Introduction
Crafting a Product Requirements Document (PRD) for medical devices is not merely a procedural step; it is a critical determinant of project success in a high-stakes industry where clarity can significantly influence the balance between innovation and failure. Approximately 70% of healthcare equipment projects encounter setbacks due to unclear requirements, underscoring the necessity of a robust PRD. This article explores the essential components and best practices for developing a PRD that not only complies with regulatory standards but also aligns with user needs, thereby enhancing the likelihood of successful product outcomes.
How can stakeholders ensure that their PRD functions as a dynamic tool, adapting to the evolving landscape of healthcare technology?
Define the Product Requirements Document (PRD) for Medical Devices
A (PRD) for medical devices is an essential formal document that delineates the critical requirements and specifications for a product. It acts as a comprehensive blueprint for the development team, detailing the objectives the product must fulfill and the constraints it must adhere to. A well-structured PRD includes:
- User needs
- Design constraints
By clearly articulating these elements, the PRD fosters a shared understanding among all stakeholders regarding project objectives, which is vital for and compliance with industry standards.
Statistics indicate that approximately 70% of healthcare equipment projects fail due to ambiguous requirements, underscoring the necessity of a robust PRD. Successful examples of PRDs in illustrate how clarity in requirements can lead to timely and cost-effective project completion. For instance, companies that have adopted comprehensive PRDs have reported significant reductions in development time and costs, with some achieving up to an 18-month decrease in time to market.
The importance of a PRD cannot be overstated; it not only guides the development process but also ensures that the final product meets both market demands and regulatory expectations. As the healthcare instrument landscape evolves, the incorporation of into PRDs is becoming increasingly vital, allowing manufacturers to adapt to changing demands and enhance product efficacy. In this context, a well-crafted PRD serves as a foundational element that supports innovation and compliance in the rapidly evolving healthcare equipment industry. plays a pivotal role in this process, offering for startups in , ensuring that projects are not only successful but also adhere to necessary regulations.
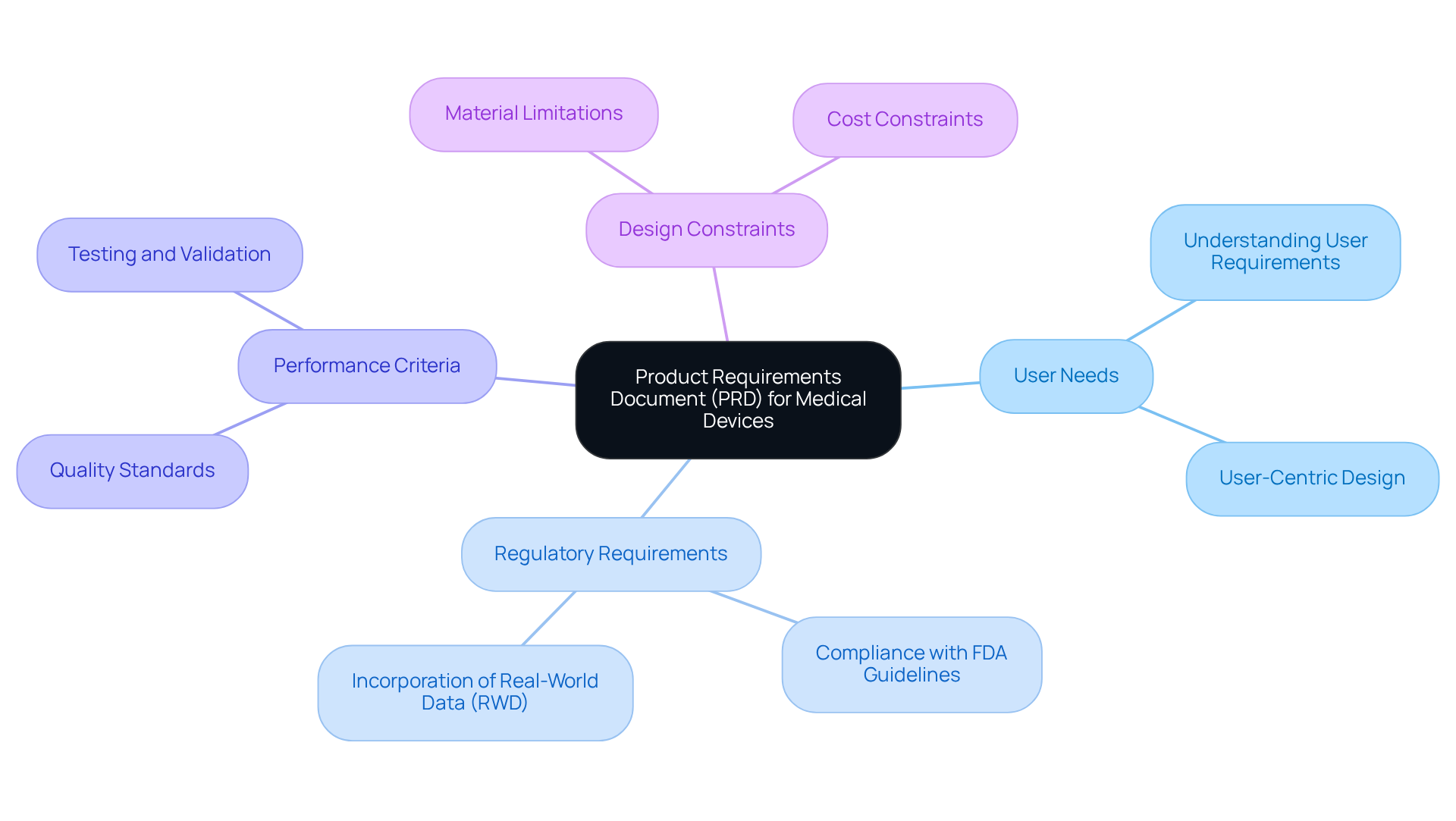
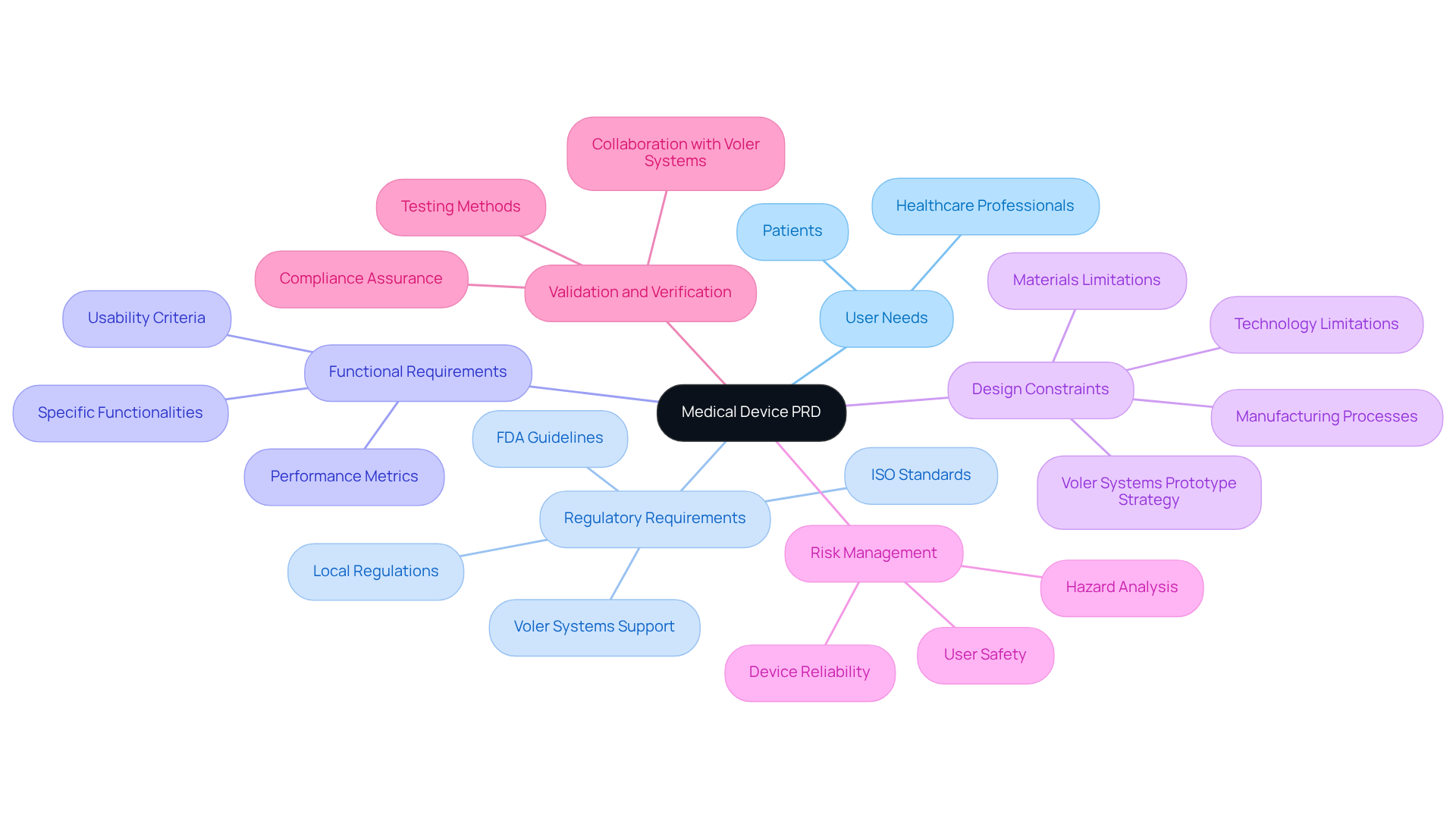
Identify Key Components of a Medical Device PRD
Key components of a medical device (PRD) include:
- User Needs: Clearly articulate the needs of end-users, encompassing both healthcare professionals and patients.
- Regulatory Requirements: Outline the applicable regulations and standards that the equipment must comply with, such as FDA guidelines, ISO standards, and any relevant local regulations. , assisting startups in effectively navigating these regulatory challenges.
- Functional Requirements: Specify the essential functions of the equipment, including performance metrics, usability criteria, and specific functionalities that address user needs.
- Design Constraints: Identify limitations related to materials, manufacturing processes, or technology. Voler Systems' facilitates adjustments to designs for production, ensuring that these constraints are addressed early in the development process.
- Risk Management: Include a to address potential hazards associated with the device, focusing on user safety and device reliability.
- Validation and Verification: Define the testing methods to ensure the item meets the specified requirements. By collaborating with Voler Systems, startups can streamline their , ensuring compliance from prototype to production.
Incorporating these elements allows managers to develop a robust PRD that serves as a solid foundation for the development process, supported by .
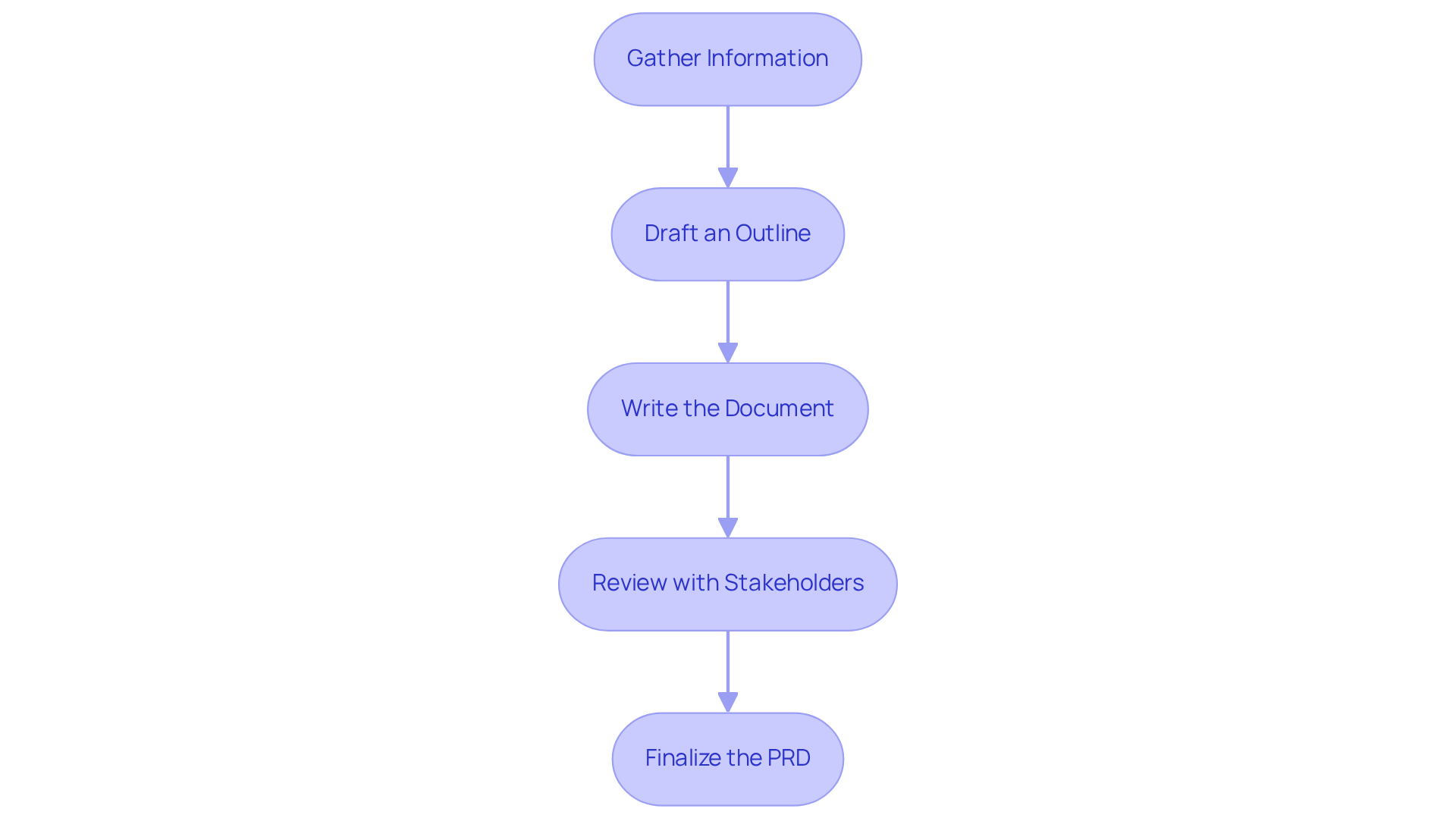
Implement a Step-by-Step Approach to Writing a PRD
To create an effective (PRD) for , follow these structured steps:
- Gather Information: Engage with stakeholders through interviews to capture and understand . This foundational step ensures that the PRD reflects both market demands and .
- Draft an Outline: Develop a comprehensive outline that incorporates key components identified during the information-gathering phase. A well-structured outline serves as a roadmap for the document.
- Write the Document: Populate the outline with detailed descriptions, focusing on . Each section should express the vision and requirements of the product in a manner that is easily comprehensible.
- Review with Stakeholders: Circulate the draft among . This collaborative review process is crucial for identifying gaps and ensuring that all perspectives are considered.
- Finalize the PRD: Complete the document by verifying its accuracy and alignment with regulatory standards. A thorough before obtaining approval, ensuring that the PRD is both comprehensive and compliant.
By employing this organized method, product managers can develop a PRD that clearly conveys the and requirements, ultimately enhancing the success rates of development.
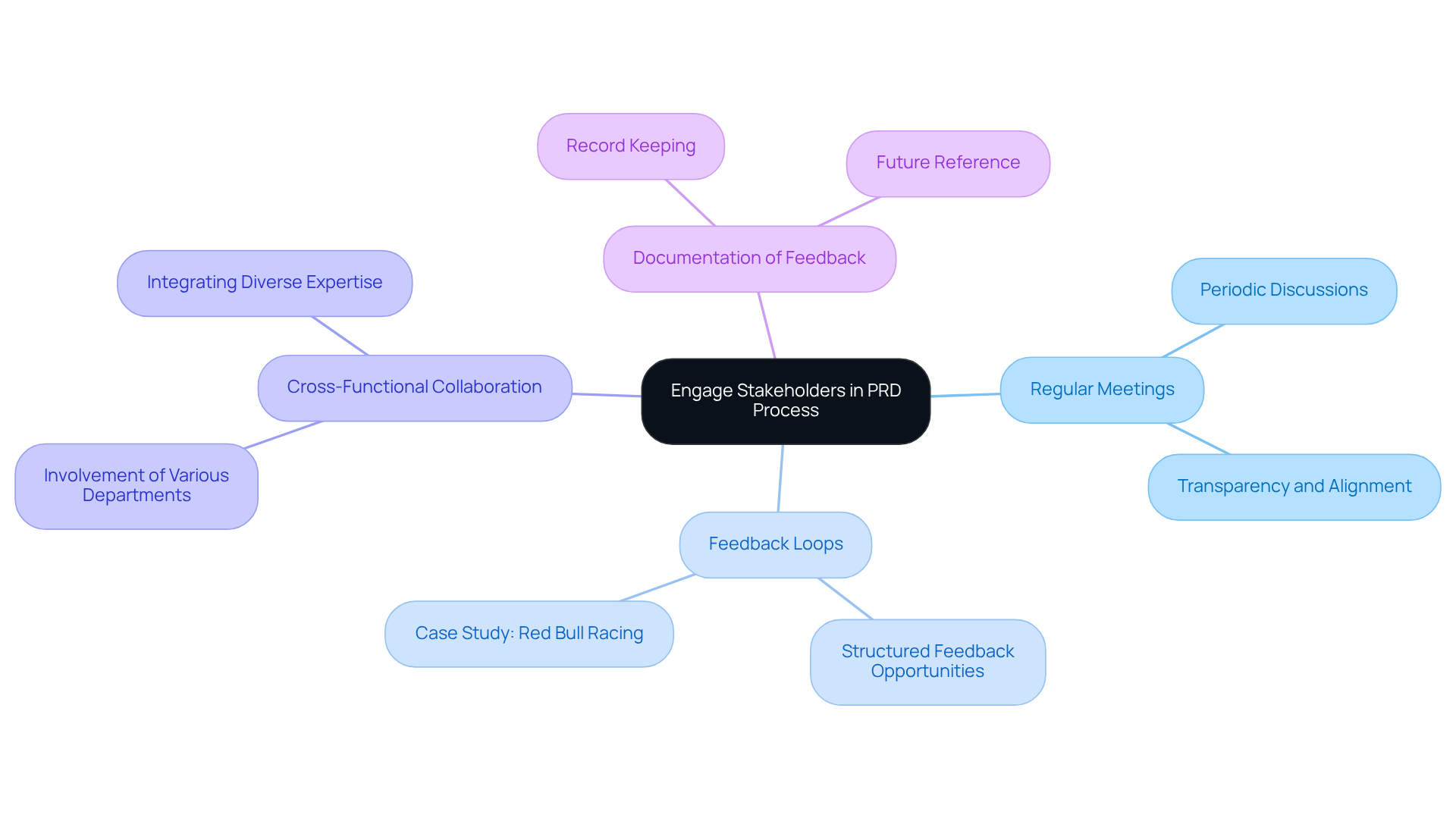
Engage Stakeholders and Gather Feedback Throughout the PRD Process
throughout the process is essential for developing effective medical products. Key strategies include:
- Regular Meetings: Schedule periodic meetings with stakeholders to discuss progress and gather input. These interactions promote transparency and ensure alignment on project objectives, demonstrating ' commitment to in .
- : Establish structured opportunities for stakeholders to provide feedback on drafts and revisions. Research indicates that organizations employing can significantly enhance their development efficiency, resulting in a more polished and market-ready product. For example, a case study on Red Bull Racing illustrated how optimized their tire-changing process, highlighting the impact of continuous improvement on operational efficiency. Similarly, has effectively implemented feedback mechanisms in their projects, ensuring compliance with emissions and ESD standards through a thorough review process.
- : Involve team members from various departments - engineering, marketing, regulatory affairs, and more - to ensure a comprehensive perspective. Successful often relies on this collaboration, as it integrates diverse expertise and viewpoints, ultimately enhancing the product's design and functionality. An examination of participatory methods in healthcare product development revealed that effective teamwork can address challenges such as managing knowledge and health outcomes, a principle that exemplifies in their project execution.
- Documentation of Feedback: Maintain a detailed record of all feedback received and how it was addressed in the PRD. This practice not only fosters accountability but also serves as a valuable reference for future projects, aligning with ' commitment to thorough for startups in healthcare technology.
By actively and fostering a culture of teamwork, managers can create a that reflects a consensus and meets the diverse needs of the project, ultimately leading to successful healthcare outcomes.
Review and Iterate the PRD for Continuous Improvement
To ensure the effectiveness of the , it is essential to implement a that encompasses the following elements:
- Scheduled Reviews: Establish regular intervals for PRD assessments to guarantee its relevance and accuracy in response to evolving project needs.
- Incorporate New Insights: Continuously update the document with new information, such as , to maintain alignment with current requirements.
- Version Control: Maintain a to track modifications, ensuring that all stakeholders are informed of the latest updates and changes.
- Feedback from Testing: Utilize insights gained from testing the item to refine the PRD, thereby .
By committing to these , managers can significantly enhance the quality of the PRD, ultimately guiding the development of successful . Research indicates that teams with well-structured PRDs experience faster and fewer late-stage surprises, which directly contributes to project success. Emphasizing a culture of not only mitigates risks but also aligns product features with user needs and business priorities, ensuring that the right product is built from the outset.
Conclusion
A well-crafted Product Requirements Document (PRD) is essential for the successful development of medical devices. It acts as a comprehensive blueprint that outlines critical requirements and specifications while fostering a shared understanding among all stakeholders involved in the project. By emphasizing clarity and precision, a robust PRD significantly reduces the risks of project failure, which statistics indicate can occur in nearly 70% of healthcare equipment projects due to ambiguous requirements.
This article highlights key components of an effective PRD, including:
- User needs
- Regulatory requirements
- Functional specifications
- Risk management strategies
The systematic, step-by-step approach to writing a PRD ensures that all necessary elements are thoroughly addressed. Furthermore, the focus on stakeholder engagement and continuous improvement underscores the importance of collaboration and adaptability throughout the development process. By incorporating feedback loops and conducting regular reviews, product managers can refine the PRD, ensuring it aligns with evolving project needs and regulatory changes.
In conclusion, mastering the PRD for medical devices transcends mere documentation; it lays the groundwork for innovation and compliance in a rapidly evolving industry. By adhering to best practices and actively involving stakeholders, organizations can enhance their development processes, ultimately leading to successful healthcare outcomes. Embracing these principles will streamline product development and ensure that the final products are effective, safe, and aligned with market demands.
Frequently Asked Questions
What is a Product Requirements Document (PRD) for medical devices?
A PRD for medical devices is a formal document that outlines the critical requirements and specifications for a product, serving as a comprehensive blueprint for the development team.
What are the key components included in a medical device PRD?
Key components of a medical device PRD include user needs, regulatory requirements, functional requirements, design constraints, risk management, and validation and verification.
Why is a PRD important in medical device development?
A PRD is important because it fosters a shared understanding among stakeholders, guides the development process, and ensures that the final product meets market demands and regulatory expectations.
What role does Voler Systems play in the PRD process?
Voler Systems provides comprehensive documentation compliance assistance for startups in health technology, helping to ensure projects are successful and adhere to necessary regulations.
How does incorporating real-world data (RWD) into a PRD benefit medical device development?
Incorporating RWD into a PRD allows manufacturers to adapt to changing demands and enhance product efficacy, supporting innovation and compliance in the healthcare equipment industry.
What impact can a well-structured PRD have on project timelines and costs?
A well-structured PRD can lead to significant reductions in development time and costs, with some companies achieving up to an 18-month decrease in time to market.
What statistics highlight the necessity of a robust PRD in healthcare equipment projects?
Statistics indicate that approximately 70% of healthcare equipment projects fail due to ambiguous requirements, underscoring the importance of having a robust PRD.
List of Sources
- Define the Product Requirements Document (PRD) for Medical Devices
- The Crucial Role of Real-World Data for Medical Device Companies (https://premierinc.com/newsroom/the-crucial-role-of-real-world-data-for-medical-device-companies)
- Navigate 2026's Medical Device Manufacturing Trends (https://mddionline.com/medical-device-regulations/navigate-2026-s-medical-device-manufacturing-trends)
- FDA Issues Updated Guidance Loosening Regulatory Approach to Certain Digital Health Tools (https://lw.com/en/insights/fda-issues-updated-guidance-loosening-regulatory-approach-to-certain-digital-health-tools)
- FDA Updates Broaden Wellness and Clinical Decision Support Software Devices Under Enforcement (https://emergobyul.com/news/fda-updates-broaden-wellness-and-clinical-decision-support-software-devices-under-enforcement)
- FDA Adapts with the Times on Digital Health: Updated Guidances on General Wellness Products and Clinical Decision Support Software | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/01/fda-adapts-with-the-times-on-digital-health-updated-guidances-on-general-wellness-products)
- Implement a Step-by-Step Approach to Writing a PRD
- Top Medical Device Regulations to Watch in 2026 (https://xtalks.com/top-medical-device-regulations-to-watch-in-2026-4549)
- Medical Device Compliance: 2025 Insights & 2026 Priorities (https://medenvoyglobal.com/blog/medical-device-compliance-2025-insights-2026-priorities)
- Medical Product Development Trends - 2026 | IDP (https://innovativedesignproducts.com/medical-product-development-trends-2026)
- Digest: January 2026 Featured news that matters in medical writing and communications - Emtex Life Science (https://emtexlifescience.com/resources/digest-january-2026-featured-news-that-matters-in-medical-writing-and-communications)
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
- Engage Stakeholders and Gather Feedback Throughout the PRD Process
- An Overview of Stakeholders, Methods, Topics, and Challenges in Participatory Approaches Used in the Development of Medical Devices: A Scoping Review - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10125077)
- Feedback loops in teacher professional development: A qualitative study in Herat Province of Afghanistan (https://tandfonline.com/doi/full/10.1080/2331186X.2021.1954466)
- How Well Does Your Organization Use Feedback Loops? (https://forbes.com/sites/danielnewman/2016/08/02/how-well-does-your-organization-use-feedback-loops)
- CDRH Proposed Guidances for Fiscal Year 2026 (FY2026) (https://fda.gov/medical-devices/guidance-documents-medical-devices-and-radiation-emitting-products/cdrh-proposed-guidances-fiscal-year-2026-fy2026)
- Stakeholders’ engagement for improved health outcomes: a research brief to design a tool for better communication and participation - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11979262)
- Review and Iterate the PRD for Continuous Improvement
- Product Requirements Document (PRD): Purpose & Best Practices (https://fictiv.com/articles/prd-product-requirements-document)
- 5 Critical Trends for Compliance and Innovation (https://mddionline.com/medical-device-regulations/2026-medical-device-quality-5-critical-trends-for-compliance-and-innovation)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)






