB quality control methods helps manufacturers significantly reduce defects, enhance product reliability, and maintain compliance with regulatory standards.
What is IPC Class 3, and why is it relevant to healthcare instruments?
IPC Class 3 criteria are applied to healthcare instruments and require the highest level of reliability. Understanding these classifications helps engineers make informed decisions regarding materials, design, and testing protocols for medical equipment.
Can you provide examples of the real-world applications of IPC guidelines?
Organizations that follow IPC-A-600 and IPC-6012 have reported improved manufacturing outcomes and reduced risks of equipment failures. Adherence to these guidelines is crucial for ensuring PCB quality control.
What common errors can occur in manufacturing tests related to IPC standards?
Common errors include insufficient testing protocols and failure to meet qualification requirements, which can lead to significant delays in product development.
How does Voler Systems apply IPC guidelines in their products?
Voler Systems utilizes its expertise in electronic design services to develop health-related products, such as wearable technology and heart pumps, while ensuring compliance with IPC guidelines. This approach helps guarantee high-quality production processes in the evolving healthcare equipment market.
