Understanding Product Development Technologies for Medical Devices
Explore the significance and evolution of product development technologies in medical...

Selecting the appropriate embedded programming services for medical devices is a crucial decision that can profoundly influence patient safety and regulatory compliance. Given the healthcare industry's stringent standards and the rapid evolution of technologies, it is essential for manufacturers to grasp the intricacies of embedded programming to deliver reliable and innovative products. However, how can one ascertain that the chosen service provider not only fulfills technical specifications but also navigates the complex landscape of healthcare regulations? This article explores best practices for assessing embedded programming services, guiding readers through key criteria that foster effective partnerships and enhance project success.
Choosing embedded programming services for healthcare instruments requires a thorough understanding of industry-specific requirements, especially the . is critical, as it outlines the lifecycle processes for healthcare software, ensuring safety, efficacy, and performance. Recent data reveals that a significant percentage of healthcare instruments fail to meet these regulatory standards, underscoring the necessity for strict adherence to compliance protocols.
For instance, when developing a wearable health monitor, the embedded programming must enable , all while adhering to . , including regulatory bodies and end-users, yields valuable insights into the essential features and compliance requirements. This collaborative approach ensures that the selected embedded programming services are tailored to meet these critical needs, ultimately enhancing the reliability and market readiness of healthcare equipment.
, with its extensive experience in developing a range of , heart pumps, and liquid biopsy platforms, is well-positioned to assist manufacturers in . Their AI-assisted engineering methodology not only expedites the development process but also guarantees that products are designed for the future of intelligent healthcare.
To ensure the success of your health equipment project, it is essential to evaluate the technical skills and of potential firms offering . Consider suppliers like , which have a proven track record in developing innovative that leverage FPGA and AI technologies, particularly for wireless health equipment designed for optimal battery life. Their compliance with industry standards, such as and ISO 14971, is vital for guaranteeing the safety and efficacy of .
Request documentation of their previous work, certifications, and compliance records to thoroughly assess their qualifications. Additionally, consider their experience with technologies pertinent to your equipment, such as or protocols. Engaging in discussions about their development processes and will yield valuable insights into their capabilities and commitment to maintaining .
Be mindful of common pitfalls in the evaluation process, such as neglecting the significance of ongoing compliance and the necessity for comprehensive documentation. By following these recommended practices, you can enhance the likelihood of selecting a supplier that will positively influence the success of your endeavor.
Effective collaboration and communication with providers are crucial for the success of . Establishing from the outset ensures that all stakeholders are aligned on objectives, timelines, and . Regular meetings and updates foster transparency, enabling the that could impact delivery.
Utilizing facilitates real-time communication and document sharing, which is essential in a field where precision and safety are paramount. Constructive feedback from both technical and non-technical stakeholders enhances the understanding of the initiative's needs, leading to improved outcomes. Successful case studies in demonstrate that significantly enhance project success rates, reducing the likelihood of costly delays and ensuring compliance with stringent industry standards.
This collaborative approach not only improves the quality of the final product but also strengthens the partnership with your service provider, ultimately contributing to the advancement of .
When selecting , it is crucial to review and . These resources provide valuable insights into the expert's experience, capabilities, and the quality of their work. Seek out that showcase similar to your own, particularly those emphasizing adherence to and innovative solutions.
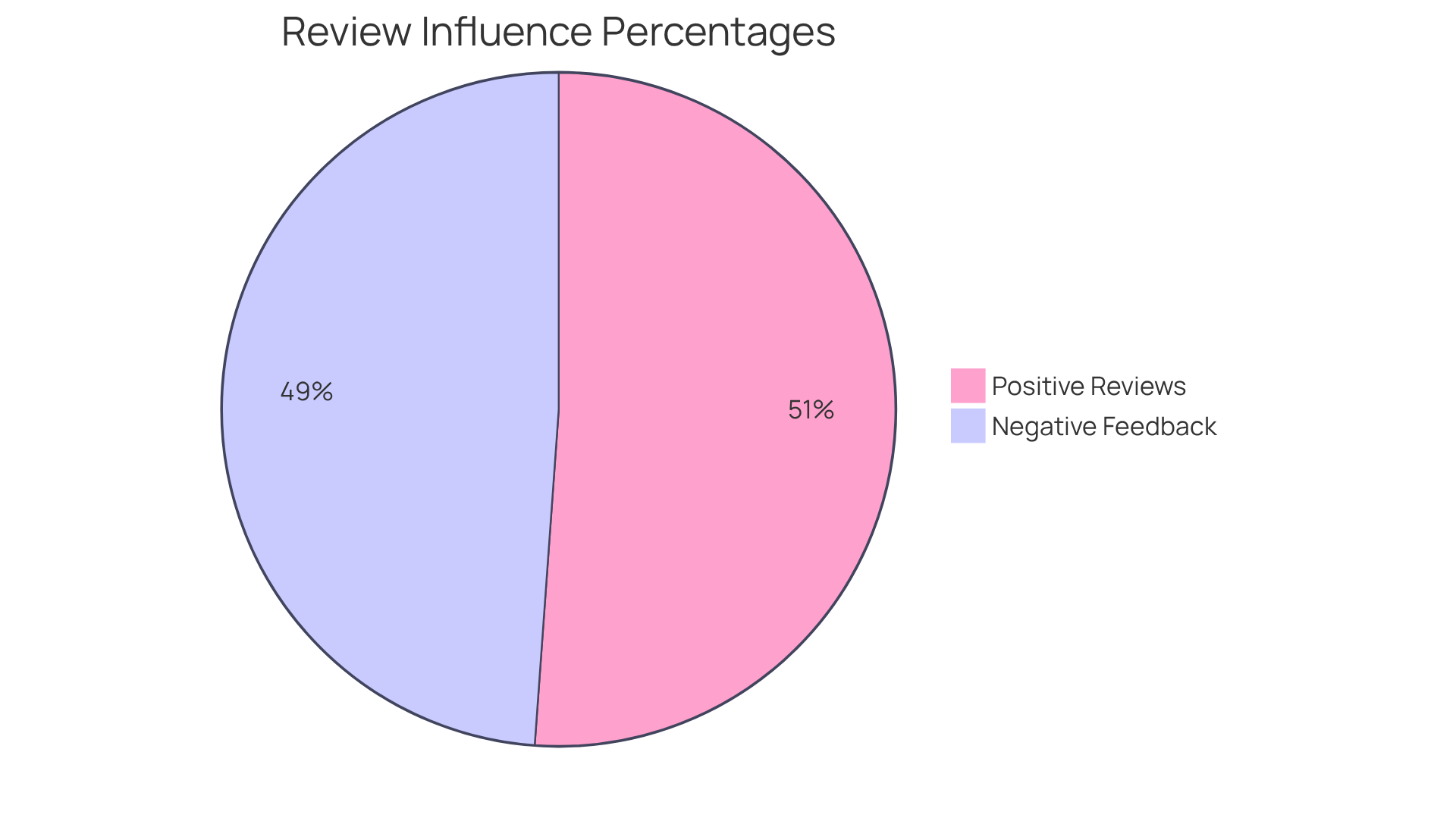
present firsthand accounts of a professional's reliability, communication, and ability to meet deadlines. Notably, 90% of review readers are influenced by , while 86% are affected by negative feedback. This highlights the importance of understanding . Engaging with former clients for can further clarify their satisfaction levels and the organization's alignment with your project’s goals and values. Conducting thorough due diligence ensures that you select a provider of who is not only technically proficient but also dedicated to delivering .
Choosing the right embedded programming services for medical devices is a critical decision that significantly impacts the safety, efficacy, and market readiness of healthcare products. Compliance with industry regulations, such as IEC 62304, is paramount, as it ensures that the development process adheres to the highest standards, ultimately safeguarding patient safety and enhancing product reliability.
This article outlines essential best practices for selecting embedded programming providers. Key considerations include:
By understanding regulatory standards and engaging with experienced providers like Voler Systems, stakeholders can make informed choices that align with their project goals. Furthermore, clear communication and collaboration with service providers can lead to more successful outcomes, as demonstrated by numerous case studies that highlight the benefits of effective partnerships in healthcare technology development.
Ultimately, selecting embedded programming services should not be taken lightly. It is vital to prioritize compliance, technical proficiency, and collaborative approaches to ensure the successful development of innovative medical devices. By adhering to these best practices, healthcare manufacturers can navigate the complexities of embedded programming, drive advancements in intelligent healthcare, and contribute to improved patient outcomes.
What are the key regulatory standards for embedded programming in healthcare?
Compliance with IEC 62304 is critical, as it outlines the lifecycle processes for healthcare software, ensuring safety, efficacy, and performance.
Why is compliance with regulatory standards important in healthcare instruments?
A significant percentage of healthcare instruments fail to meet regulatory standards, highlighting the necessity for strict adherence to compliance protocols to ensure safety and effectiveness.
What specific requirements must be considered when developing wearable health monitors?
Embedded programming for wearable health monitors must enable real-time data processing and secure data transmission while adhering to patient privacy regulations such as HIPAA.
How can collaboration with stakeholders improve the development of healthcare instruments?
Collaborating with stakeholders, including regulatory bodies and end-users, provides valuable insights into essential features and compliance requirements, ensuring that embedded programming services are effectively tailored to meet critical needs.
What experience does Voler Systems have in the healthcare sector?
Voler Systems has extensive experience in developing a range of healthcare products, including wearable technology, heart pumps, and liquid biopsy platforms.
How does Voler Systems approach the development process for healthcare products?
Voler Systems employs an AI-assisted engineering methodology that expedites the development process while ensuring that products are designed for the future of intelligent healthcare.
