Master FMEA Failure Mode: A Step-by-Step Guide for Medical Devices
Master FMEA failure modes in medical devices for enhanced safety and reliability.

Failure Mode and Effects Analysis (FMEA) is a fundamental aspect of engineering, especially in sectors where safety and reliability are paramount. This systematic methodology enables teams to identify potential failure modes in products and processes, allowing for proactive measures that enhance performance and reduce risks. As the complexity of contemporary engineering challenges increases, organizations must consider how to effectively implement FMEA. This implementation should not only meet regulatory standards but also promote innovation and build trust in their products.
The term refers to the FMEA full form in engineering, which stands for Failure Mode and Effects Analysis, a systematic approach used to identify potential defect modes within a product or process and assess their impact on performance. Developed in the 1940s by the U.S. military, FMEA aimed to enhance reliability in military systems. The core principle revolves around predicting issues before they occur, allowing teams to implement corrective measures proactively. By analyzing how components may fail and the consequences of those failures, engineers can prioritize risks and bolster reliability, particularly in critical sectors such as medical devices and aerospace.
In the realm of medical equipment manufacturing, the importance of robust testing systems is paramount. These systems are essential for ensuring that products comply with standards from prototype through to production, especially in the area of wearable medical devices. Furthermore, comprehending and addressing common errors in manufacturing tests is crucial for sustaining quality and efficiency in electronic device design. By integrating the FMEA full form in engineering with effective testing strategies, manufacturers can significantly reduce the risk of issues and enhance the overall reliability of their products.
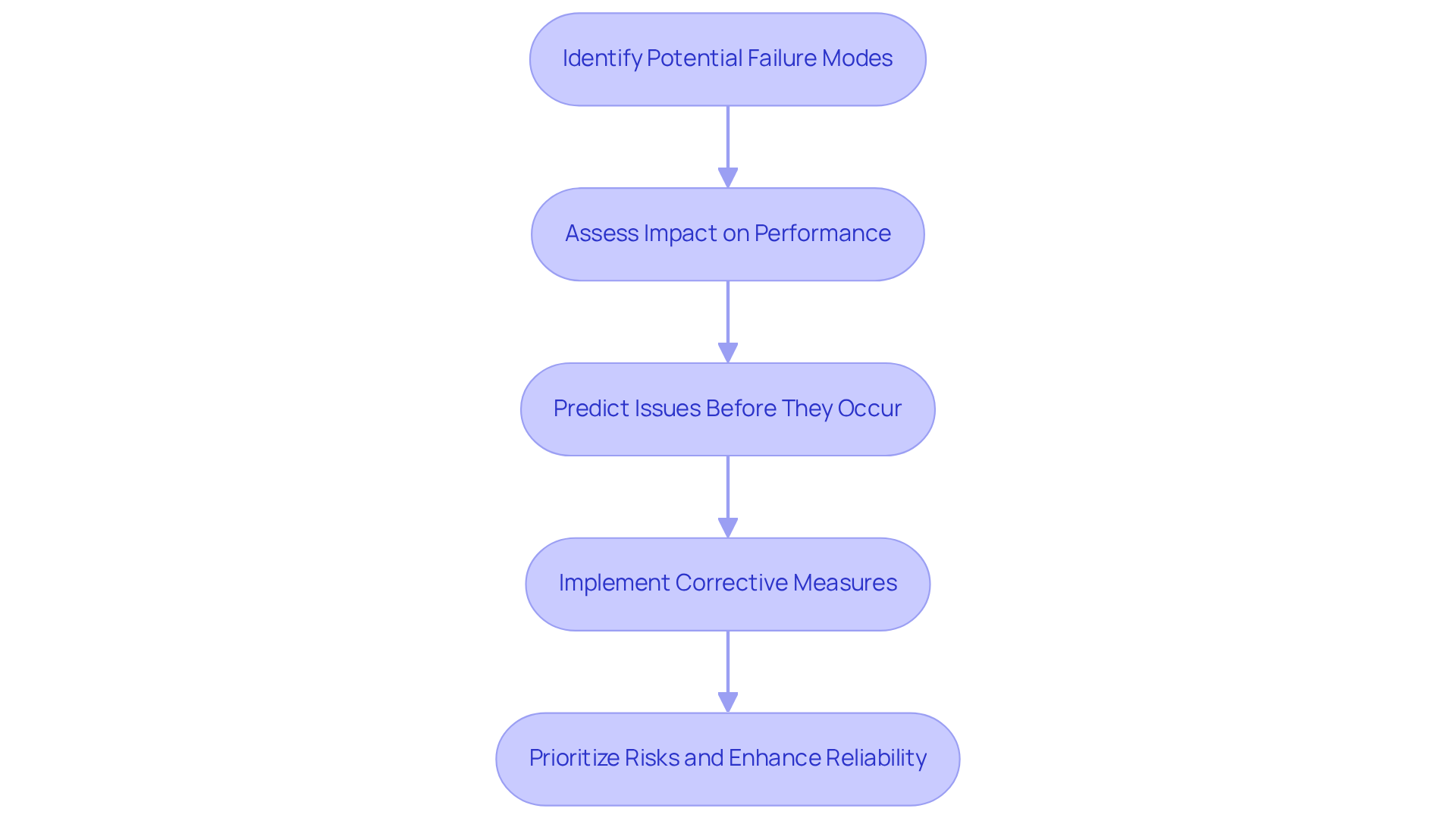
The FMEA full form in engineering is Failure Mode and Effects Analysis, which is a critical component that significantly enhances product reliability and safety by identifying potential malfunction modes early in the design phase. This proactive approach enables teams to tackle issues before they develop into costly problems or safety risks. In the medical device, FMEA is essential for meeting stringent regulatory standards, as it allows manufacturers to conduct comprehensive evaluations of potential risks. By prioritizing risk modes based on their severity and likelihood, organizations can allocate resources effectively, ensuring that the most critical threats are addressed first. This not only improves quality but also fosters customer trust and satisfaction.
For instance, a medical device manufacturer utilized FMEA during the design phase of a wearable health monitor, identifying battery failure types early on. This proactive identification led to design modifications that enhanced both safety and performance. Voler Systems exemplifies expertise in supporting legacy test equipment for medical device firms, ensuring compliance and reliability in their products. Their innovative development of a calf-worn tool for monitoring motion and circumference in knee replacement rehabilitation further demonstrates how FMEA principles can improve patient safety and product effectiveness. Customer feedback has underscored the success of these applications, highlighting their significant impact on product reliability and patient safety in the healthcare sector.
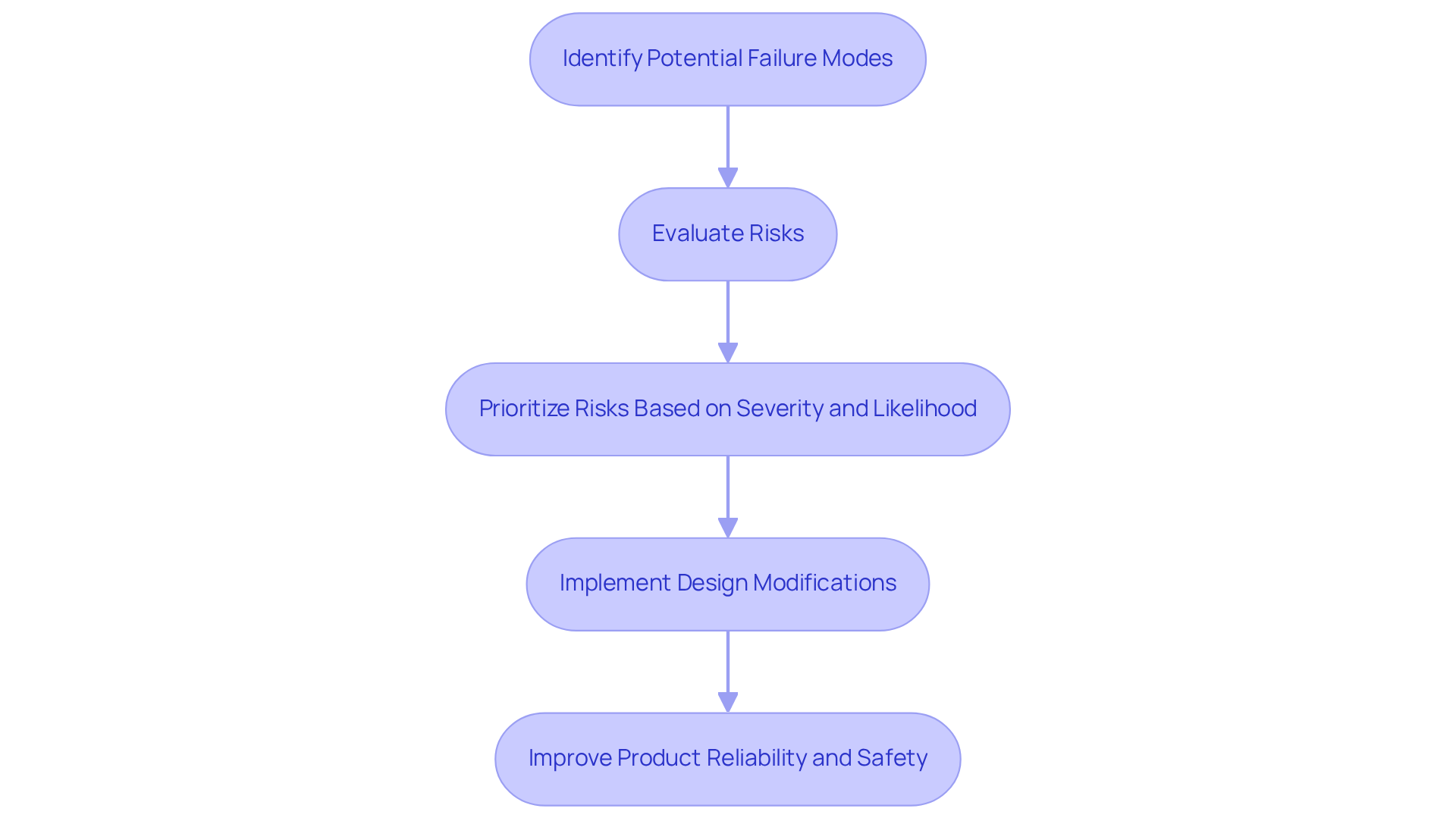
The FMEA full form in engineering refers to Failure Mode and Effects Analysis, which is a critical tool in the development of medical equipment, systematically identifying potential modes of malfunction and their implications for patient safety. Its application is particularly vital for devices such as pacemakers and insulin pumps, where any failure could lead to severe health consequences. By employing the FMEA full form in engineering, manufacturers can proactively mitigate risks, ensuring compliance with stringent regulatory standards and enhancing product reliability.
Recent case studies illustrate the effectiveness of FMEA in this sector. For example, a medical equipment manufacturer utilized FMEA during the design phase of a wearable health monitor, successfully identifying a potential battery failure mode early in the development process. This proactive approach led to design modifications that significantly improved the safety and performance of the device, highlighting the tangible benefits of the FMEA full form in engineering.
Statistics further highlight the importance of FMEA in medical device innovation. The global FMEA market is projected to grow at a compound annual growth rate (CAGR) of approximately 9.2% over the forecast period, reflecting the increasing complexity of manufacturing processes and the need for robust quality assurance practices. Additionally, the FMEA software market is expected to reach around USD 22.18 billion by 2031, driven by technological advancements and regulatory demands.
Expert insights stress the necessity of treating FMEA documentation, the FMEA full form in engineering, as dynamic and evolving to enhance risk management practices. Regular updates and continuous improvement of risk assessment documents ensure their relevance throughout the product lifecycle, ultimately leading to safer and more reliable medical equipment. By integrating FMEA into their quality management processes, manufacturers can prioritize risks based on severity and likelihood, focusing on the most critical issues and fostering a culture of continuous improvement. Furthermore, Voler Systems' innovative solutions in incorporating edge AI into medical equipment are transforming modern healthcare, equipping manufacturers to address the challenges of today's medical landscape. Voler Systems also specializes in developing a range of medical devices, including wearable technologies, designed with risk assessment principles to enhance safety and performance.
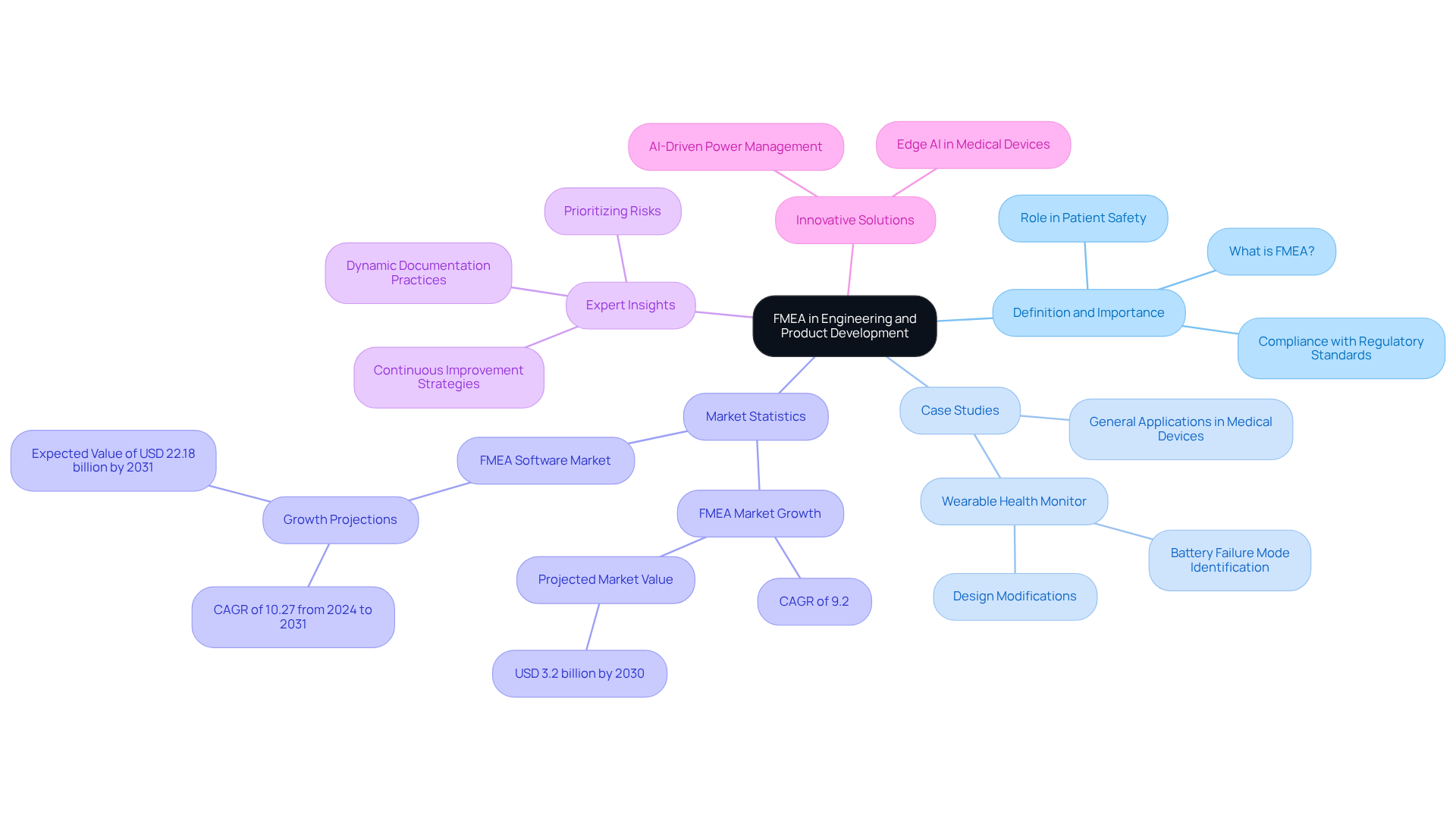
The FMEA process is a systematic approach that encompasses several critical steps to ensure thorough analysis and risk mitigation:
By adhering to these steps, organizations can effectively identify and address potential failures, significantly enhancing product reliability and safety. This structured approach not only fosters a culture of quality but also aligns with industry standards, ensuring compliance with best practices in engineering, such as understanding the FMEA full form in engineering.
Failure Mode and Effects Analysis (FMEA) is not merely a methodology; it is an essential strategy that significantly enhances engineering practices by identifying potential failures before they manifest. By systematically analyzing how components can fail and the implications of those failures, FMEA empowers engineers to implement proactive solutions, thereby ensuring product reliability and safety across various industries, particularly in critical sectors such as healthcare and aerospace.
The article underscores key insights regarding the historical development of FMEA, its importance in regulatory compliance, and its application in real-world scenarios, such as the design of medical devices. The structured process of FMEA, which encompasses steps from defining the scope to continuous monitoring and updating, serves as a comprehensive blueprint for effectively mitigating risks. Case studies, including those involving wearable health monitors, illustrate the tangible benefits of employing FMEA, demonstrating how early identification of potential issues can lead to enhanced safety and performance.
Ultimately, the integration of FMEA into engineering practices cultivates a culture of quality and continuous improvement. As the complexity of manufacturing processes escalates, the demand for robust risk management strategies becomes increasingly critical. Embracing FMEA not only enhances product reliability but also fosters trust with customers and stakeholders. Organizations are encouraged to adopt this systematic approach to safeguard their innovations and ensure that they meet the highest standards of safety and effectiveness in an ever-competitive landscape.
What does FMEA stand for in engineering?
FMEA stands for Failure Mode and Effects Analysis.
What is the core concept of FMEA?
The core concept of FMEA is to systematically identify potential defect modes within a product or process and assess their impact on performance, allowing teams to predict issues before they occur and implement corrective measures proactively.
When was FMEA developed and by whom?
FMEA was developed in the 1940s by the U.S. military to enhance reliability in military systems.
How does FMEA benefit engineering, particularly in critical sectors?
FMEA helps engineers prioritize risks and bolster reliability by analyzing how components may fail and the consequences of those failures, which is particularly important in critical sectors such as medical devices and aerospace.
Why are robust testing systems important in medical equipment manufacturing?
Robust testing systems are essential for ensuring that products comply with standards from prototype through to production, particularly in the area of wearable medical devices.
What role does FMEA play in manufacturing tests?
FMEA helps in comprehending and addressing common errors in manufacturing tests, which is crucial for sustaining quality and efficiency in electronic device design.
How can manufacturers enhance the reliability of their products using FMEA?
By integrating FMEA with effective testing strategies, manufacturers can significantly reduce the risk of issues and enhance the overall reliability of their products.
