Best Practices for Connected Product Development in Medical Devices
Introduction The rapid evolution of healthcare technology necessitates a new approach to...

The convergence of engineering disciplines within mechatronics is reshaping the medical device sector, leading to the emergence of intelligent healthcare solutions. This article outlines best practices for mastering the mechatronics design process, emphasizing the critical roles of:
in enhancing device functionality and improving patient outcomes. As the industry evolves, engineers must navigate the complexities of regulatory standards to ensure both innovation and compliance in their designs.
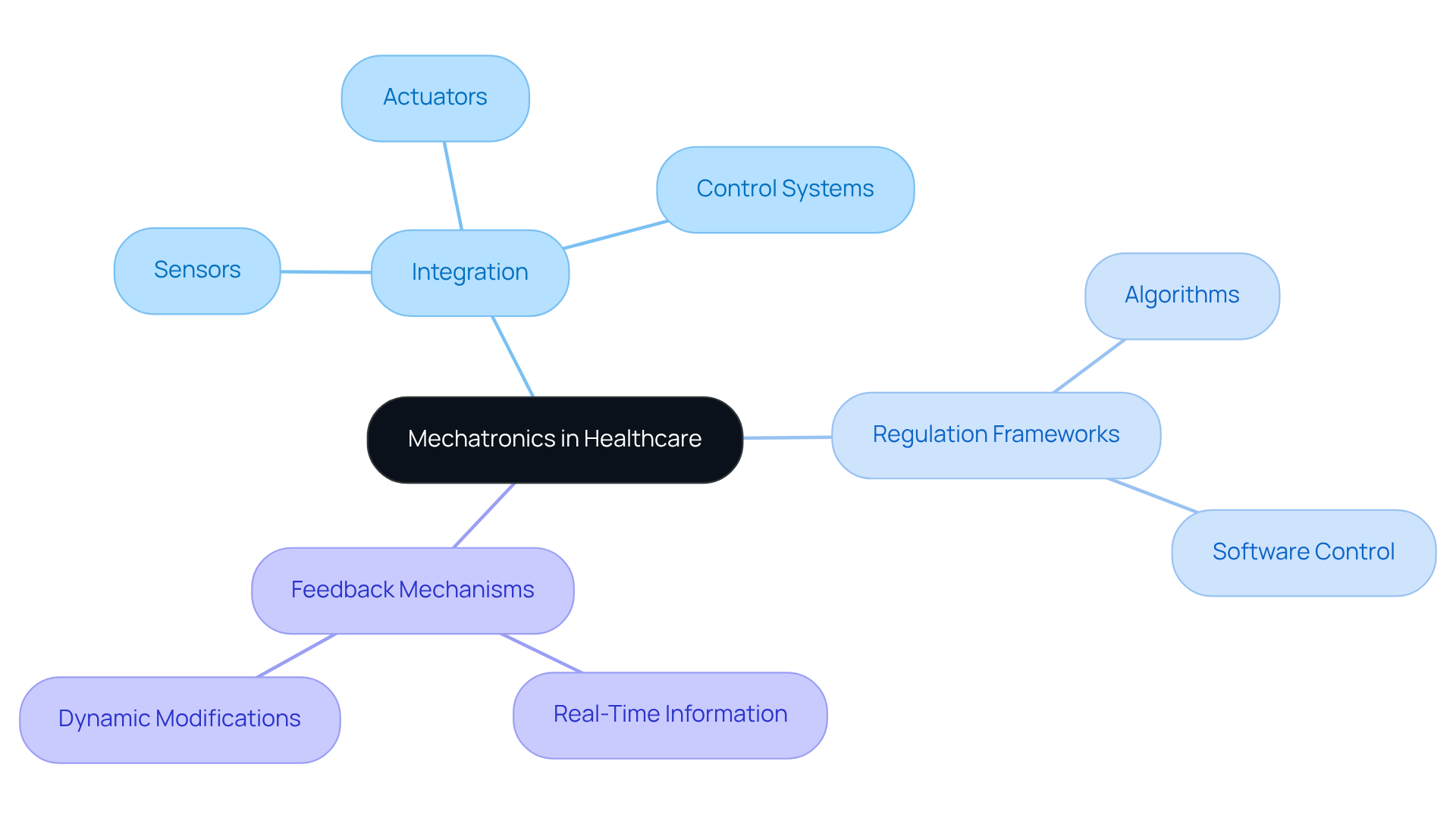
Mechatronics represents a convergence of multiple engineering disciplines, essential for advancing intelligent systems in healthcare. In the field of medical equipment, mechatronics entails the integration of various components, such as sensors, actuators, and control systems, which together enhance functionality and performance. The core principles of mechatronics in healthcare technology include:
A comprehensive grasp of these principles is vital for engineers involved in the mechatronics design process of healthcare equipment creation, as they determine the operational capabilities of apparatus in practical applications, ensuring adherence to rigorous safety and performance standards. Furthermore, as the industry shifts towards value-based models, the mechatronics design process is expected to demonstrate measurable improvements in function and recovery, underscoring the importance of effectively integrating mechatronics principles. As healthcare technology evolves, the mechatronics design process will be crucial in enhancing patient outcomes and operational efficiency.
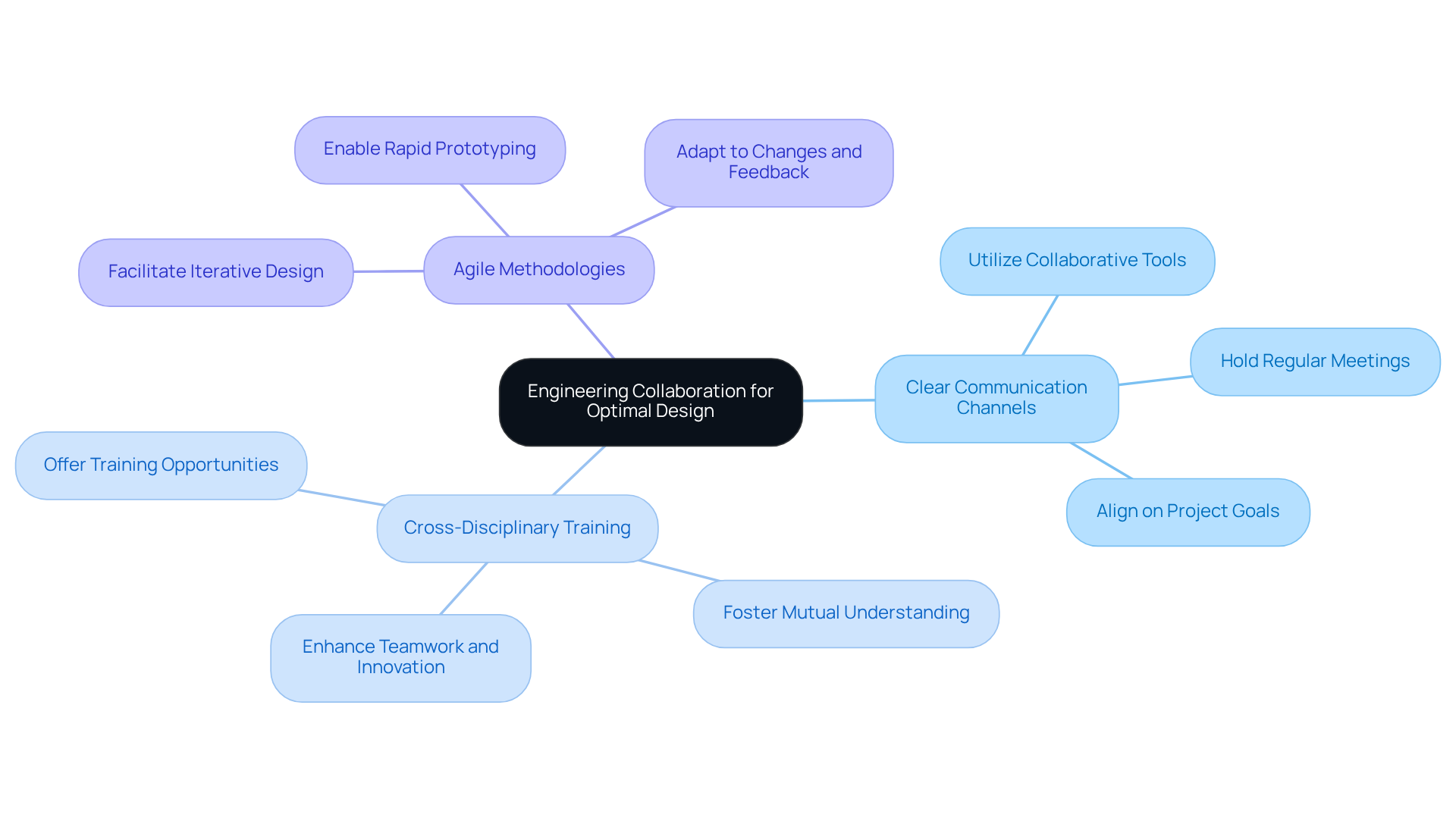
Effective collaboration among various engineering disciplines is essential for the successful creation of healthcare equipment. To achieve this, consider the following best practices:
Real-world instances, such as the advancement of wearable health technology, illustrate how collaborative teams can develop innovative solutions that not only satisfy user requirements but also adhere to strict regulatory standards. By leveraging diverse expertise, these teams enhance the quality and functionality of healthtech products, ultimately leading to better patient outcomes. Ultimately, such collaboration not only meets regulatory standards but also significantly enhances patient care.
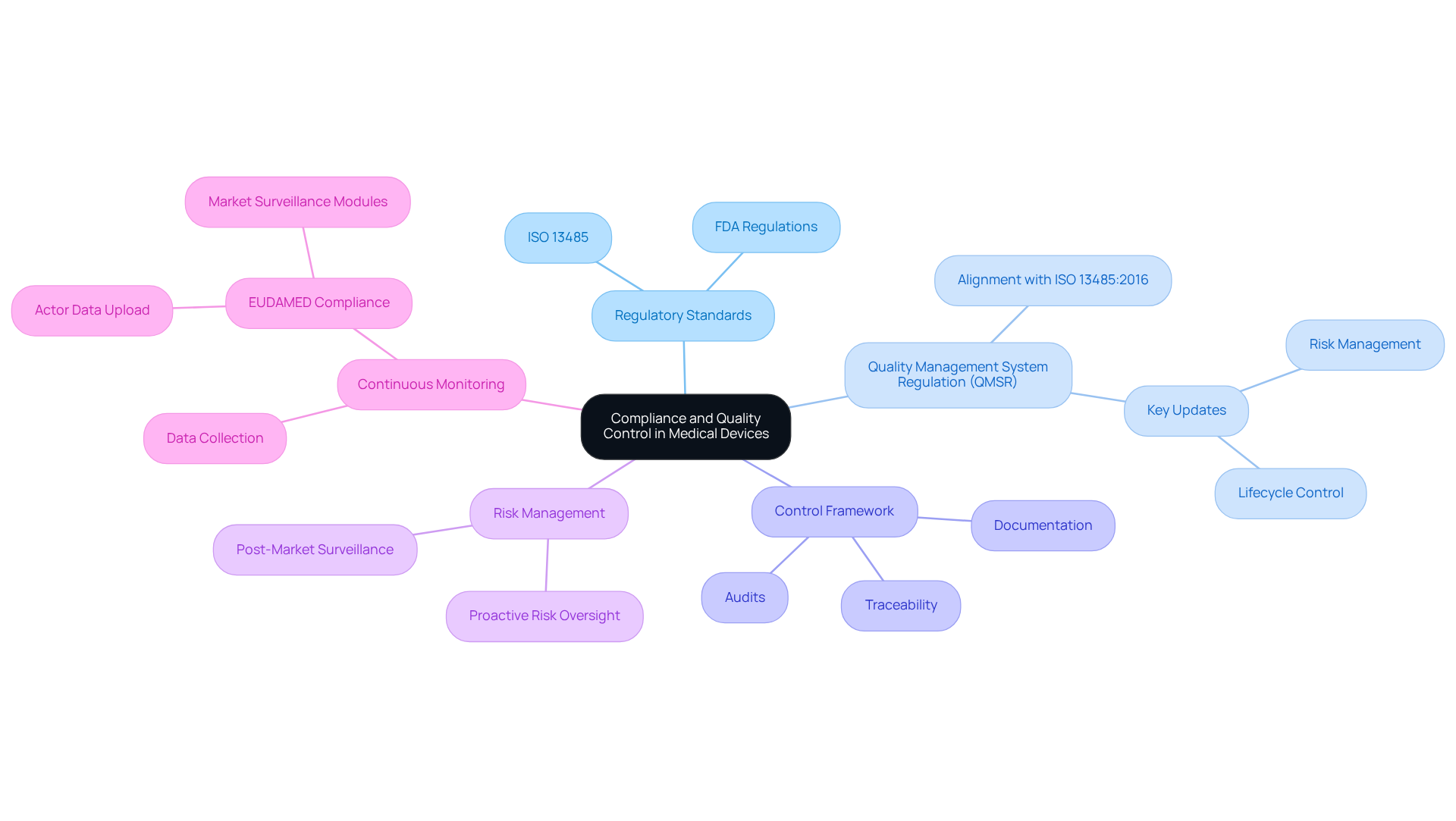
Understanding standards such as ISO 13485 and FDA regulations is critical for establishing effective quality management systems in the medical device sector. The recent updates to the Quality Management System Regulation (QMSR) align U.S. regulations with ISO 13485:2016, emphasizing risk management and lifecycle control, which are now front-line requirements for manufacturers. This alignment enhances compliance and inspection efficiency, facilitating manufacturers' navigation of regulatory landscapes.
Establishing a robust control framework is essential. This framework must encompass thorough documentation, proactive risk management, and traceability to justify and record all development decisions. The revised regulations demand enhanced traceability and organized procedures for examining complaints and performing post-market surveillance, emphasizing the necessity for careful control measures. Failure to implement these practices can lead to significant risks, including regulatory penalties and compromised product safety.
Regular evaluations of development processes and compliance checks should be scheduled to identify potential issues proactively. Regular audits are essential for ensuring adherence to regulatory requirements and for maintaining a proactive stance on risk oversight throughout the product lifecycle. The QMSR encourages continuous monitoring and data collection, which can significantly enhance regulatory preparedness and readiness for inspections. Additionally, companies must ensure that Actor data is uploaded to EUDAMED before May 2026 to avoid delays in the approval process.
By following these practices, engineers can effectively reduce risks linked to non-compliance, ultimately resulting in the creation of safer and more efficient healthcare instruments.
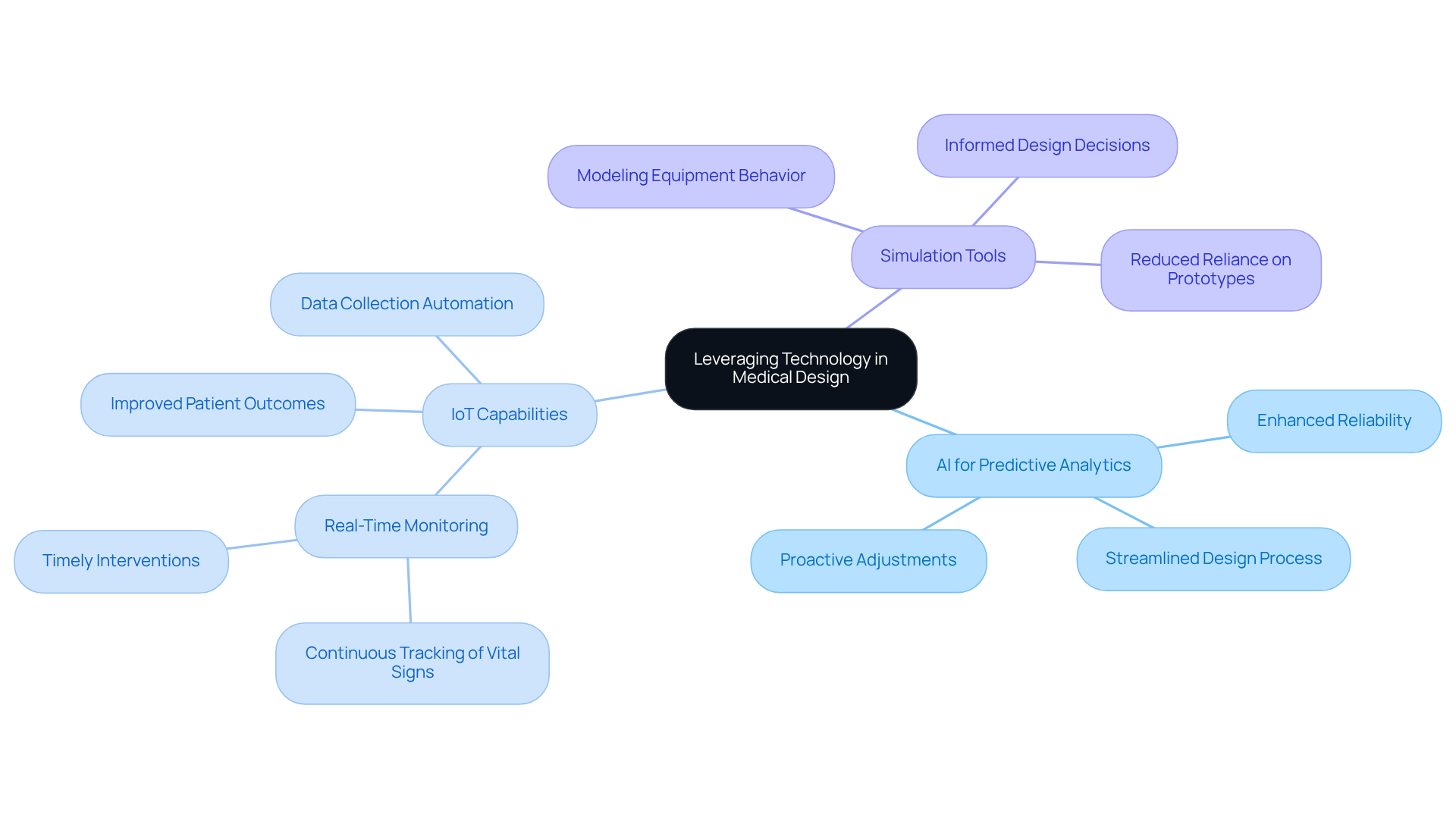
Integrating advanced technologies such as Artificial Intelligence (AI) and the Internet of Things (IoT) is crucial for enhancing the structure and functionality of medical devices. Here are best practices for leveraging these technologies:
By adopting these technologies, engineers can improve the mechatronics design process to create medical tools that are more efficient and reliable. These tools meet the evolving needs of healthcare providers and patients. The integration of IoT not only enhances the functionality of equipment but also supports operational efficiency in healthcare environments, allowing for improved patient monitoring and streamlined processes. For example, case studies on remote patient monitoring, including those from Voler Systems, show how IoT technology facilitates continuous tracking of patients' vital signs, resulting in early interventions and enhanced patient outcomes. As emphasized by Brian Charlesworth, CEO of Ultraviolet, "AI is transforming the manner in which healthcare instruments are created, developed, and introduced to the market - from innovative surgical tools to next-gen health wearables." The adoption of these technologies is not merely advantageous; it is imperative for medical device manufacturers aiming to thrive in a competitive and rapidly evolving market. For further insights, check out our resources section to explore the latest electronic design trends and success stories.
The integration of mechatronics in medical device design presents both opportunities and challenges that are critical to advancing healthcare technology. Integrating mechanical and electronic systems enables engineers to create solutions that enhance patient care and operational efficiency. Understanding the core principles of mechatronics-integration, regulatory frameworks, and feedback mechanisms serves as the foundation for developing reliable healthcare technologies that meet rigorous standards.
This article emphasizes how interdisciplinary collaboration, adherence to regulatory standards, and the strategic application of technologies like AI and IoT are essential for success. Establishing clear communication, fostering cross-disciplinary training, and implementing agile methodologies are vital practices that drive successful outcomes in healthcare equipment design. Furthermore, navigating regulatory landscapes and adopting modern technologies not only mitigate risks but also ensure that medical devices are safe, effective, and aligned with market demands.
The significance of mastering the mechatronics design process cannot be overstated. As the healthcare industry continues to evolve, embracing these best practices will be crucial for engineers and manufacturers aiming to create cutting-edge medical devices. By prioritizing collaboration, compliance, and technological integration, stakeholders can contribute to improved patient outcomes and a more efficient healthcare system. Ultimately, the commitment to these principles will determine the future success of medical device innovation and patient safety.
What is mechatronics?
Mechatronics is the convergence of multiple engineering disciplines that is essential for advancing intelligent systems, particularly in healthcare. It involves the integration of components like sensors, actuators, and control systems to enhance the functionality and performance of medical equipment.
What are the core principles of mechatronics in healthcare technology?
The core principles include Integration, Regulation Frameworks, and Feedback Mechanisms. Integration focuses on combining mechanical and electronic systems, Regulation Frameworks involve algorithms and software for operational oversight, and Feedback Mechanisms use sensors for real-time information to adapt equipment operations.
How does integration work in mechatronics?
Integration in mechatronics involves the seamless combination of mechanical and electronic systems, enabling devices to perform complex tasks autonomously. This is crucial for creating dependable systems in critical healthcare environments.
What role do regulation frameworks play in healthcare technology?
Regulation frameworks involve algorithms and software that oversee equipment operations, ensuring accuracy and dependability, which are vital for the safe functioning of medical instruments.
How do feedback mechanisms enhance healthcare equipment?
Feedback mechanisms utilize sensors to provide real-time information, allowing for dynamic modifications in equipment operations. This enhances user safety and ensures that health equipment can adapt to changing conditions.
Why is a comprehensive understanding of mechatronics principles important for engineers?
A comprehensive understanding of mechatronics principles is vital for engineers involved in the design process of healthcare equipment, as it determines the operational capabilities of devices and ensures adherence to safety and performance standards.
How is the mechatronics design process expected to evolve in the healthcare industry?
As the healthcare industry shifts towards value-based models, the mechatronics design process is expected to demonstrate measurable improvements in function and recovery, highlighting the importance of effectively integrating mechatronics principles to enhance patient outcomes and operational efficiency.
