Best Practices for Embedded Services in Medical Device Development
Introduction In the complex landscape of medical device development, regulatory...

In the healthcare sector, navigating the regulatory landscape poses significant challenges that can critically impact the success of medical devices. Companies must not only adhere to stringent regulatory standards but also leverage cutting-edge technologies to enhance reliability and performance. Organizations must effectively navigate the complexities of product development to ensure compliance and foster innovation. This article explores best practices for industrial product development in medical devices, providing insights that empower teams to navigate obstacles and achieve sustainable growth.
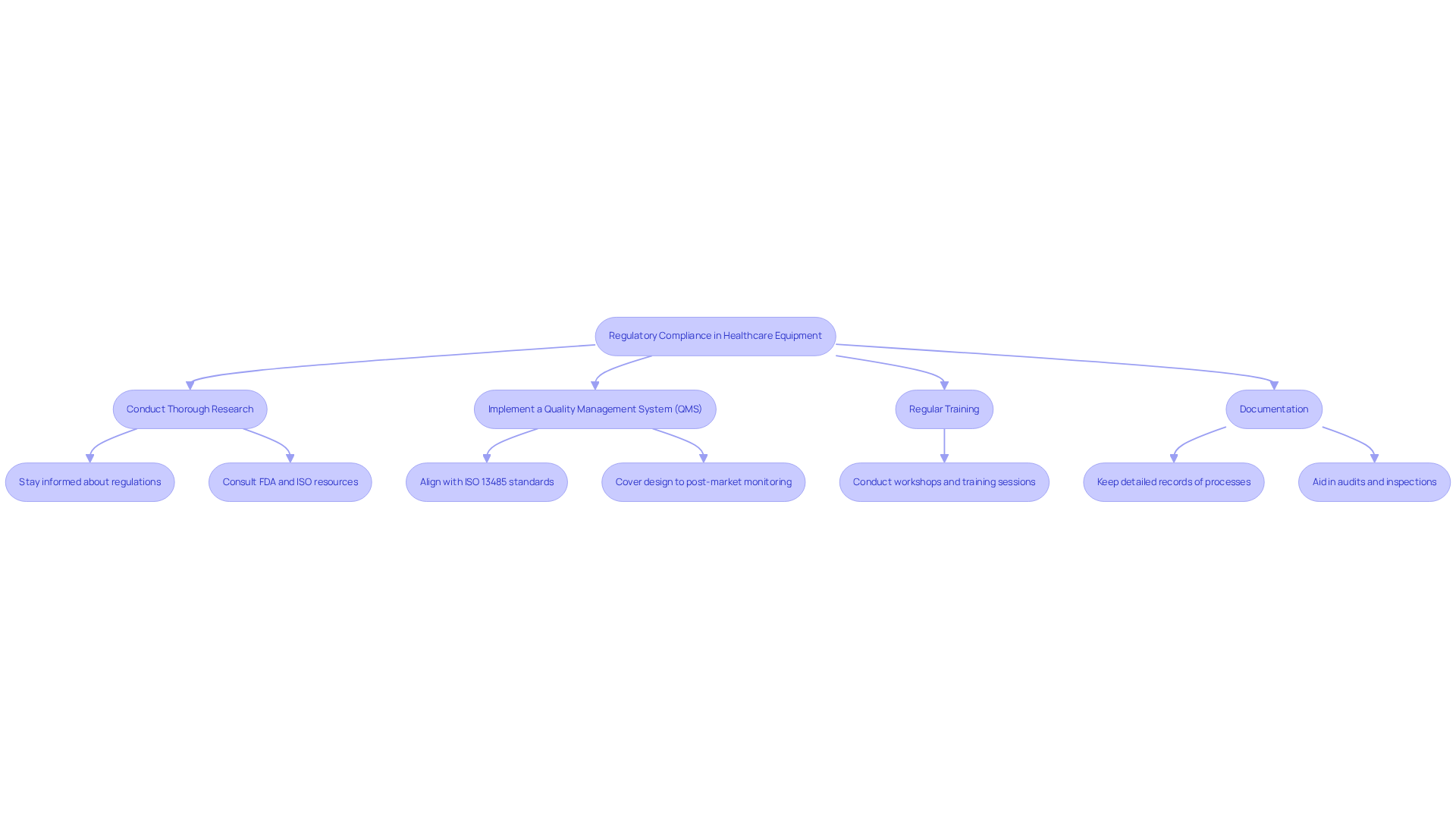
In the healthcare equipment sector, navigating regulatory compliance is a critical challenge that can significantly impact a company's success. Comprehending regulations such as the FDA's Quality System Regulation (QSR) and ISO 13485 is essential, as these standards govern the design, manufacturing, and post-market monitoring of health-related products.
Key Strategies for Compliance:
Real-World Example: A healthcare equipment company that adopted a comprehensive QMS experienced a 30% reduction in compliance-related issues during FDA inspections, underscoring the effectiveness of these practices. The implementation of a robust QMS not only mitigates compliance risks but also positions startups for sustainable growth in a competitive market.
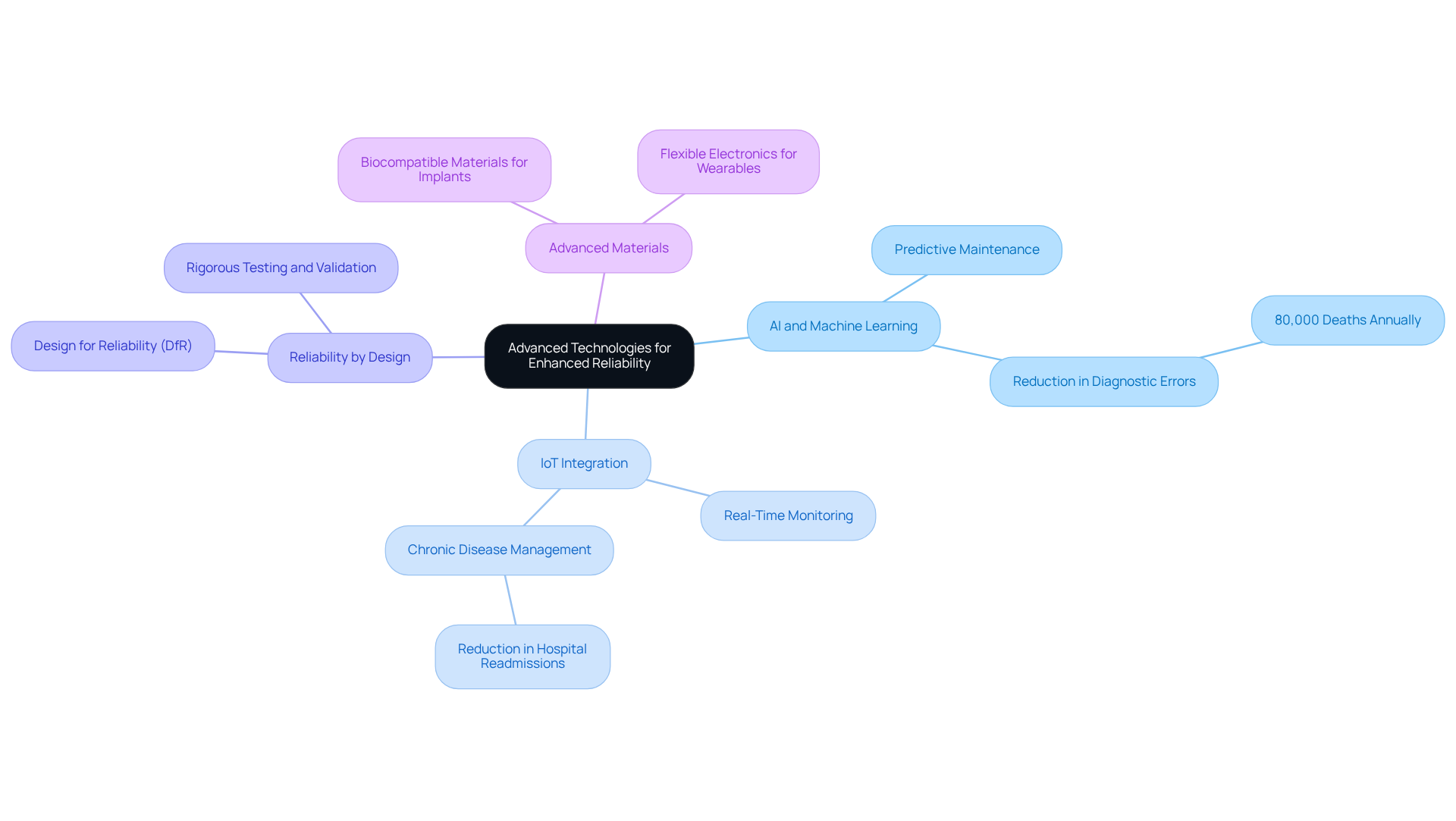
Integrating advanced technologies into medical equipment development is not just beneficial; it is essential for enhancing reliability and performance in healthcare. AI, machine learning, and IoT are critical technologies that drive innovative solutions, significantly improving patient care and operational efficiency.
Best Practices:
Real-World Example: A wearable health monitoring tool that integrated IoT capabilities enabled continuous patient monitoring, resulting in a 40% reduction in emergency hospital visits due to timely interventions based on real-time data. This case illustrates the transformative impact of IoT integration on patient care and operational efficiency in healthcare settings.
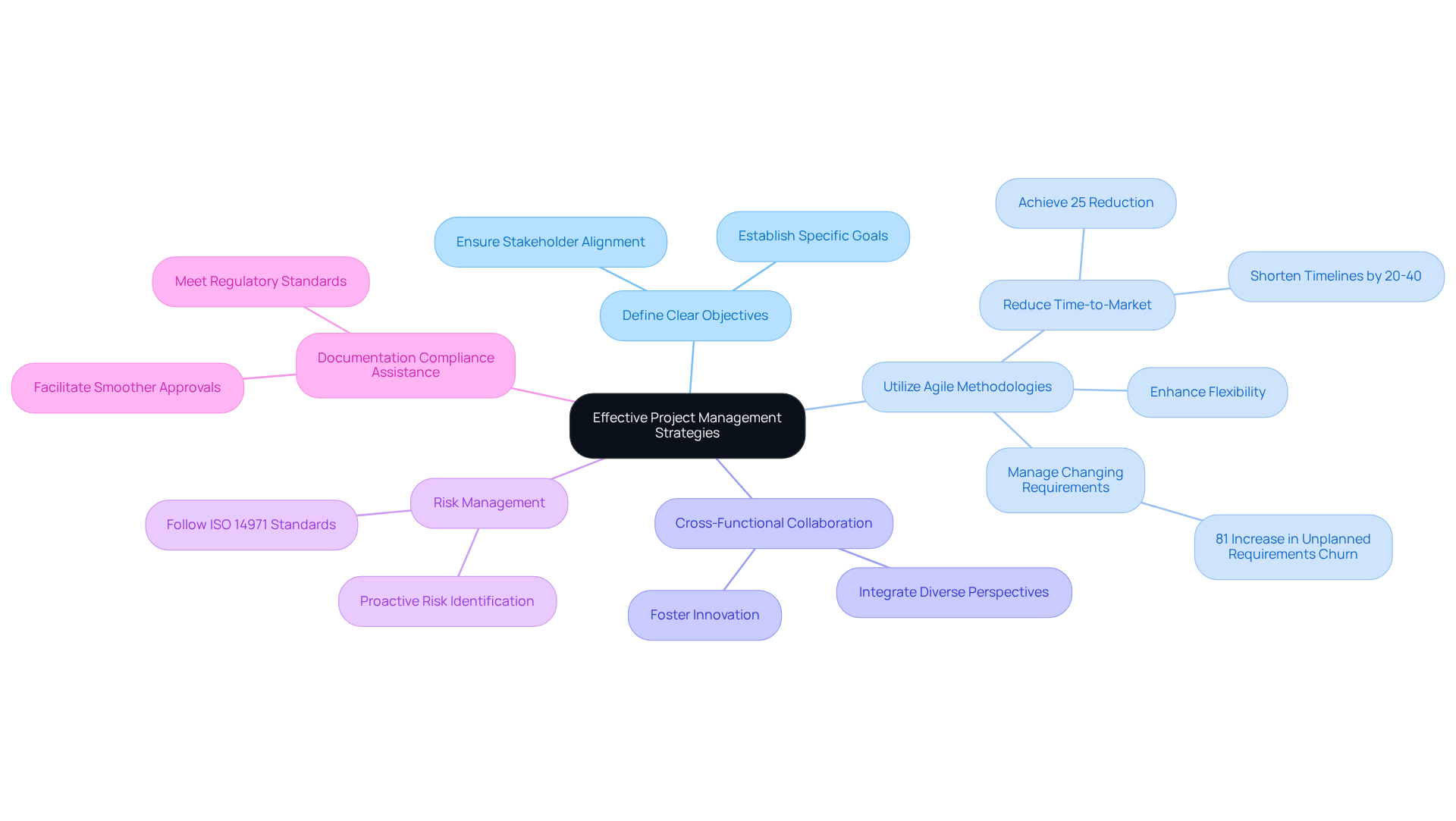
Navigating the complexities of regulatory compliance and technical challenges can hinder project success in the healthcare equipment sector. However, applying efficient strategies can significantly enhance the creation process, particularly by leveraging Voler Systems' expertise in innovative healthcare product design through software development and advanced engineering solutions.
Define Clear Objectives: Establish specific, measurable goals for each project phase. This clarity helps teams work together effectively and keeps everyone informed about progress, ensuring that all stakeholders are aligned.
Utilize Agile Methodologies: Adopting agile project management techniques enhances flexibility and responsiveness to changes throughout the creation process. With its iterative approach, agile allows teams to adapt to changing requirements as they arise, which is crucial in a regulated environment where compliance is paramount. Notably, the healthcare equipment sector has experienced a 29% rise in the time taken to specify requirements and an 81% rise in unexpected requirements turnover, underscoring the necessity for agile practices. Organizations that embrace agile methodologies, similar to those supported by Voler Systems, can achieve a 25% reduction in time-to-market for new offerings, consistent with research indicating that agile-oriented design approaches can shorten healthcare product creation timelines by 20% to 40%.
Cross-Functional Collaboration: Encourage collaboration among various departments, including engineering, regulatory, and marketing. This ensures that all viewpoints are integrated into the progress process, fostering innovation and compliance, which are critical for successful project outcomes.
Risk Management: Implement a proactive risk management plan that identifies potential risks early and outlines mitigation strategies. This method is vital for managing the intricacies of industrial product development in healthcare, where compliance with regulations is crucial. Following standards such as ISO 14971, which emphasizes risk management for healthcare products, is essential for guaranteeing compliance and reliability in product design.
Documentation Compliance Assistance: Integrating documentation compliance assistance is essential for startups in the healthcare equipment sector. Ensuring that all documentation meets regulatory standards not only facilitates smoother approvals but also enhances product reliability and safety.
Common Pitfalls: It is also crucial to identify possible challenges in agile implementation, such as the cultural resistance mentioned by 72% of healthcare equipment R&D leaders. For instance, a company that faced challenges in adopting agile methodologies found that addressing team concerns and providing adequate training significantly improved their transition. Proactively addressing these challenges can transform potential pitfalls into opportunities for innovation and success in engineering design projects.
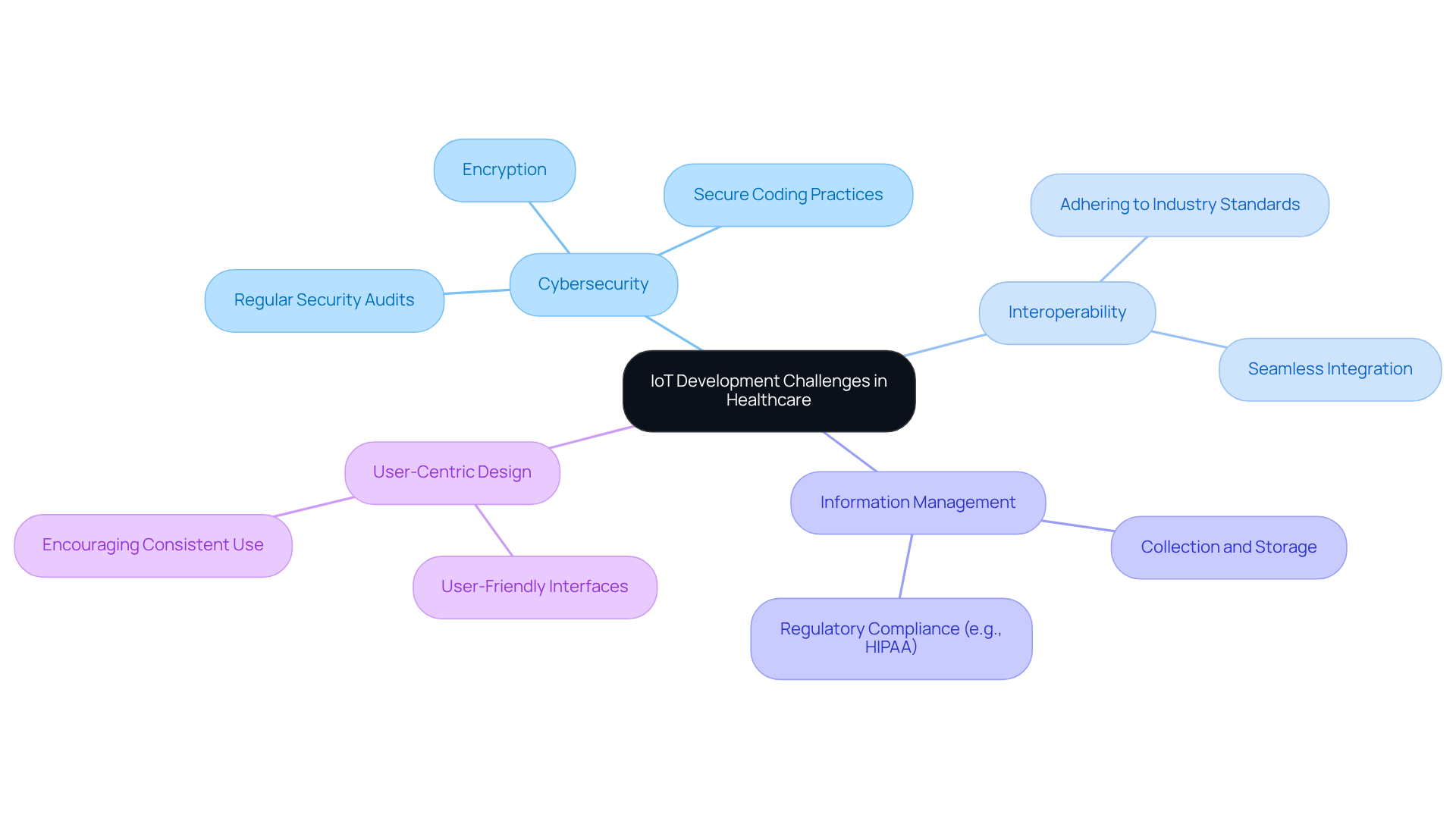
Integrating IoT technologies in healthcare equipment often leads to significant security and interoperability challenges. Addressing these challenges effectively can lead to enhanced product reliability and patient safety.
Real-World Example: A connected insulin delivery system that emphasized cybersecurity and interoperability successfully reduced data breaches by 50%. The need for robust security measures is underscored by the rising incidence of healthcare-related ransomware attacks, which increased by 30% in 2025.
In medical device development, the adherence to established practices is essential for achieving compliance and success. Organizations can navigate the complexities of this highly regulated industry by implementing regulatory standards, leveraging advanced technologies, and adopting effective project management strategies. These practices facilitate smoother product development, as evidenced by case studies showing improved patient safety and operational efficiency.
Key insights from this discussion highlight the importance of:
Each of these elements plays a vital role in mitigating risks, fostering innovation, and ensuring that medical devices meet the highest standards of quality and safety. Real-world examples demonstrate how these strategies have led to significant improvements in compliance and patient outcomes, underscoring their effectiveness in practice.
Embracing these best practices is crucial for organizations aiming to thrive in a competitive landscape. As the medical device landscape continues to evolve, staying ahead of regulatory changes and technological advancements will be crucial. Organizations should invest in training, adopt innovative solutions, and foster collaboration to meet current standards and prepare for future advancements in healthcare technology. The commitment to excellence in medical device development will not only enhance market competitiveness but also contribute to better health outcomes for patients worldwide.
Why is regulatory compliance important in the healthcare equipment sector?
Regulatory compliance is crucial as it significantly impacts a company's success by ensuring that products meet safety and quality standards, thereby protecting public health.
What are some key regulations that healthcare equipment companies need to understand?
Companies need to understand the FDA's Quality System Regulation (QSR) and ISO 13485, which govern the design, manufacturing, and post-market monitoring of health-related products.
What is a Quality Management System (QMS) and why is it important?
A QMS is a structured system that aligns with ISO 13485 standards, covering all aspects of product creation from design to post-market monitoring. It is essential for achieving compliance documentation and streamlining regulatory processes, particularly for startups.
How can companies stay updated on regulatory standards?
Companies should conduct thorough research and regularly consult resources such as the FDA and ISO websites to stay informed about the latest regulations and guidelines.
Why is regular training necessary for team members in healthcare equipment companies?
Regular training ensures that all team members are well-versed in compliance requirements and quality control processes, helping to maintain high standards within the organization.
What role does documentation play in regulatory compliance?
Detailed documentation of all design and manufacturing processes is vital for audits and inspections, and it helps identify areas for improvement, especially for startups facing regulatory challenges.
Can you provide an example of the benefits of implementing a Quality Management System?
A healthcare equipment company that adopted a comprehensive QMS experienced a 30% reduction in compliance-related issues during FDA inspections, highlighting the effectiveness of these practices in mitigating compliance risks and supporting sustainable growth.
