Introduction
Mastering PCB design for medical devices is not merely a technical requirement; it is a critical factor influencing patient safety and device reliability. This article explores best practices that engineers can adopt to navigate the complexities of PCB layout, component placement, and compliance with stringent regulatory standards. Given the rapid advancements in technology and increasing regulatory scrutiny, what are the most effective strategies to ensure both innovation and adherence to safety guidelines in medical device design?
Understand PCB Design Fundamentals
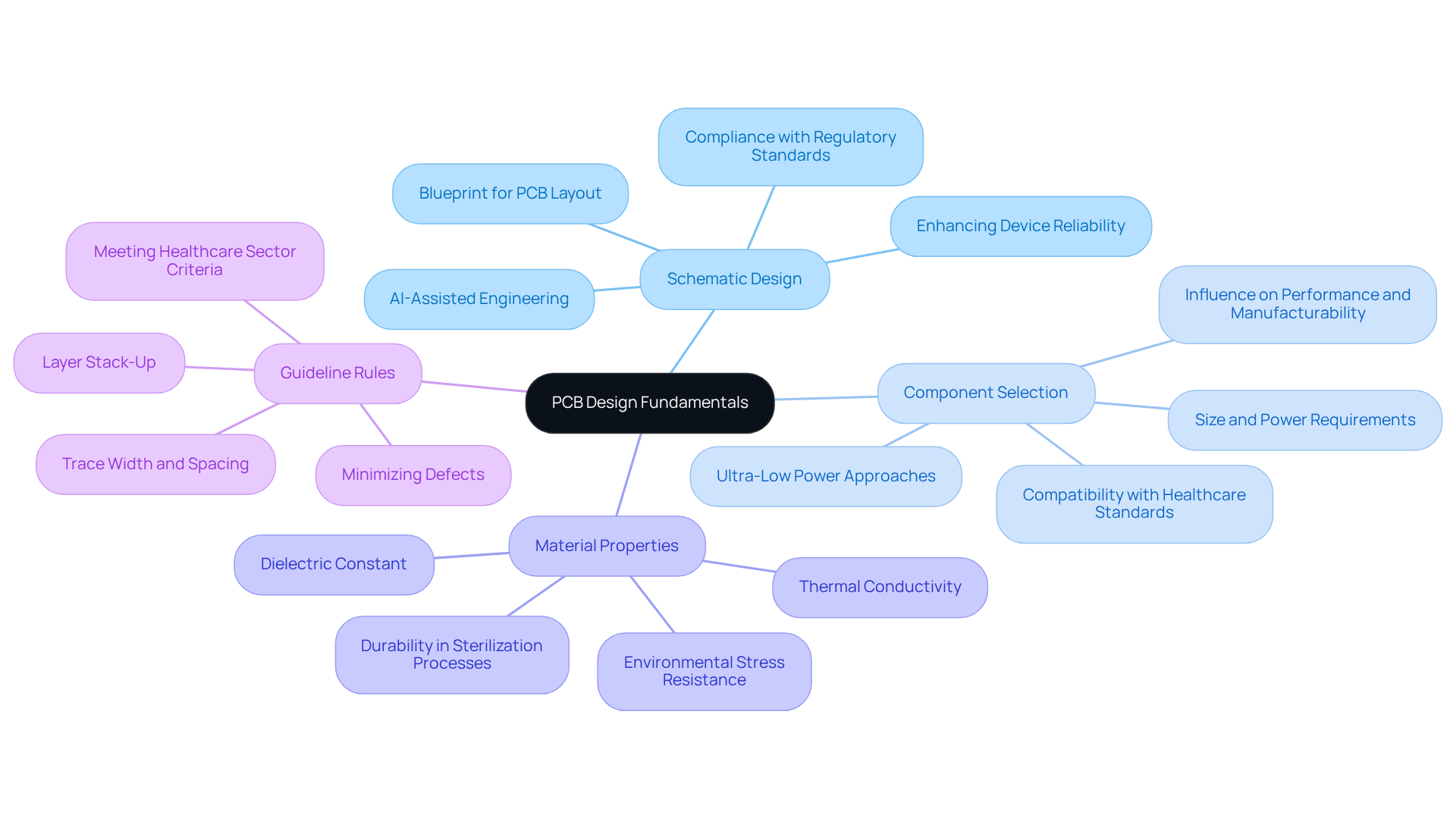
Mastering PCB creation for medical equipment begins with a solid grasp of essential concepts that shape the development process. The key elements include:
- Schematic Design: This serves as the blueprint for the PCB layout, where a well-structured schematic defines the circuit's functionality. This initial step is crucial, as it establishes the foundation for all subsequent phases of development. Industry specialists emphasize that effective schematic planning is vital for ensuring compliance with and enhancing device reliability. employs to refine the design process, ensuring that products are compliant and designed for the future of intelligent healthcare.
- : The selection of components is critical. Engineers must evaluate factors such as size, power requirements, and compatibility with healthcare standards. The right components not only influence the system's performance but also its manufacturability and adherence to safety regulations. prioritizes , which enhance battery longevity in wireless medical equipment, a key consideration for wearable technology.
- Material Properties: A thorough understanding of the properties of materials used in PCBs, including dielectric constant and thermal conductivity, is essential for ensuring . Advanced materials can significantly enhance durability, particularly in devices that must endure sterilization processes and environmental stress.
- : Familiarity with , such as trace width, spacing, and layer stack-up, is crucial for manufacturability and performance. Adhering to these guidelines minimizes the risk of defects and ensures that the final product meets the .
By mastering these fundamentals, engineers can develop models and that not only meet functional requirements but also comply with the rigorous standards necessary for medical equipment, ultimately enhancing patient safety and effectiveness. Furthermore, common pitfalls in PCB creation, such as neglecting documentation and traceability, should be avoided to ensure compliance and reliability. ' expertise in test systems further assures in electronic equipment development, supporting manufacturers from prototype to production.
Optimize Component Placement Strategies
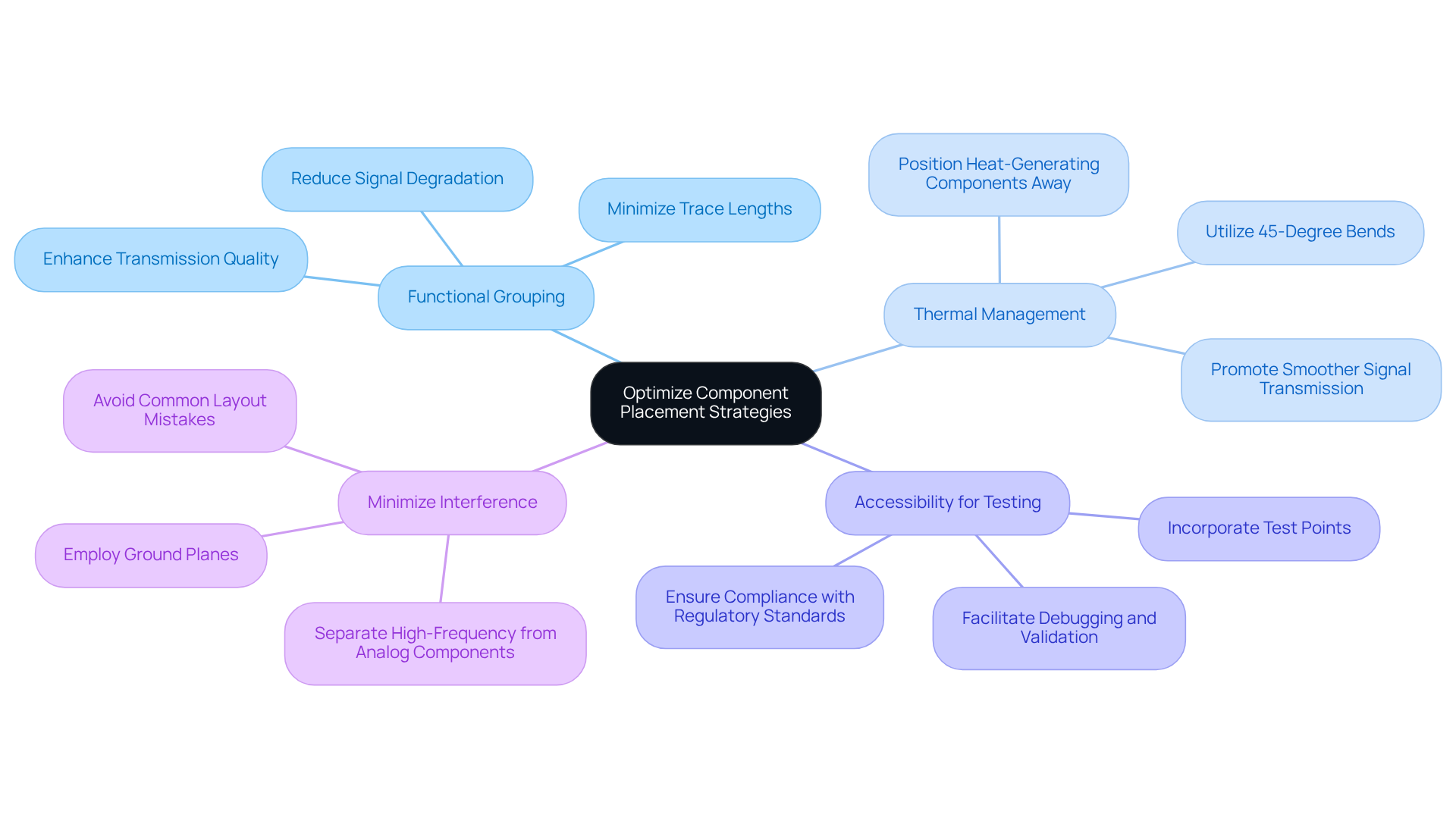
Enhancing component arrangement is crucial in PCB creation for . Here are key practices to improve performance and compliance:
- : Strategically position components that interact closely to minimize trace lengths. This approach reduces signal degradation and enhances overall circuit efficiency. Not only does it simplify the design, but it also improves transmission quality, which is essential for .
- : Place heat-generating components away from sensitive areas to prevent overheating. Effective is vital for ensuring the reliability and longevity of equipment, particularly in environments with frequent temperature variations. Utilizing 45-degree bends and curved traces can further minimize reflection points, promoting smoother signal transmission.
- : Incorporate test points in easily accessible locations to facilitate . This practice is essential for ensuring that equipment can be effectively tested and maintained, ultimately aiding in compliance with regulatory standards. The integration of robust test systems from is crucial, as they ensure that products meet the required quality and reliability standards throughout the design and manufacturing process.
- : Keep high-frequency components separated from analog components to reduce electromagnetic interference (EMI). Employ ground planes to mitigate interference risks, ensuring that the equipment operates reliably in electrically noisy environments. Avoiding common layout mistakes, such as improper trace routing and insufficient grounding, is also essential for maintaining signal integrity.
By applying these strategies, designers can significantly enhance the performance and manufacturability of healthcare products, ensuring compliance with stringent regulatory standards. plays a pivotal role in this process, offering innovative solutions that .
Implement Effective Routing Techniques
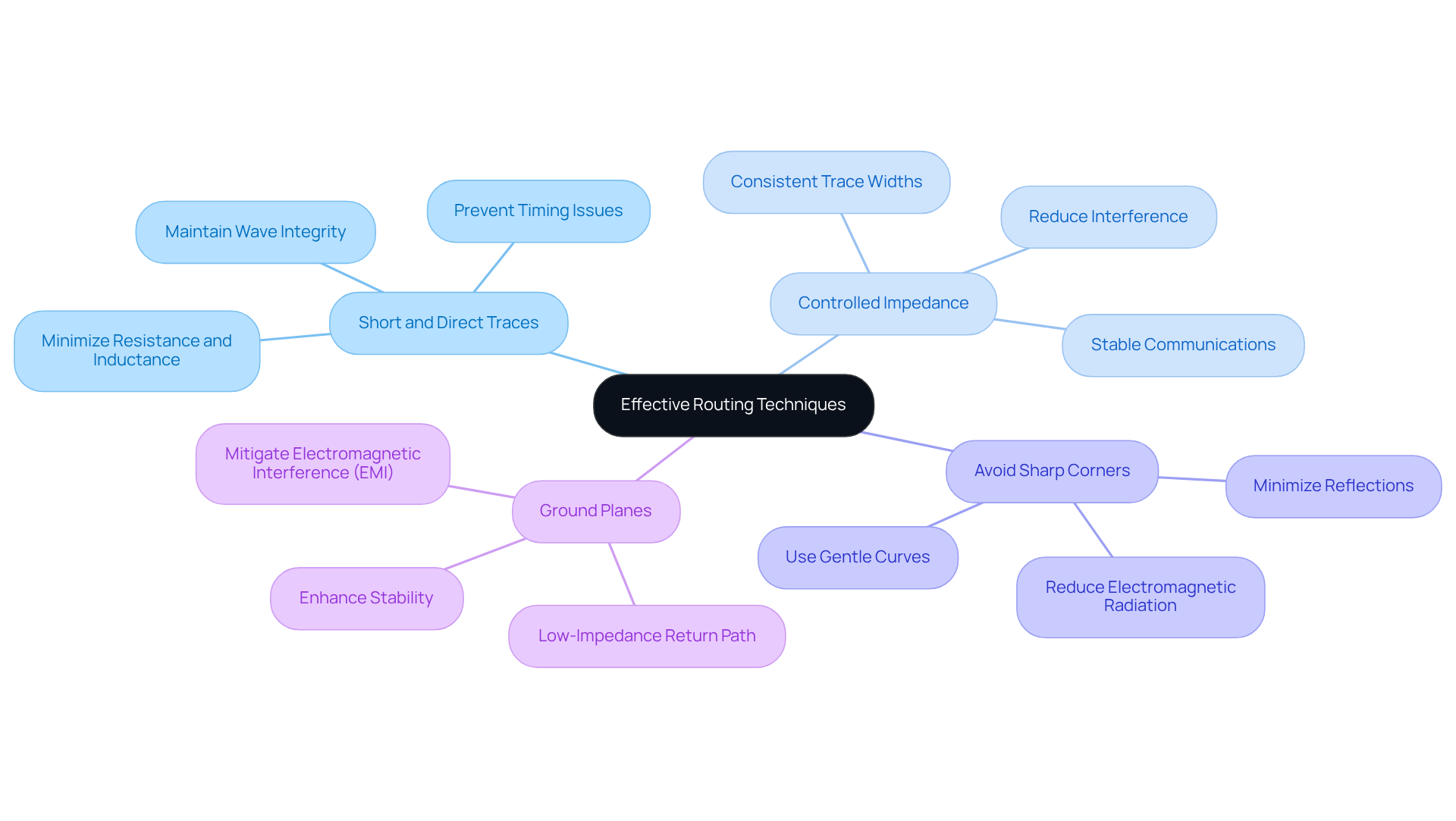
Effective are essential for ensuring the performance and reliability of . Key practices include:
- : Traces should be kept as short as possible to minimize resistance and inductance, which can degrade signal quality. This is particularly crucial for high-frequency waves to prevent timing issues and maintain wave integrity.
- : Consistent trace widths and spacing are vital for managing impedance, especially in . This practice ensures stable communications and reduces the likelihood of interference.
- Avoid Sharp Corners: Gentle curves should be used instead of sharp corners in routing to minimize reflections and maintain . Sharp angles can increase electromagnetic radiation, adversely affecting overall performance.
- : should be utilized to provide a low-impedance return path for transmissions, which helps mitigate electromagnetic interference (EMI). This technique enhances the , particularly in environments where reliability is paramount.
Incorporating , such as automated test equipment and analyzers, is crucial for validating these . These systems ensure that meet stringent quality and reliability standards. Furthermore, as the integration of Edge AI continues to reshape modern healthcare, employing these routing strategies can enhance the efficiency of tools utilizing advanced technologies. By adhering to these , designers can significantly bolster the performance and reliability of , ensuring they function as intended in critical applications.
Ensure Compliance with Regulatory Standards
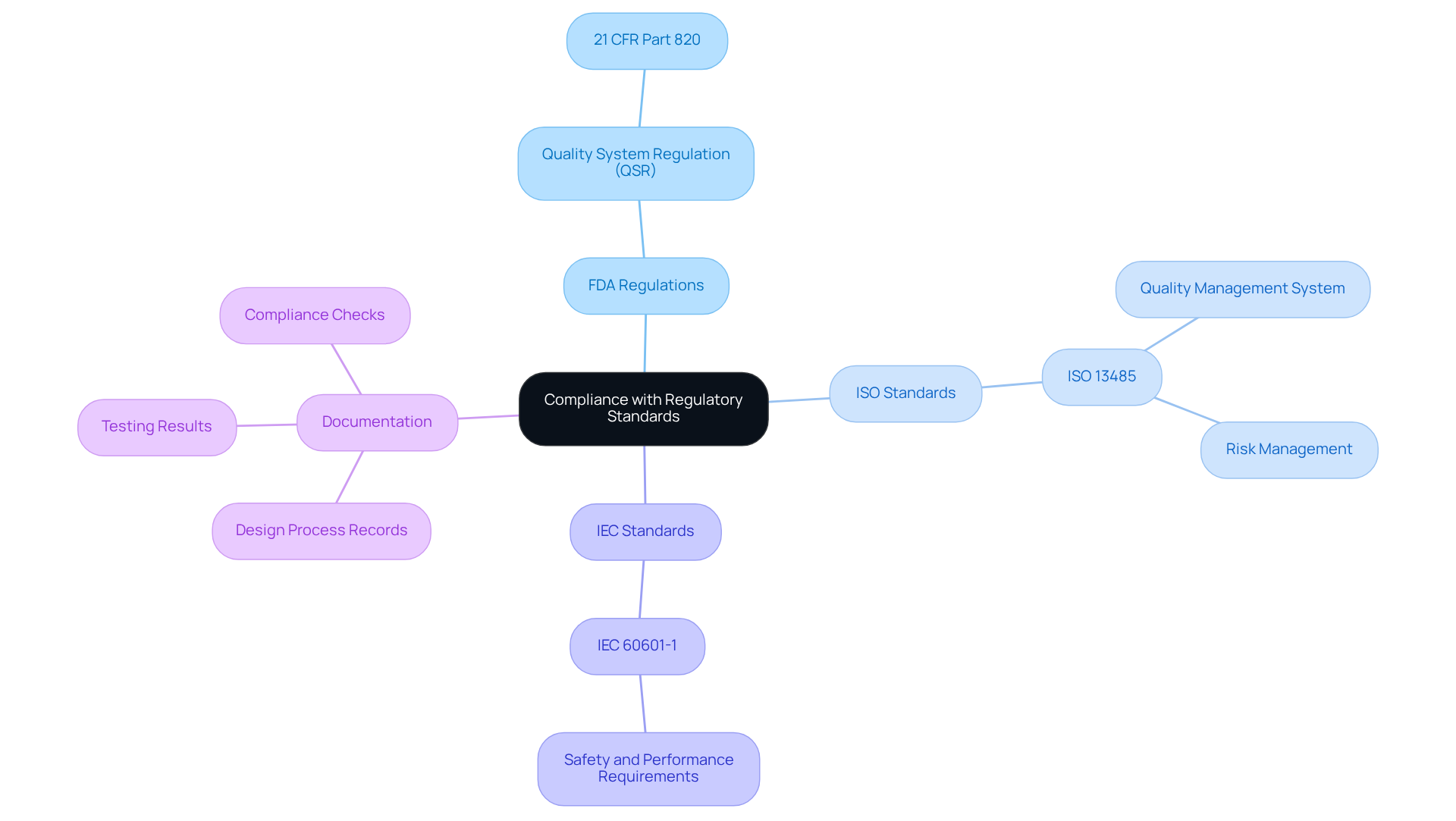
Adhering to regulatory standards is essential in PCB development for healthcare equipment. Key considerations include:
- : Understanding the FDA's Quality System Regulation (QSR) is crucial. It is imperative that your design aligns with the requirements specified in 21 CFR Part 820, which outlines the necessary to be followed throughout the product lifecycle.
- : is vital for establishing a quality management system in healthcare product manufacturing. This standard emphasizes , ensuring that products meet safety and efficacy requirements.
- IEC Standards: Familiarity with is necessary, as it specifies safety and performance requirements for electrical equipment used in healthcare. This knowledge is vital for ensuring reliability in clinical settings.
- Documentation: Maintaining , testing results, and compliance checks is crucial. Thorough record-keeping facilitates audits and , demonstrating adherence to industry standards.
By prioritizing compliance, designers can ensure that their are not only safe and effective but also positioned for successful market entry.
Conclusion
Mastering PCB design for medical devices is a multifaceted process that relies on a comprehensive understanding of fundamental principles, strategic planning, and strict adherence to industry standards. The intricate relationship between schematic design, component selection, and material properties establishes the foundation for creating reliable and effective medical equipment. By concentrating on these essential elements, engineers can ensure that their designs not only function as intended but also comply with the rigorous requirements of the healthcare sector.
Key strategies such as:
- Optimizing component placement
- Implementing effective routing techniques
- Adhering to regulatory standards
significantly enhance the performance and manufacturability of medical PCBs. Techniques like functional grouping, thermal management, and maintaining controlled impedance are vital for minimizing interference and ensuring signal integrity. Furthermore, compliance with FDA, ISO, and IEC standards is crucial for safeguarding patient safety and facilitating successful market entry.
In summary, the importance of best practices in PCB design for medical devices is paramount. As the healthcare landscape continues to evolve, adopting these principles will not only foster the development of innovative and reliable medical technologies but also contribute to the overall advancement of patient care. It is essential for engineers and designers to remain diligent in their pursuit of excellence, ensuring that every PCB created meets the highest standards of quality and compliance.
Frequently Asked Questions
What are the key elements of PCB design for medical equipment?
The key elements include schematic design, component selection, material properties, and guideline rules.
Why is schematic design important in PCB development?
Schematic design serves as the blueprint for the PCB layout, defining the circuit's functionality and establishing the foundation for all subsequent development phases. It is crucial for compliance with regulatory standards and enhancing device reliability.
What factors should engineers consider when selecting components for PCBs?
Engineers must evaluate size, power requirements, and compatibility with healthcare standards. Proper component selection influences system performance, manufacturability, and adherence to safety regulations.
How do material properties affect PCB design?
Understanding material properties, such as dielectric constant and thermal conductivity, is essential for ensuring long-term reliability and performance. Advanced materials can enhance durability, especially in devices that undergo sterilization processes.
What are guideline rules in PCB design, and why are they important?
Guideline rules include trace width, spacing, and layer stack-up, which are crucial for manufacturability and performance. Adhering to these guidelines minimizes defects and ensures compliance with the healthcare sector's stringent criteria.
What are common pitfalls to avoid in PCB creation?
Common pitfalls include neglecting documentation and traceability, which can jeopardize compliance and reliability.
How does Voler Systems contribute to PCB design for medical equipment?
Voler Systems employs AI-assisted engineering to refine the design process, ensuring compliance and enhancing the reliability of products. They also prioritize ultra-low power approaches for improved battery longevity in wireless medical equipment.
List of Sources
- Understand PCB Design Fundamentals
- Designing PCBs for Medical Devices: Key Considerations & Compliance Tips (https://blog.picamfg.com/designing-pcbs-for-medical-devices-key-considerations-compliance-tips)
- How PCB Design Helps The Medical Field - Consett Magazine (https://consettmagazine.com/2023/10/18/how-pcb-design-helps-the-medical-field-92873)
- PCB Design in Medical Devices: Enhancing Diagnostic Precision And Reliability - product - News - Sunsoar (https://oem-pcb.com/news/pcb-design-in-medical-devices-enhancing-diagn-82319565.html)
- Medical Device PCB Design — FDA Compliant Layouts (https://medium.com/@raypcb/medical-device-pcb-design-fda-compliant-layouts-c69a4f9b0d79)
- linkedin.com (https://linkedin.com/posts/devinekevin_designing-pcbs-for-medical-devices-key-considerations-activity-7417592684997083137-dZj7)
- Optimize Component Placement Strategies
- Component Placement Guidelines for PCB Design and Assembly (https://acceleratedassemblies.com/blog/component-placement-guidelines-for-pcb-design-and-assembly)
- Component Placement Best Practices for High-Reliability ADAS PCBs (https://allpcb.com/blog/pcb-knowledge/component-placement-best-practices-for-high-reliability-adas-pcbs.html)
- Medical Device PCB Design: Top 10 Essential Guidelines (https://911eda.com/articles/medical-device-pcb-design-guidelines)
- Medical Device PCB Design — FDA Compliant Layouts (https://medium.com/@raypcb/medical-device-pcb-design-fda-compliant-layouts-c69a4f9b0d79)
- suntsu.com (https://suntsu.com/blog/pcb-component-selection-essential-tips-for-optimal-design-and-performance)
- Implement Effective Routing Techniques
- Medical Device PCB Design: Top 10 Essential Guidelines (https://911eda.com/articles/medical-device-pcb-design-guidelines)
- Medical Device PCB Design — FDA Compliant Layouts (https://medium.com/@raypcb/medical-device-pcb-design-fda-compliant-layouts-c69a4f9b0d79)
- richpcba.com (https://richpcba.com/blog/top-medical-pcba-designs-for-efficient-healthcare-solutions)
- Ensuring Signal Integrity in Patient Monitoring PCBs: Best Practices for Trace Routing and Grounding (https://allpcb.com/blog/pcb-design/ensuring-signal-integrity-in-patient-monitoring-pcbs-best-practices-for-trace-routing-and-grounding.html)
- Ensure Compliance with Regulatory Standards
- totalboardsolutions.com (https://totalboardsolutions.com/compliance-for-pcbs-within-medical-device-regulations)
- FDA Warning Letter Statistics on Medical Devices in the Past Fiscal Year 2025 - ECA Academy (https://gmp-compliance.org/gmp-news/fda-warning-letter-statistics-on-medical-devices-in-the-past-fiscal-year-2025)
- Medical PCB Assembly: Reliability and Compliance Challenges - LSTPCB (https://lstpcb.com/news/medical-pcb-assembly-reliability-and-compliance-challenges)
- greenlight.guru (https://greenlight.guru/blog/iso-13485-qms-medical-device)
- How Will FDA's New Rules Impact Your Health Gadgets? (https://spectrum.ieee.org/fda-medical-device-rules)





