Understanding the Software Product Development Process for Medical Devices
Introduction Understanding the software product development process for medical devices...

The field of embedded systems design requires a strategic approach that integrates innovation with practical functionality. Following best practices in the design process streamlines development and enhances product quality and reliability. The fast-paced advancements in technology and shifting user demands create challenges for designers. This article outlines four essential practices that guide the embedded systems design process, providing insights for achieving success in this competitive field.
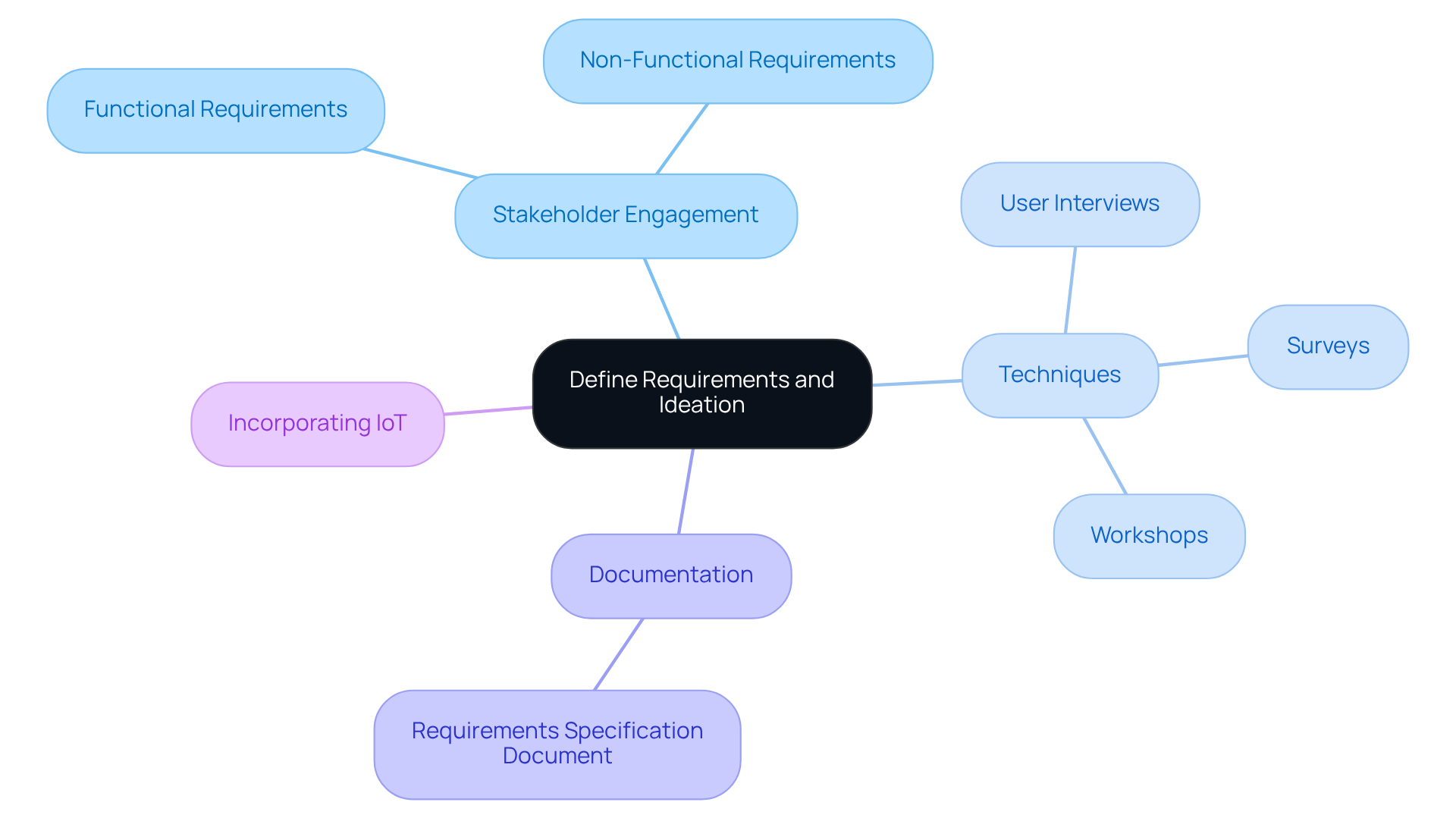
The initial stage of the embedded systems design process hinges on accurately specifying project requirements, a critical factor for achieving successful outcomes. Engaging stakeholders is crucial for gathering functional and non-functional requirements. This ensures a comprehensive consideration of all system aspects. Techniques such as user interviews, surveys, and workshops effectively elicit detailed insights. Furthermore, creating user personas and scenarios aids in visualizing the end product's usage, thus guiding the ideation phase. Documenting these requirements in a structured format, such as a requirements specification document, is vital for maintaining clarity throughout the project lifecycle. This foundational work not only aligns the team but also significantly reduces the risk of scope creep and miscommunication during the embedded systems design process. Incorporating IoT concepts from the beginning is vital for ensuring that the final product meets contemporary requirements. Utilizing an AI-driven engineering approach enhances design efficiency, facilitating the development of adaptive embedded solutions that respond to evolving technology demands, particularly in the medical device field.
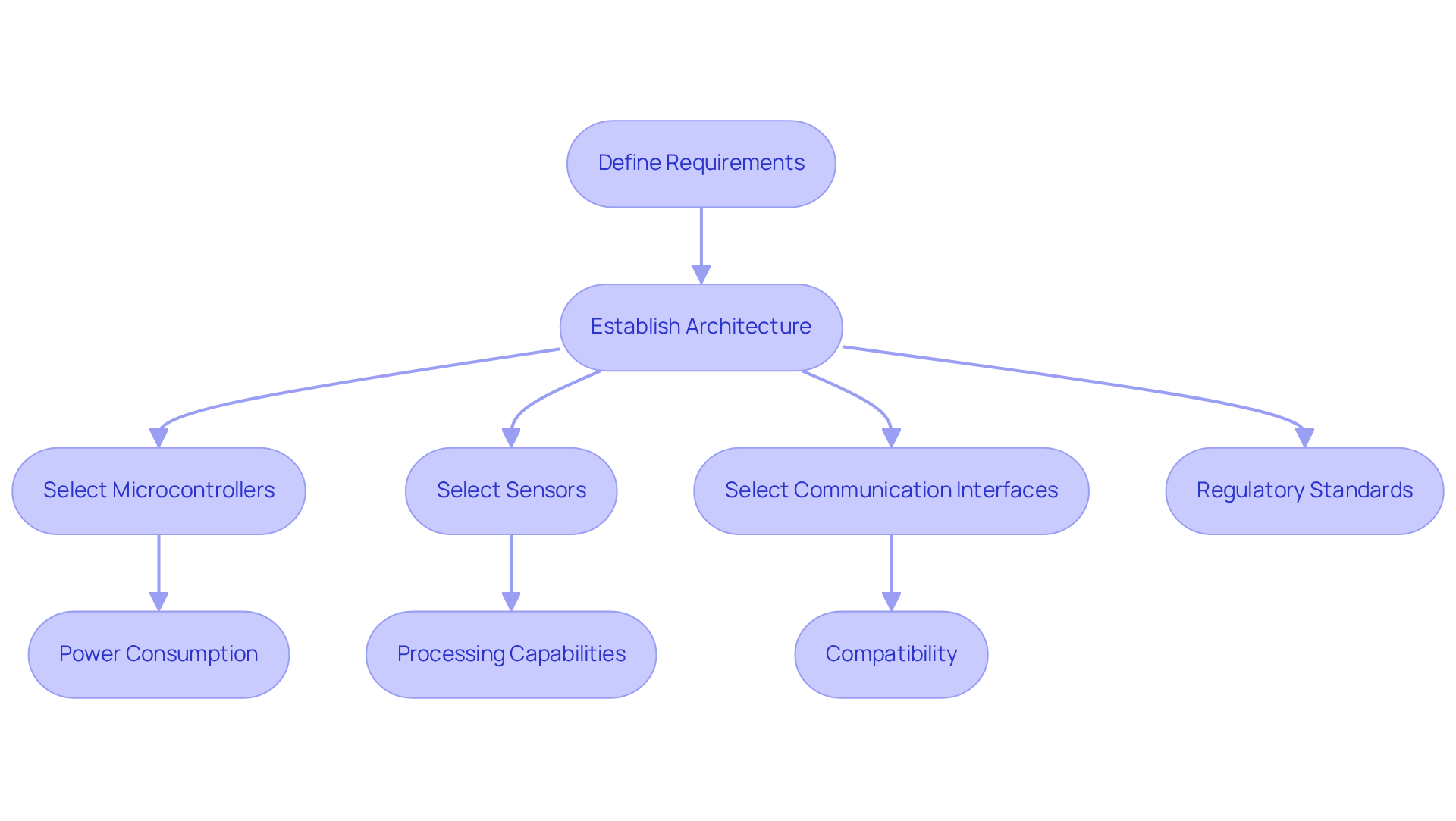
Once the requirements are defined, the next critical step in the embedded systems design process is to establish a robust architecture. This embedded systems design process involves the selection of suitable microcontrollers, sensors, and communication interfaces that align with the project's objectives. A top-down approach proves beneficial in the embedded systems design process, beginning with a high-level architecture diagram that clearly outlines the major components and their interactions. Key considerations in the embedded systems design process include:
Navigating regulatory standards requires careful consideration of these factors to ensure the apparatus meets user needs.
Given the growing aging population, manufacturers must prioritize durable components that can endure the requirements of long-term care equipment, such as pacemakers and insulin pumps. The average life expectancy in the U.S. has increased to 79 years, and the number of adults aged 65 and older is expected to surpass 98 million by 2060, creating a demand for dependable medical equipment.
Utilizing simulation tools in the embedded systems design process can validate architectural decisions before physical implementation, allowing for adjustments that enhance performance while managing costs. In competitive markets such as medical equipment and IoT solutions, maintaining a balance between performance and cost is essential. Manufacturers are increasingly focusing on overall expenses, including testing, packaging, and shipping, to keep product costs reasonable while ensuring high quality and compliance with regulatory requirements. By meticulously choosing components according to these criteria, companies can optimize the embedded systems design process and enhance the manufacturability of their products.
Additionally, it is vital to consider the user environment when designing wearable sensors. Elements like moisture, temperature, and user behavior can greatly influence the efficiency and longevity of the apparatus. At Voler Systems, we excel in ultra-low power strategies, ensuring optimal power management for wireless medical devices. Our dedication to AI-driven innovation further improves the design approach, enabling smarter, more efficient solutions that satisfy the needs of contemporary healthcare. Ultimately, the design choices made today will shape the future of healthcare technology.
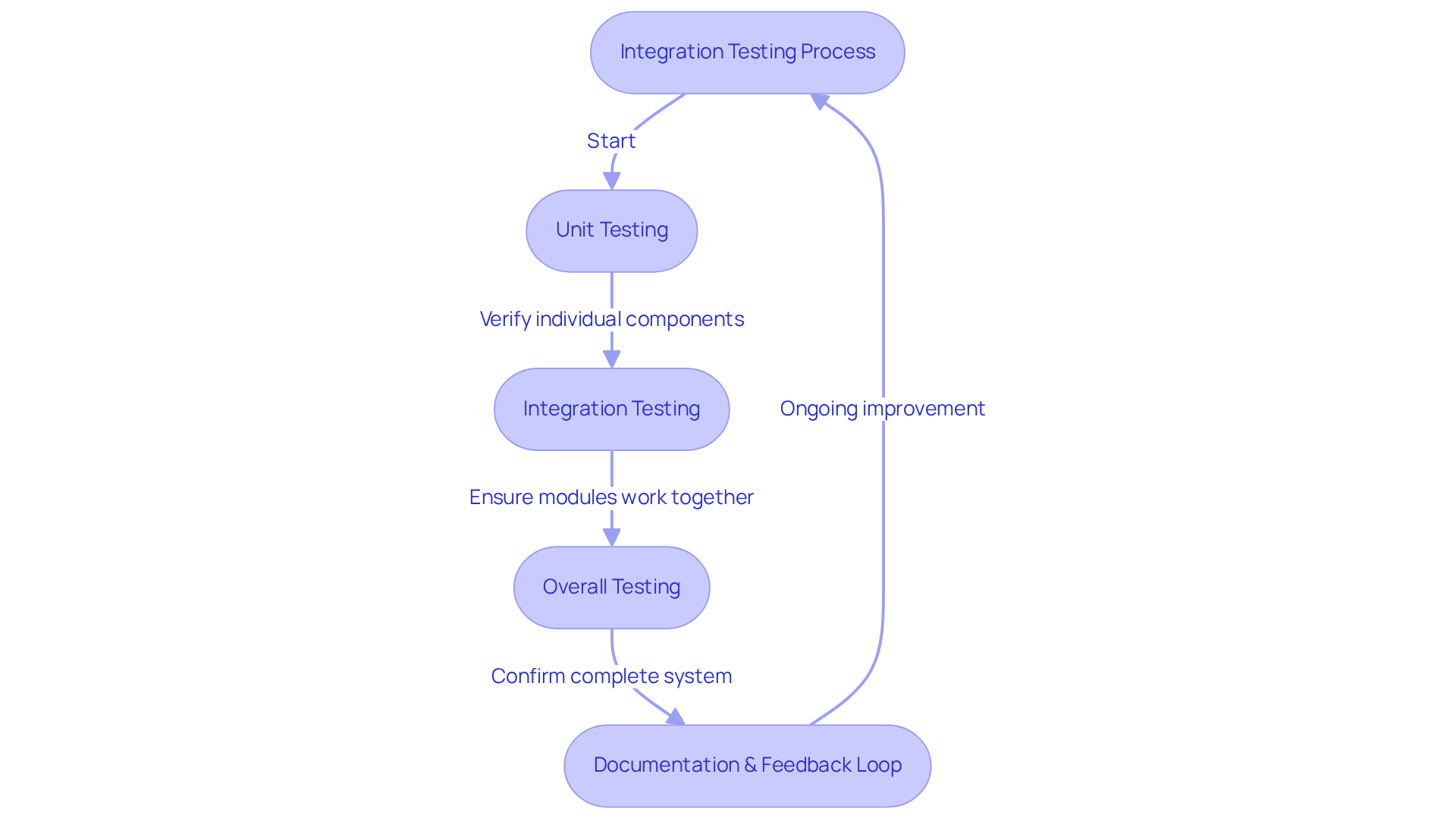
Integration testing serves as a pivotal phase in the embedded systems design process, where the merging and evaluation of individual components occur as a unified system. Initiating this process early in the development cycle is essential for identifying integration issues before they escalate into significant problems. The implementation of automated testing frameworks markedly improves testing efficiency and accuracy. Key strategies include:
Additionally, documenting test results and maintaining a continuous feedback loop with the development team is essential for fostering ongoing improvement and ensuring quality assurance. This structured approach not only mitigates risks but also aligns with industry best practices within the embedded systems design process, ultimately leading to higher quality products in the medical device sector. By adhering to this structured methodology, organizations can significantly enhance product quality and compliance within the medical device industry.
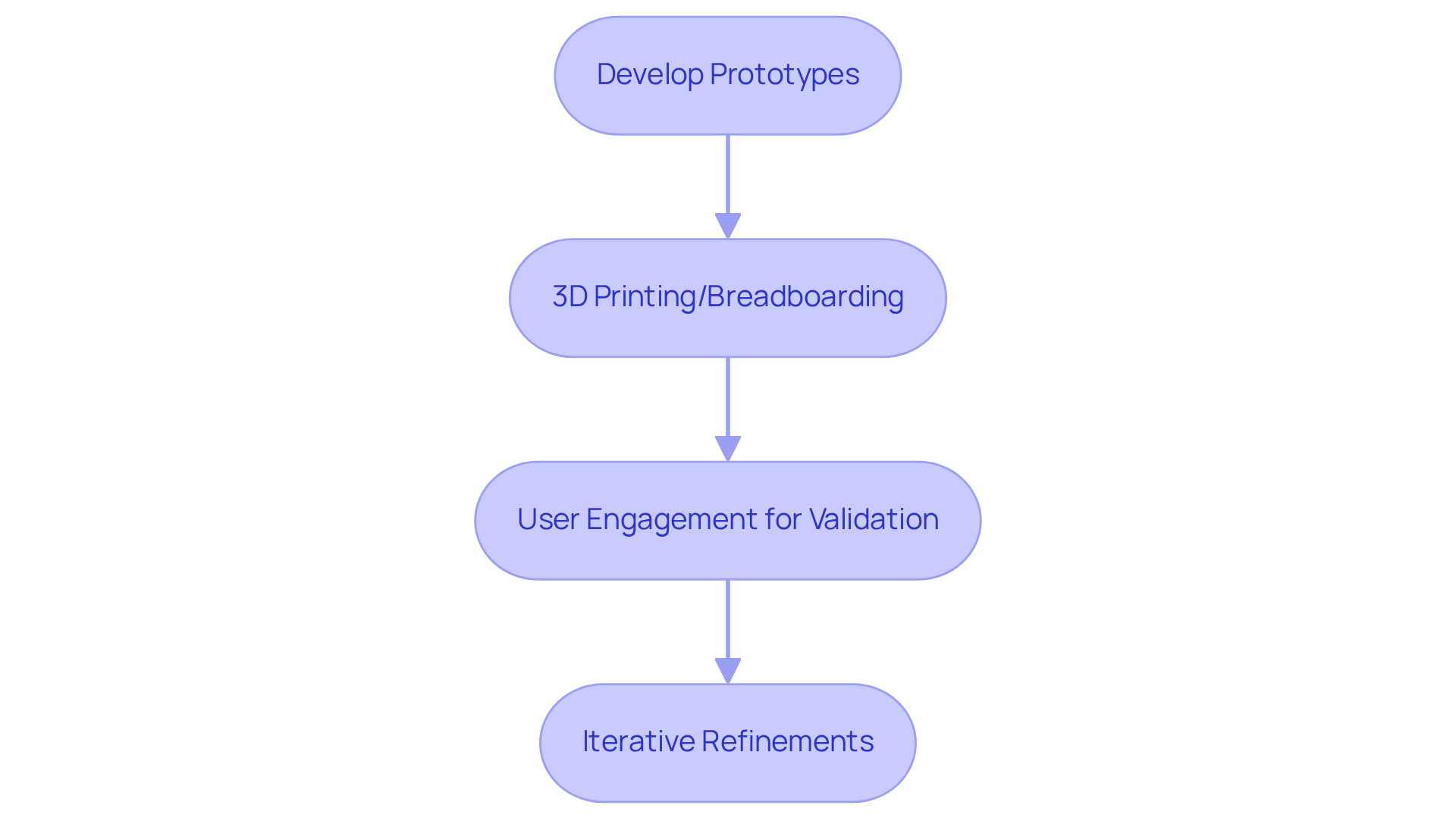
Effective prototyping is essential in embedded systems development, as it transforms abstract ideas into tangible solutions.
3D printing and breadboarding enable rapid iterations and testing, reducing the time from concept to functional prototype.
Engaging end-users in the validation process is vital. Their feedback on usability and functionality directly informs necessary design refinements.
This iterative approach enhances product alignment with user expectations and identifies discrepancies against initial requirements early.
Addressing these issues early mitigates the risk of costly changes in later development stages, enhancing the success of medical devices.
The design of embedded systems necessitates a strategic approach to ensure successful outcomes in a complex landscape. Prioritizing the definition of requirements and engaging stakeholders from the outset minimizes misunderstandings and aligns project objectives. This phase establishes a foundation for robust system architecture and the careful selection of components that meet performance and regulatory standards.
Key practices, including integration testing and effective prototyping, are essential for ensuring quality assurance and aligning products with user needs. Early testing and user feedback enhance product reliability and streamline development by addressing potential issues before they escalate. The incorporation of innovative technologies, such as AI-driven approaches and simulation tools, further supports the creation of adaptive, high-quality embedded solutions tailored for modern healthcare challenges.
The importance of adhering to best practices in embedded systems design is paramount. As the demand for reliable medical devices continues to grow, following structured methodologies and embracing user-centered design will be critical in developing solutions that meet the evolving needs of the industry. Organizations that embrace these principles will not only improve their product offerings but also play a pivotal role in shaping the future of healthcare technology.
What is the first stage of the embedded systems design process?
The first stage of the embedded systems design process is defining requirements and ideation.
Why is defining project requirements important?
Accurately specifying project requirements is critical for achieving successful outcomes in embedded systems design.
How can stakeholders be engaged to gather requirements?
Stakeholders can be engaged through techniques such as user interviews, surveys, and workshops to gather functional and non-functional requirements.
What are user personas and scenarios, and how do they help?
User personas and scenarios are tools used to visualize the end product's usage, which aids in guiding the ideation phase of the design process.
Why is documenting requirements important?
Documenting requirements in a structured format, such as a requirements specification document, is vital for maintaining clarity and alignment throughout the project lifecycle.
How does proper requirement definition reduce project risks?
Proper requirement definition significantly reduces the risk of scope creep and miscommunication during the embedded systems design process.
What role do IoT concepts play in the design process?
Incorporating IoT concepts from the beginning is essential for ensuring that the final product meets contemporary requirements.
How does an AI-driven engineering approach benefit embedded systems design?
An AI-driven engineering approach enhances design efficiency and facilitates the development of adaptive embedded solutions that respond to evolving technology demands, especially in the medical device field.
