Introduction
Implementing a robust Failure Mode and Effects Analysis (FMEA) is crucial for ensuring the safety and reliability of medical devices. In this field, even minor oversights can lead to significant consequences. This guide explores the intricacies of FMEA, providing insights into how organizations can systematically identify potential failures and mitigate risks before they affect patient care. Given the increasing regulatory demands and the complexity of modern medical technologies, manufacturers must consider how to remain compliant while proactively safeguarding against potential failures.
Define Failure Mode and Effects Analysis (FMEA)
A system FMEA example illustrates a systematic and organized approach designed to identify potential malfunction types within a system, product, or procedure, and evaluate their impact on overall performance. In the context of medical equipment, FMEA plays a crucial role in anticipating and mitigating risks associated with functionality and safety. The process involves deconstructing the apparatus into its individual components, identifying how each component could fail, and assessing the consequences of these failures. This proactive risk management tool is vital for ensuring that medical equipment not only complies with stringent regulatory standards but also operates reliably in real-world scenarios.
The significance of Failure Mode and Effects Analysis in ensuring medical equipment safety is underscored by alarming statistics; for instance, the theoretical maximum Risk Priority Number (RPN) is 150, indicating the potential severity of issues if left unaddressed. Practical applications of Failure Mode and Effects Analysis have demonstrated its effectiveness, as evidenced by research conducted at Taranaki Base Hospital, which identified 35 error types in a Rapid Response System (RRS) and highlighted the critical need for user feedback in improving patient monitoring procedures. Common errors in manufacturing tests, such as:
- Insufficient test coverage
- Inappropriate test conditions
- Failure to document results
can substantially impact product quality. By utilizing a system FMEA example and addressing these prevalent challenges, organizations can enhance the safety and reliability of their medical equipment, ultimately leading to improved patient outcomes and compliance with industry regulations.
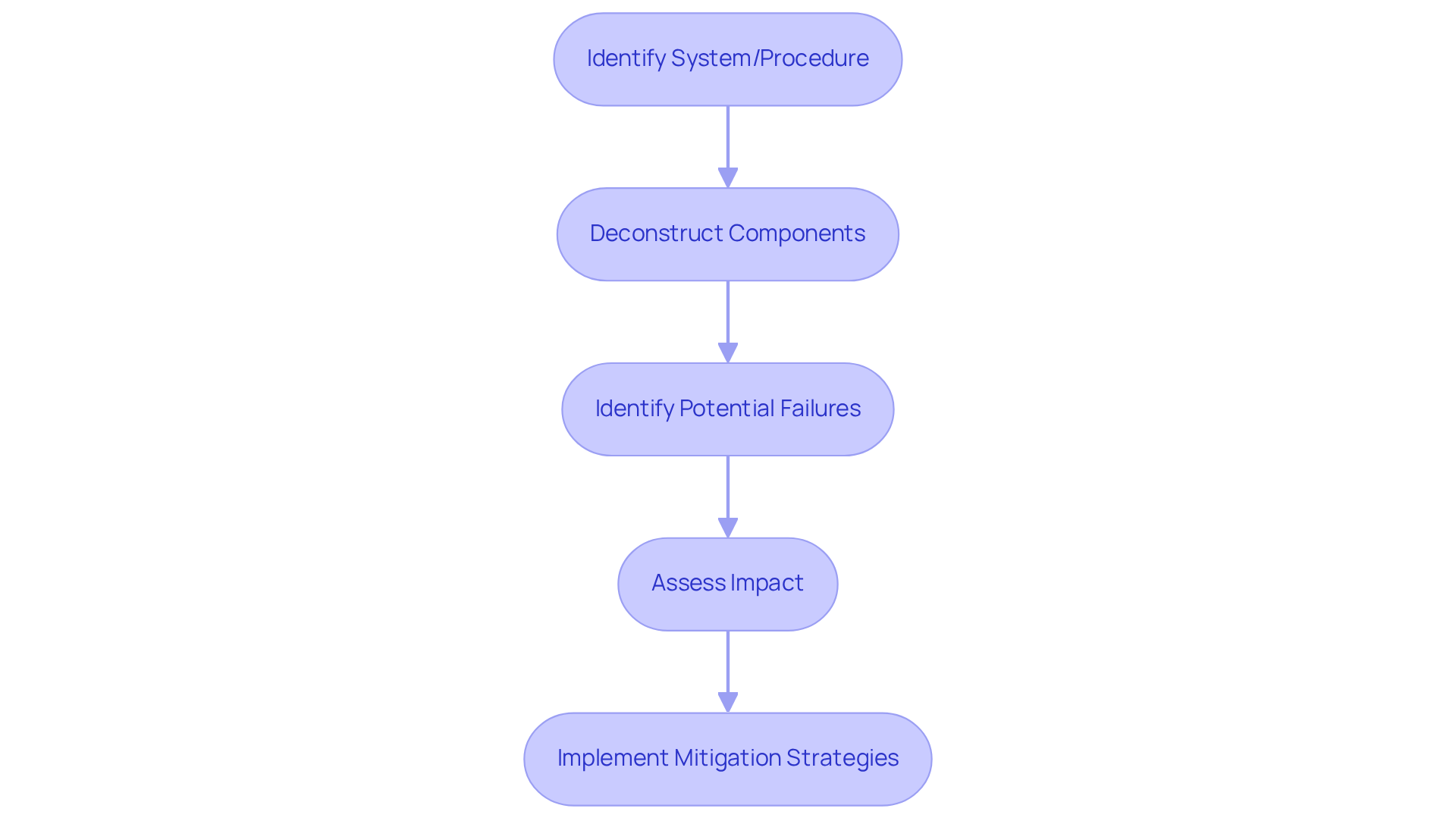
Understand the Importance of FMEA in Medical Device Design
FMEA plays a crucial role in the design of medical equipment for several key reasons. It facilitates the early identification of potential failure modes during the development process, enabling teams to proactively address issues before they escalate into costly recalls or safety incidents. This proactive approach is essential for meeting regulatory requirements, including those established by the FDA and ISO standards, which mandate comprehensive risk evaluations for medical products. By systematically assessing risks, manufacturers such as Voler Systems can significantly enhance product reliability and patient safety, thereby fostering improved market acceptance and customer trust.
Furthermore, FMEA promotes a culture of continuous improvement within organizations. It encourages teams to learn from past mistakes and refine their designs, ultimately leading to the creation of more durable and safer medical products. For instance, Voler Systems has successfully developed an innovative calf-worn tool for motion and circumference monitoring, which is vital for knee replacement rehabilitation. This device not only meets stringent medical equipment standards but also exemplifies how failure mode and effects analysis can guide the development phase to ensure compliance and safety.
Case studies have shown that organizations effectively implementing Failure Mode and Effects Analysis have reduced the incidence of recalls by as much as 30%, thereby safeguarding their reputation and financial stability. As regulatory scrutiny intensifies, integrating failure modes and effects analysis into the design process becomes essential for maintaining compliance and securing the long-term success of medical products in the marketplace. Regulatory experts emphasize that failure mode and effects analysis is not merely a compliance tool but a strategic approach that can significantly impact a company's market success.
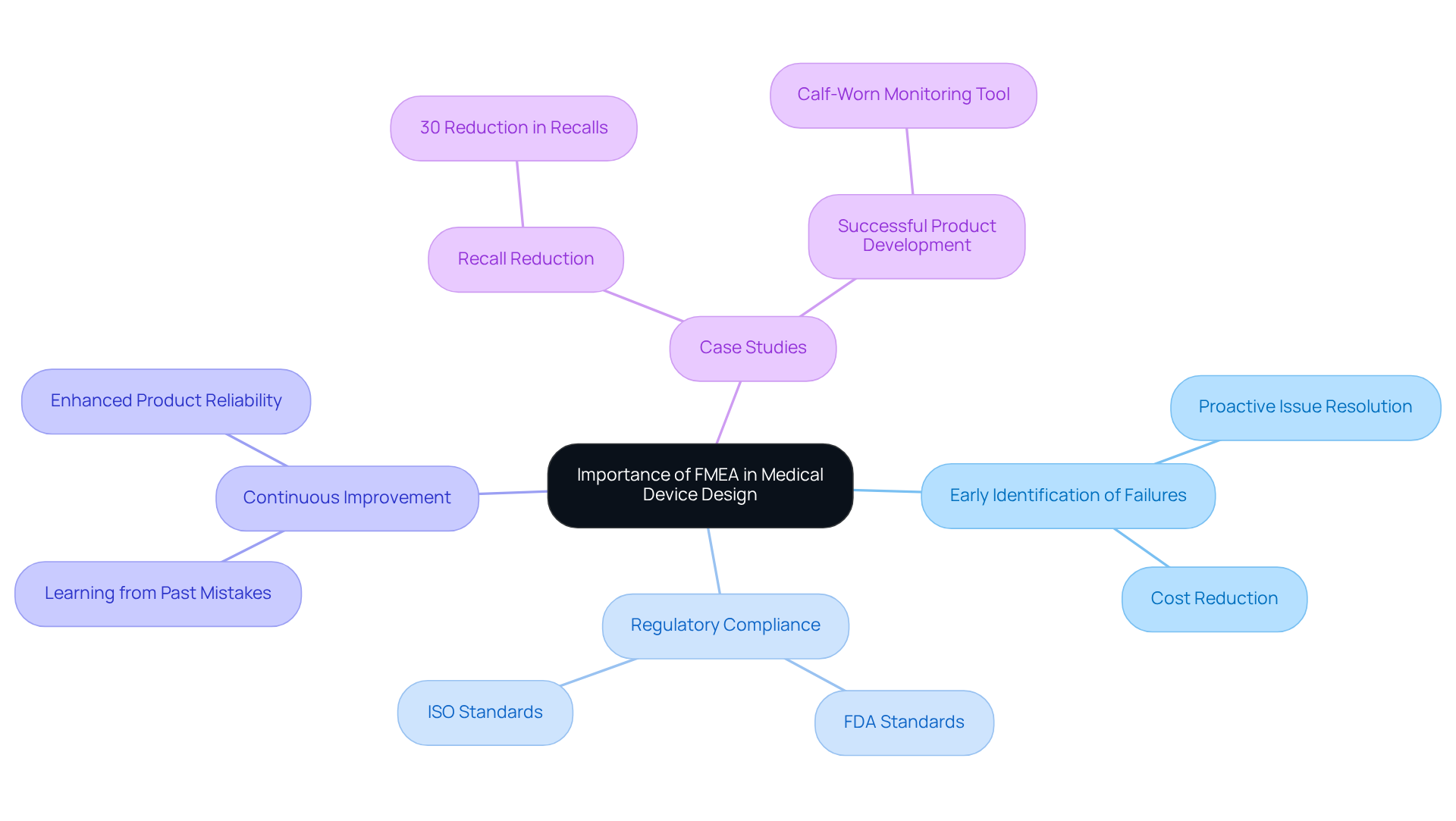
Implement FMEA: Step-by-Step Process for Medical Device Design
- Define the Scope: Clearly outline the system, subsystem, or component to be analyzed, specifying the boundaries of the analysis and the intended application of the apparatus. This essential step ensures that the system FMEA example for the failure mode and effects analysis process is targeted and relevant, particularly concerning the compliance assistance provided by Voler Systems for medical product startups.
- Assemble the FMEA Team: Gather a cross-functional team that includes design engineers, quality assurance professionals, and regulatory experts. Diverse perspectives enhance the analysis, ensuring comprehensive coverage of potential issues. Voler Systems emphasizes collaboration in this phase to streamline hardware development and ensure all regulatory requirements are met.
- Identify Functions: List the functions of the apparatus or system components. Understanding the intended functions is crucial for recognizing possible malfunction modes and ensuring that all aspects of the device are considered.
- Identify Malfunction Types: For each function, brainstorm potential malfunction modes. Consider how each component could fail to perform its intended function, which is vital for a thorough risk assessment.
- Assess Effects of Issues: Evaluate the consequences of each issue mode. Determine how these shortcomings could affect the device's performance, safety, and adherence to regulatory standards, which is essential in the medical device sector.
- Assign Severity Ratings: Rate the severity of each impact effect on a scale (e.g., 1 to 10), where 1 is negligible and 10 is catastrophic. This prioritization helps focus efforts on the most critical issues, as illustrated in a system FMEA example, that could impact patient safety.
- Identify Causes: For each malfunction mode, identify potential causes. Examine design flaws, manufacturing defects, or user errors that could lead to issues, ensuring a comprehensive understanding of risks. Voler Systems' prototype strategy aids in identifying these risks early in the design process.
- Assign Occurrence Ratings: Rate the likelihood of each cause occurring on a similar scale. This evaluation aids in understanding which setbacks are more likely and require proactive management.
- Determine Detection Ratings: Evaluate how easily each issue can be detected before it reaches the customer. A lower rating indicates a greater possibility of issues going unnoticed, highlighting areas for improvement in quality control.
- Calculate Risk Priority Number (RPN): Multiply the severity, occurrence, and detection ratings for each error mode to obtain the RPN. This number helps prioritize which issues to address first, guiding resource allocation effectively.
- Develop Action Plans: For high-priority failure modes (those with high RPNs), create action plans to mitigate risks. This may involve design changes, additional testing, or process improvements to enhance product reliability, serving as a system FMEA example that aligns with Voler Systems' commitment to supporting startups in navigating compliance from prototype to production.
- Document and Review: Maintain a comprehensive failure mode and effects analysis report that records findings, action plans, and follow-up activities. Consistently assess and revise the failure mode and effects analysis as the design progresses to ensure continuous compliance and safety. The FDA's revised guidance stresses the necessity for manufacturers to incorporate risk management across the entire product lifecycle, underscoring the importance of a robust failure modes and effects analysis.
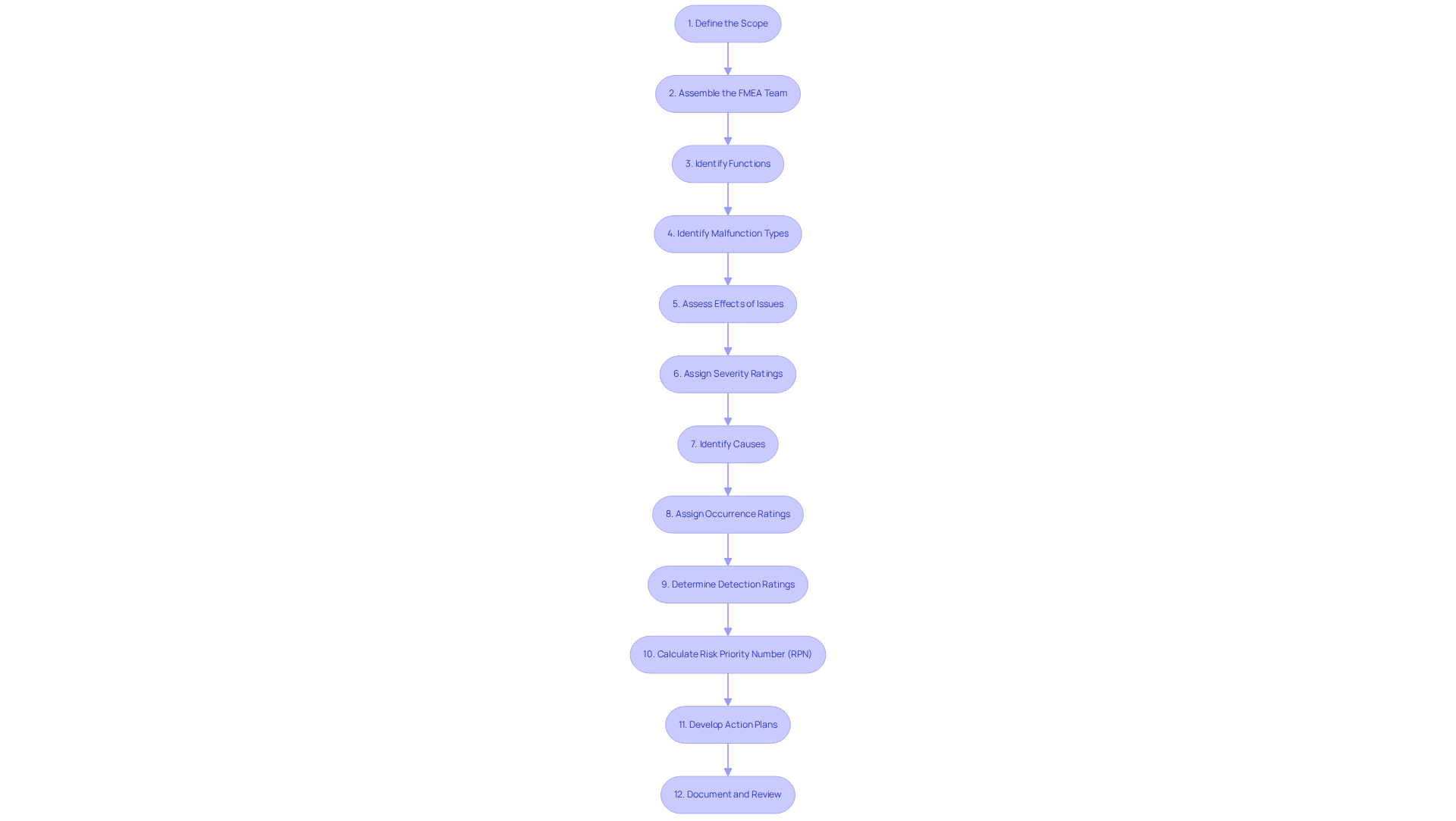
To effectively implement FMEA in medical device design, consider utilizing the following tools and resources:
- FMEA Software: Tools such as Relyence, RiskWatch, and FMEA-Pro streamline the analysis process by automating documentation and fostering collaboration among team members. This not only enhances efficiency but also improves precision. Industry experts indicate that these software solutions can significantly reduce the time allocated to documentation, enabling teams to concentrate on critical analysis.
- Templates and Checklists: Standardized failure mode and effects analysis templates ensure consistency in evaluation, while checklists help confirm adherence to all essential steps, thereby minimizing the risk of oversight during the procedure. Leveraging templates from successful case studies, such as those offered by Minitab Engage, can further elevate the quality of your analysis.
- Training Resources: Investing in training programs or workshops focused on failure mode and effects analysis methodologies can substantially enhance team capabilities. Online courses and webinars provide valuable insights and practical examples that can be directly applied to projects. For instance, a recent webinar highlighted that effective training can lead to a 30% reduction in errors during the failure mode and effects analysis process.
- Industry Guidelines: Consulting guidelines from organizations like the FDA and ISO is essential for adhering to best practices in executing failure mode and effects analysis. These documents offer critical frameworks and standards that can enhance the quality of your analysis. The FDA underscores that compliance with these guidelines can avert common pitfalls that may result in product recalls.
- Case Studies: Examining successful failure mode and effects analysis case studies in medical device development allows teams to draw insights from practical experiences. For example, a case study involving Relyence demonstrated that a systematic approach to failure mode and effects analysis led to a 40% decrease in design defects, underscoring the tangible benefits of thorough examination.
- Collaboration Tools: Employing project management and collaboration tools such as Jira or Trello improves communication among team members throughout the FMEA process. This ensures alignment and keeps everyone informed, fostering a more cohesive working environment. Effective collaboration has been shown to enhance project outcomes by as much as 25%, according to recent industry reports.
Conclusion
Implementing Failure Mode and Effects Analysis (FMEA) is crucial for ensuring the safety and reliability of medical devices. This systematic approach identifies potential failure modes and evaluates their impact on overall performance, fostering a culture of proactive risk management. By understanding and applying FMEA principles, organizations can enhance compliance with regulatory standards and improve patient outcomes.
The article outlines the critical steps for executing FMEA effectively, including:
- Defining the scope
- Assembling a cross-functional team
- Assessing risks
- Developing action plans
Key insights emphasize the importance of early identification of potential failure modes, the use of specialized tools and resources, and the role of continuous improvement in the design process. Case studies illustrate that companies utilizing FMEA have successfully reduced recalls and improved product reliability, underscoring its value in the medical device industry.
In conclusion, integrating FMEA into the design and development of medical devices is not merely a regulatory requirement but a strategic necessity that leads to safer products and improved patient care. By embracing this structured methodology, organizations can mitigate risks, enhance their market reputation, and contribute to the advancement of healthcare technology. It is imperative for medical device manufacturers to prioritize FMEA as a cornerstone of their design processes, ensuring that safety and reliability remain at the forefront of their innovations.
Frequently Asked Questions
What is Failure Mode and Effects Analysis (FMEA)?
FMEA is a systematic and organized approach used to identify potential malfunction types within a system, product, or procedure, and evaluate their impact on overall performance, particularly in the context of medical equipment.
How does FMEA benefit medical equipment safety?
FMEA helps anticipate and mitigate risks associated with the functionality and safety of medical equipment, ensuring compliance with regulatory standards and reliable operation in real-world scenarios.
What is the process involved in conducting an FMEA?
The process involves deconstructing the apparatus into its individual components, identifying how each component could fail, and assessing the consequences of these failures.
What is the significance of the Risk Priority Number (RPN) in FMEA?
The theoretical maximum RPN is 150, indicating the potential severity of issues if failures are left unaddressed, highlighting the importance of FMEA in risk management.
Can you provide an example of FMEA application in a medical context?
Research conducted at Taranaki Base Hospital identified 35 error types in a Rapid Response System (RRS), demonstrating the effectiveness of FMEA and the critical need for user feedback to improve patient monitoring procedures.
What are some common errors identified in manufacturing tests?
Common errors include insufficient test coverage, inappropriate test conditions, and failure to document results, which can significantly impact product quality.
How can organizations improve the safety and reliability of medical equipment using FMEA?
By utilizing FMEA to address prevalent challenges in manufacturing and testing, organizations can enhance the safety and reliability of their medical equipment, leading to improved patient outcomes and compliance with industry regulations.
List of Sources
- Define Failure Mode and Effects Analysis (FMEA)
- Failure mode and effect analysis (FMEA) to identify and mitigate failures in a hospital rapid response system (RRS) - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC8857483)
- linkedin.com (https://linkedin.com/pulse/failure-mode-effects-analysis-fmea-software-market-dsaxf)
- Patient Safety Risks of AI in the OR Exposed & Other Medtech News (https://mddionline.com/artificial-intelligence/frightening-medical-ai-problems-exposed-other-medtech-news)
- Understand the Importance of FMEA in Medical Device Design
- CDRH New (https://fda.gov/medical-devices/medical-devices-news-and-events/cdrh-new-news-and-updates)
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- FDA Medical Device Safety Action Plan: Good or Bad for Medtech? - Medtec China (https://en.medtecchina.com/media/policies-regulations/fda-medical-device-safety-action-plan-good-or-bad-for-medtech)
- Medical Device Regulatory News and Updates (https://pureglobal.com/resources/regulatory-updates)
- Medical Device Industry News & Regulatory Updates: What Changed in 2026 and What's Coming Next (https://meddeviceguide.com/blog/medical-device-industry-news-regulatory-updates-2026)
- Implement FMEA: Step-by-Step Process for Medical Device Design
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- Identify Tools and Resources for Effective FMEA Implementation
- clarkstonconsulting.com (https://clarkstonconsulting.com/insights/2026-medical-device-industry-trends)
- The Hidden Causes Behind Device Recalls (https://spectrum.ieee.org/medical-device-recalls)
- FDA Updates Broaden Wellness and Clinical Decision Support Software Devices Under Enforcement (https://emergobyul.com/news/fda-updates-broaden-wellness-and-clinical-decision-support-software-devices-under-enforcement)
- visuresolutions.com (https://visuresolutions.com/medtech-and-pharma-guide/best-risk-management-fmea-tools)





