Introduction
Failure Mode and Effects Analysis (FMEA) is a fundamental component in ensuring safety and reliability in the development of medical devices. As the healthcare landscape continues to evolve, the stakes for manufacturers are increasingly high; even a single oversight can result in severe consequences for patient safety and regulatory compliance.
This article explores the detailed steps involved in conducting an effective FMEA analysis, providing insights into proactive risk identification and mitigation, as well as strategies for enhancing product design and compliance.
What challenges may emerge during this essential process, and how can manufacturers effectively navigate these obstacles to ensure their devices not only meet but surpass safety standards?
Define FMEA and Its Importance in Medical Device Development
Failure Mode and Effects Assessment (FMEA) is a systematic approach designed to identify potential malfunction types in products or processes, assess their impacts, and prioritize them based on severity and likelihood of occurrence. In the realm of medical equipment development, FMEA is crucial as it enables manufacturers to foresee and mitigate risks related to functionality and safety. By implementing FMEA, teams can address potential failures during the design phase, thereby significantly enhancing product reliability and ensuring compliance with regulatory standards such as ISO 14971. This proactive strategy not only safeguards patient safety but also streamlines the development process, reducing the likelihood of costly redesigns and regulatory hurdles.
Given the increasing demand for medical equipment, Voler Systems supports manufacturers by leveraging its expertise in developing a diverse array of products, including:
- Wearable technology
- Heart pumps
- Liquid biopsy platforms
Recent case studies indicate that a considerable percentage of medical adverse events can be prevented through effective risk management practices like FMEA, highlighting the significance of addressing FMEA failure in safeguarding healthcare outcomes. Additionally, ongoing regulatory updates highlight the necessity of incorporating FMEA into quality management systems, reinforcing its position as a fundamental element of medical device safety.
Conduct Step-by-Step FMEA Analysis for Your Device
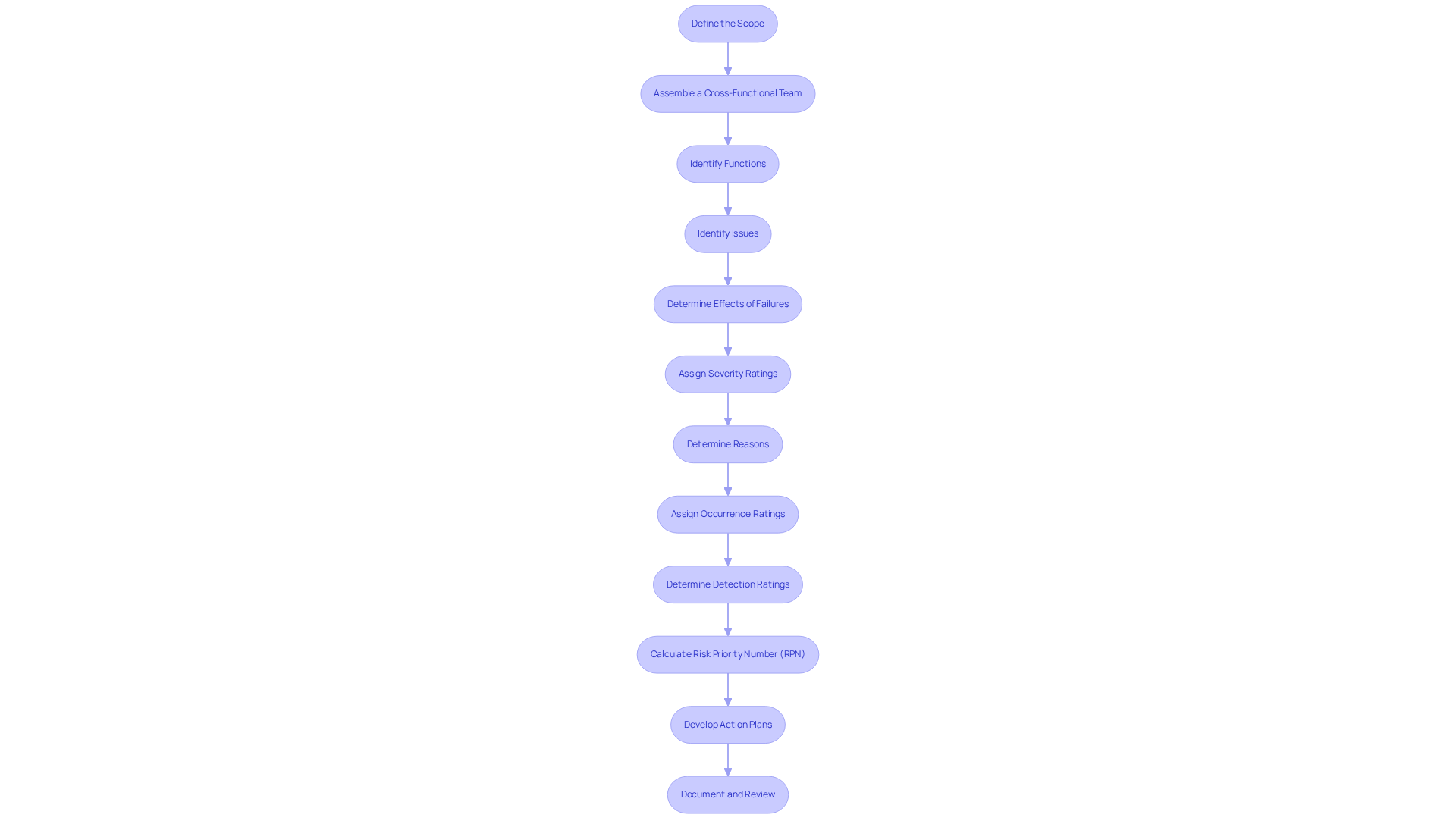
To conduct a thorough FMEA analysis, follow these steps:
- Define the Scope: Clearly outline the system, subsystem, or component under analysis. Establish boundaries to focus the analysis effectively, ensuring compliance with regulatory standards.
- Assemble a Cross-Functional Team: Gather a team with diverse expertise, including design, engineering, quality assurance, and regulatory affairs. A multidisciplinary group enhances the detection of error modes and their impacts, improving overall analysis quality and ensuring that all regulatory aspects are considered.
- Identify Functions: List the intended functions of the apparatus or system. Understanding what the device is designed to accomplish is essential for recognizing potential issues and ensuring adherence to industry regulations.
- Identify Issues: For each function, brainstorm possible problem scenarios. Consider how each function could potentially face FMEA failure in performing as intended, utilizing historical data and previous analyses to ensure comprehensive identification.
- Determine Effects of Failures: Assess the consequences of each failure mode. Evaluate the impact on equipment performance, safety, and regulatory adherence, prioritizing risks according to severity and clinical perspective in relation to FMEA failure.
- Assign Severity Ratings: Rate the severity of each effect on a scale (e.g., 1 to 10), where 10 represents the most severe impact on patient safety or device functionality. A severity rating of 10 indicates potential for death or life-threatening harm.
- Determine Reasons: For each issue type, identify possible causes. This step aids in understanding why a setback, such as FMEA failure, might occur, including design, manufacturing, software, or user handling errors.
- Assign Occurrence Ratings: Rate the likelihood of each cause occurring, again on a scale (e.g., 1 to 10). This quantifies the risk associated with each malfunction mode.
- Determine Detection Ratings: Assess how easily each malfunction can be detected before it reaches the end user. A lower rating suggests a greater chance of undetected issues, highlighting the necessity for effective monitoring and compliance checks.
- Calculate Risk Priority Number (RPN): Multiply the severity, occurrence, and detection ratings to obtain the RPN for each error type. This number assists in prioritizing which issues require prompt attention, especially in the context of FMEA failure, directing risk reduction efforts.
- Develop Action Plans: For high-priority failure modes (high RPN), create action plans to mitigate risks. This may include design changes, additional testing, or enhanced monitoring to ensure compliance with regulatory standards, supported by Voler Systems' expertise in documentation compliance.
- Document and Review: Maintain an active record of the failure mode and effects analysis, performing regular evaluations to revise it as the design progresses or new information is obtained. This ensures that the failure mode and effects analysis remains relevant and effective throughout the product lifecycle, aligning with documentation compliance requirements.
Integrate FMEA Findings into Design and Compliance Strategies
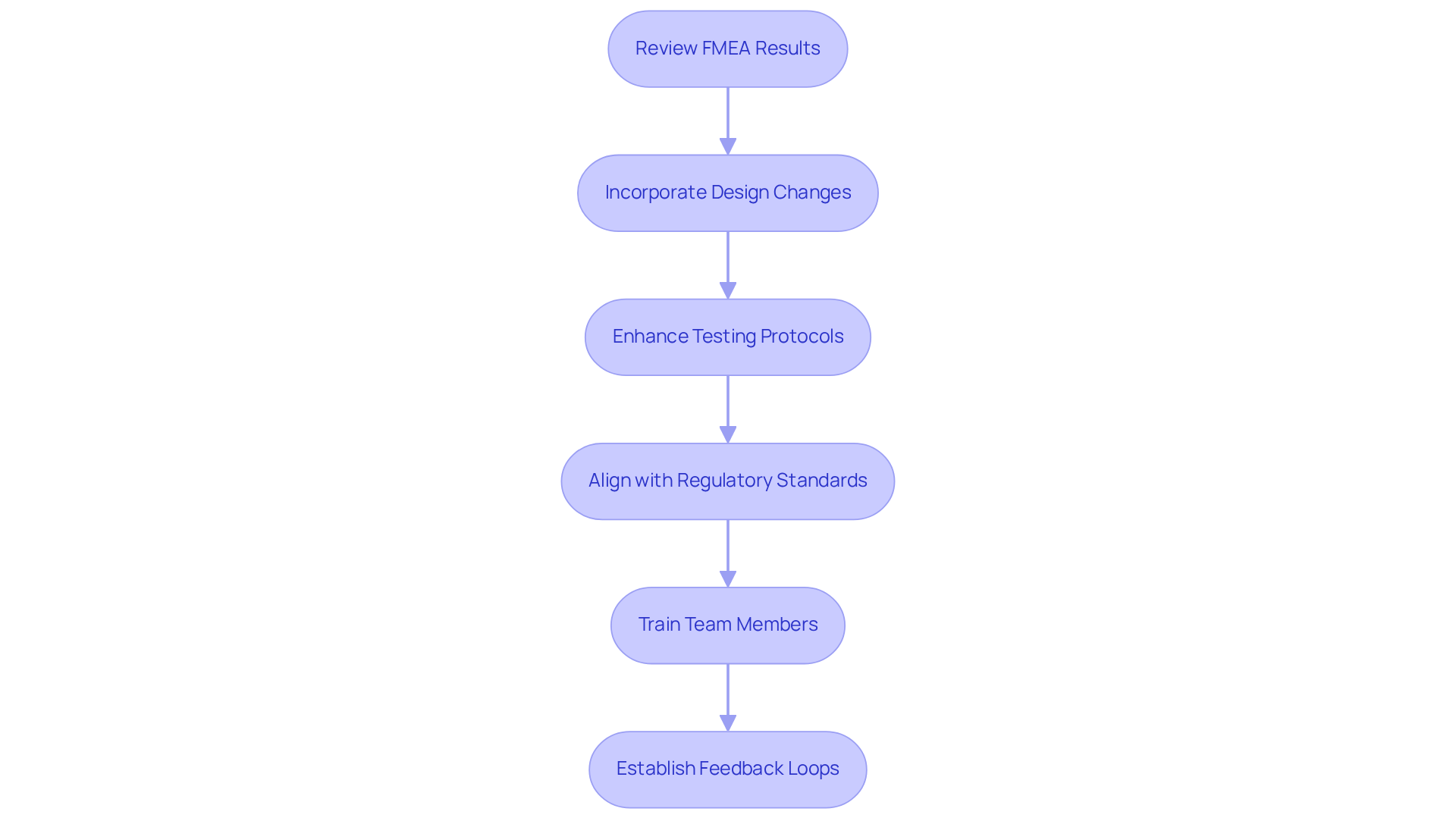
Incorporating failure mode and effects analysis results into design and compliance strategies is essential for enhancing the safety and effectiveness of medical devices. The following key steps should be considered:
- Review Failure Mode and Effects Analysis Results: Conduct regular assessments of failure mode and effects analysis outcomes with the design team. This ensures that all identified risks are understood and prioritized effectively.
- Incorporate Design Changes: Utilize insights from the failure mode and effects analysis to inform necessary design modifications. For example, if a malfunction related to a component's durability is identified, consider using stronger materials or alternative designs to mitigate risks.
- Enhance Testing Protocols: Adjust testing procedures to focus on high-risk areas identified in the failure modes and effects analysis. This may involve implementing additional stress testing or failure mode simulations to address potential issues prior to market release.
- Align with Regulatory Standards: Ensure that findings from the failure mode and effects analysis are thoroughly documented and aligned with regulatory standards, such as those specified in ISO 14971. This documentation is crucial for demonstrating compliance during audits and submissions.
- Train Team Members: Provide comprehensive training for team members on the significance of Failure Mode and Effects Analysis and its application in their roles. This fosters a culture of safety and compliance throughout the organization.
- Establish Feedback Loops: Create mechanisms for ongoing feedback from post-market surveillance and user reports to continuously refine the failure mode and effects analysis process and update design strategies as necessary.
Address Common Challenges and Solutions in FMEA Implementation
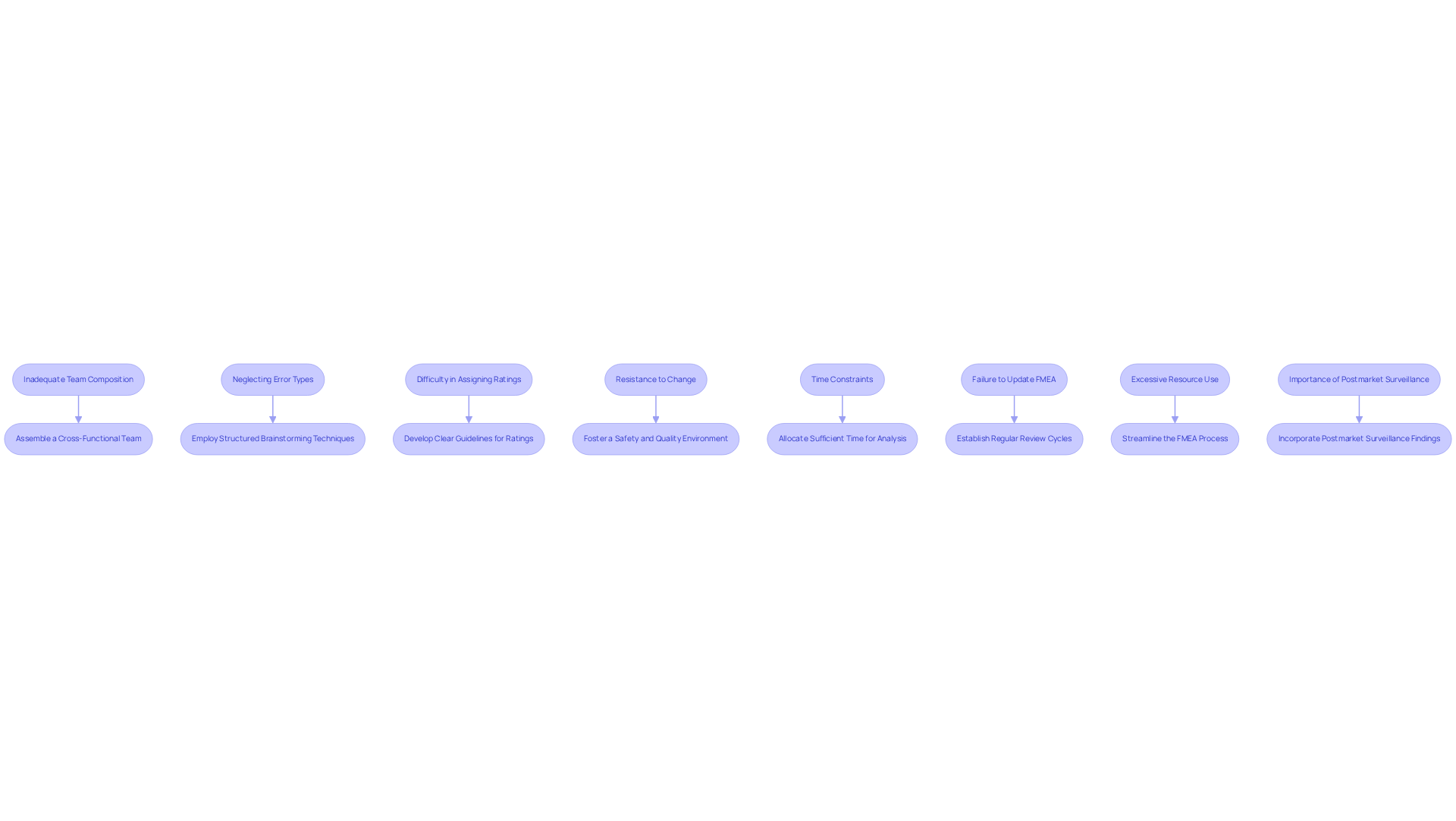
Implementing FMEA failure analysis presents several challenges that require careful consideration. Below are common issues along with suggested solutions:
- Inadequate Team Composition: A lack of diverse expertise can lead to incomplete analyses. To address this, it is essential to assemble a cross-functional team that includes members from design, engineering, quality assurance, and regulatory affairs. This diversity enhances the team's ability to efficiently recognize potential issues.
- Neglecting Error Types: Teams may overlook critical error types due to insufficient brainstorming. To mitigate this, structured brainstorming techniques, such as the Delphi method, should be employed to encourage comprehensive input from all team members, ensuring that no potential FMEA failure is overlooked.
- Difficulty in Assigning Ratings: Teams often struggle with consistently assigning severity, occurrence, and detection ratings. Developing clear guidelines and examples for rating criteria is crucial to ensure uniformity across the team, which is vital for accurate risk assessment.
- Resistance to Change: Team members may resist adopting changes based on findings from the failure mode and effects analysis. It is important to foster an environment that prioritizes safety and quality by emphasizing the significance of FMEA failure in safeguarding patients and maintaining compliance with regulatory standards, which can help reduce resistance.
- Time Constraints: Teams may feel pressured to expedite the failure mode and effects analysis process. Allocating sufficient time for thorough analysis is necessary, along with emphasizing the long-term benefits of a complete FMEA failure analysis in reducing future costs and risks associated with device failures.
- Failure to Update FMEA: FMEAs can become outdated as designs evolve. Establishing regular review cycles for the FMEA document is essential to ensure it addresses potential FMEA failures and remains current and reflective of the latest design changes and risk evaluations, thereby maintaining its relevance throughout the product lifecycle.
- Excessive Resource Use: Conducting FMEA can be resource-intensive, leading to challenges in deployment. Streamlining the FMEA process by utilizing tools and methodologies that enhance efficiency, such as integrating Fault Tree Analysis (FTA), can help reduce complexity.
- Importance of Postmarket Surveillance: Continuous monitoring of medical instruments after approval is crucial for identifying potential risks. Incorporating postmarket surveillance findings into the FMEA process ensures the ongoing safety and effectiveness of the devices.
Conclusion
Implementing Failure Mode and Effects Analysis (FMEA) is crucial for enhancing the safety and reliability of medical devices. By proactively identifying potential failures and their impacts, manufacturers can significantly improve patient safety and ensure compliance with stringent regulatory standards. This systematic approach not only addresses risks during the design phase but also streamlines the development process, ultimately leading to improved healthcare outcomes.
The article presents a comprehensive step-by-step guide for conducting FMEA, highlighting the importance of:
- Assembling a cross-functional team
- Identifying potential failure modes
- Integrating findings into design and compliance strategies
Key challenges in FMEA implementation, such as inadequate team composition and resistance to change, are addressed with practical solutions that promote a culture of safety and continuous improvement.
Ultimately, the significance of FMEA in medical device development is paramount. It serves as a vital tool for manufacturers to mitigate risks, enhance product performance, and uphold patient safety. By adopting FMEA as a fundamental component of their quality management systems, organizations can not only meet regulatory demands but also contribute to the advancement of safer and more effective medical technologies. Taking action now to implement these practices ensures a future where medical devices are designed with the utmost consideration for patient health and safety.
Frequently Asked Questions
What is FMEA?
FMEA stands for Failure Mode and Effects Assessment, which is a systematic approach used to identify potential malfunction types in products or processes, assess their impacts, and prioritize them based on severity and likelihood of occurrence.
Why is FMEA important in medical device development?
FMEA is crucial in medical device development because it enables manufacturers to foresee and mitigate risks related to functionality and safety, thereby enhancing product reliability and ensuring compliance with regulatory standards.
How does FMEA contribute to patient safety?
By implementing FMEA during the design phase, teams can address potential failures, which safeguards patient safety and reduces the likelihood of adverse events.
What are some benefits of using FMEA in the development process?
Benefits of using FMEA include improved product reliability, streamlined development processes, reduced likelihood of costly redesigns, and fewer regulatory hurdles.
What regulatory standards emphasize the importance of FMEA?
FMEA is emphasized in regulatory standards such as ISO 14971, which focuses on the application of risk management to medical devices.
What types of products does Voler Systems support in relation to FMEA?
Voler Systems supports manufacturers in developing various products, including wearable technology, heart pumps, and liquid biopsy platforms.
How effective is FMEA in preventing medical adverse events?
Recent case studies suggest that a significant percentage of medical adverse events can be prevented through effective risk management practices like FMEA, underscoring its importance in safeguarding healthcare outcomes.
Why is it necessary to incorporate FMEA into quality management systems?
Ongoing regulatory updates highlight the necessity of incorporating FMEA into quality management systems to reinforce its role as a fundamental element of medical device safety.
List of Sources
- Define FMEA and Its Importance in Medical Device Development
- 2026 Commences with a Look Back, a Look Forward and New Legislation and Guidance (https://emergobyul.com/news/2026-commences-look-back-look-forward-and-new-legislation-and-guidance)
- FMEA Without Tears: an Expert Perspective (https://aami.org/news/fmea-without-tears-an-expert-perspective)
- Medical devices Archives (https://statnews.com/topic/medical-devices)
- FDA's Risk Management Expectations for Medical Devices: What Manufacturers Need to Know (https://gardner.law/news/fda-risk-management-expectations-medical-devices)
- Healthcare Application of Failure Mode and Effect Analysis (FMEA): Is There Room in the Infectious Disease Setting? A Scoping Review - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11719677)
- Conduct Step-by-Step FMEA Analysis for Your Device
- FMEA team performance in health care: A qualitative analysis of team member perceptions - PubMed (https://pubmed.ncbi.nlm.nih.gov/19920449)
- 2026 Commences with a Look Back, a Look Forward and New Legislation and Guidance (https://emergobyul.com/news/2026-commences-look-back-look-forward-and-new-legislation-and-guidance)
- FMEA for Medical Devices: Design, Process & Use Risk Analysis Explained (https://meddeviceguide.com/blog/fmea-medical-devices-guide)
- How to do FMEA for Medical Devices: Step-by-Step Guide to Risk Management - SoftComply (https://softcomply.com/fmea-medical-devices-risk-management-guide)
- Integrate FMEA Findings into Design and Compliance Strategies
- 2026 Medical Device Manufacturing: Optimism Amid Uncertainty (https://mddionline.com/manufacturing/2026-medical-device-manufacturing-outlook-optimism-grows-despite-lingering-uncertainty)
- 2026 Predictions for Medical Device & Life Sciences: AI, Wearables, and Navigating Regulatory Change (https://jamasoftware.com/blog/2026-predictions-for-medical-device-life-sciences-ai-wearables-and-navigating-regulatory-change)
- FDA's Risk Management Expectations for Medical Devices: What Manufacturers Need to Know (https://gardner.law/news/fda-risk-management-expectations-medical-devices)
- Medical device compliance (https://siemens.com/en-us/digital-thread/integrated-lifecycle-management/medical-device-compliance)
- Address Common Challenges and Solutions in FMEA Implementation
- 2026 Medical Device Manufacturing: Optimism Amid Uncertainty (https://mddionline.com/manufacturing/2026-medical-device-manufacturing-outlook-optimism-grows-despite-lingering-uncertainty)
- Failure Mode and Effects Analysis (FMEA) for Medical Devices (https://vem-medical.com/fmea-for-medical-devices)
- The Challenge of Deploying Failure Modes and Effects Analysis in Complex System Applications—Quantification and Analysis (https://mdpi.com/2071-1050/14/3/1397)
- 2025 Medical Device Regulatory Challenges: Key Trends & Solutions (https://mastercontrol.com/gxp-lifeline/medical-device-regulatory-challenge)
- Assembling the Correct FMEA Team. (https://accendoreliability.com/assembling-correct-fmea-team)





