4 Best Practices to Manage Product Risk in Medical Device Development
Learn best practices for managing product risk in medical device development effectively.

In the realm of medical equipment development, ensuring patient safety and device efficacy is paramount. This article examines four best practices that not only ensure compliance with stringent regulatory standards but also prioritize user needs, ultimately enhancing the effectiveness of medical devices. However, as the landscape of regulations and user expectations evolves, how can developers maintain compliance while fostering innovation?
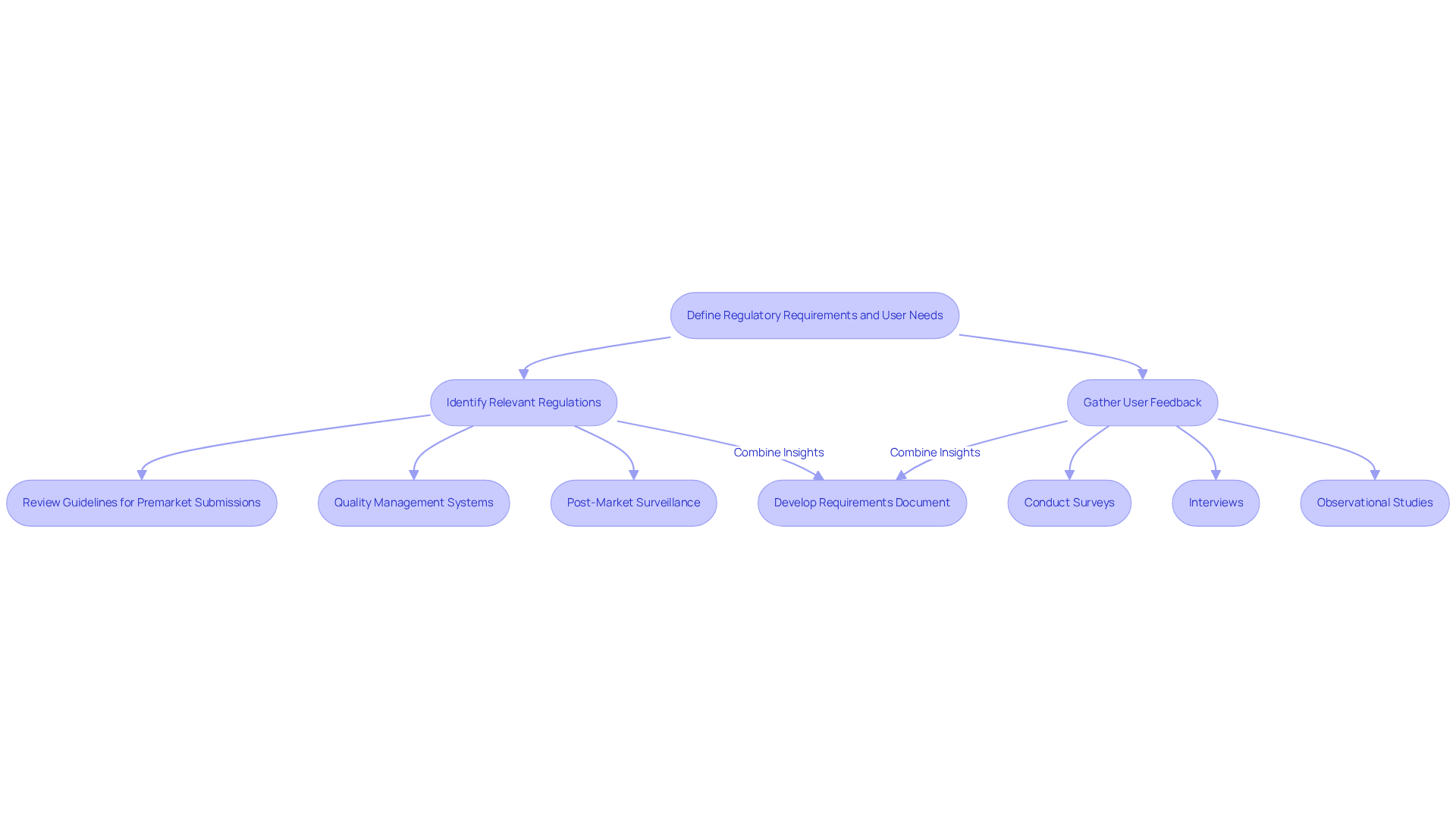
In the medical equipment development sector, navigating compliance requirements is a critical undertaking. Start by identifying the relevant regulations from bodies such as the FDA or EMA, which govern the safety and efficacy of medical devices. This requires a review of guidelines for premarket submissions and quality management systems. Additionally, it includes post-market surveillance. Recent proposals published on December 16, 2025, aim to streamline these processes, enhancing predictability while maintaining patient safety. Significantly, the feedback period for these proposals extends from January 7, 2026, to March 5, 2026, allowing stakeholders to contribute their insights.
At the same time, interact with prospective participants through surveys, interviews, and observational studies to collect insights into their needs and challenges. This dual strategy in medical equipment development ensures that the device meets compliance standards while also addressing user needs. For example, a study published in the Journal of Medical Devices emphasizes that devices created with feedback from individuals significantly lower the chance of usability mistakes, improving patient safety and satisfaction.
To apply this practice successfully, develop a requirements document that details both compliance standards and client needs related to medical equipment development. This document should be routinely revised to incorporate new regulations and feedback, ensuring that the development process stays aligned with both compliance and stakeholder expectations. Additionally, the introduction of regulatory sandboxes will support evidence generation for innovative technologies, allowing for more flexible compliance pathways. Ultimately, aligning compliance with user feedback can significantly enhance device effectiveness and patient safety.
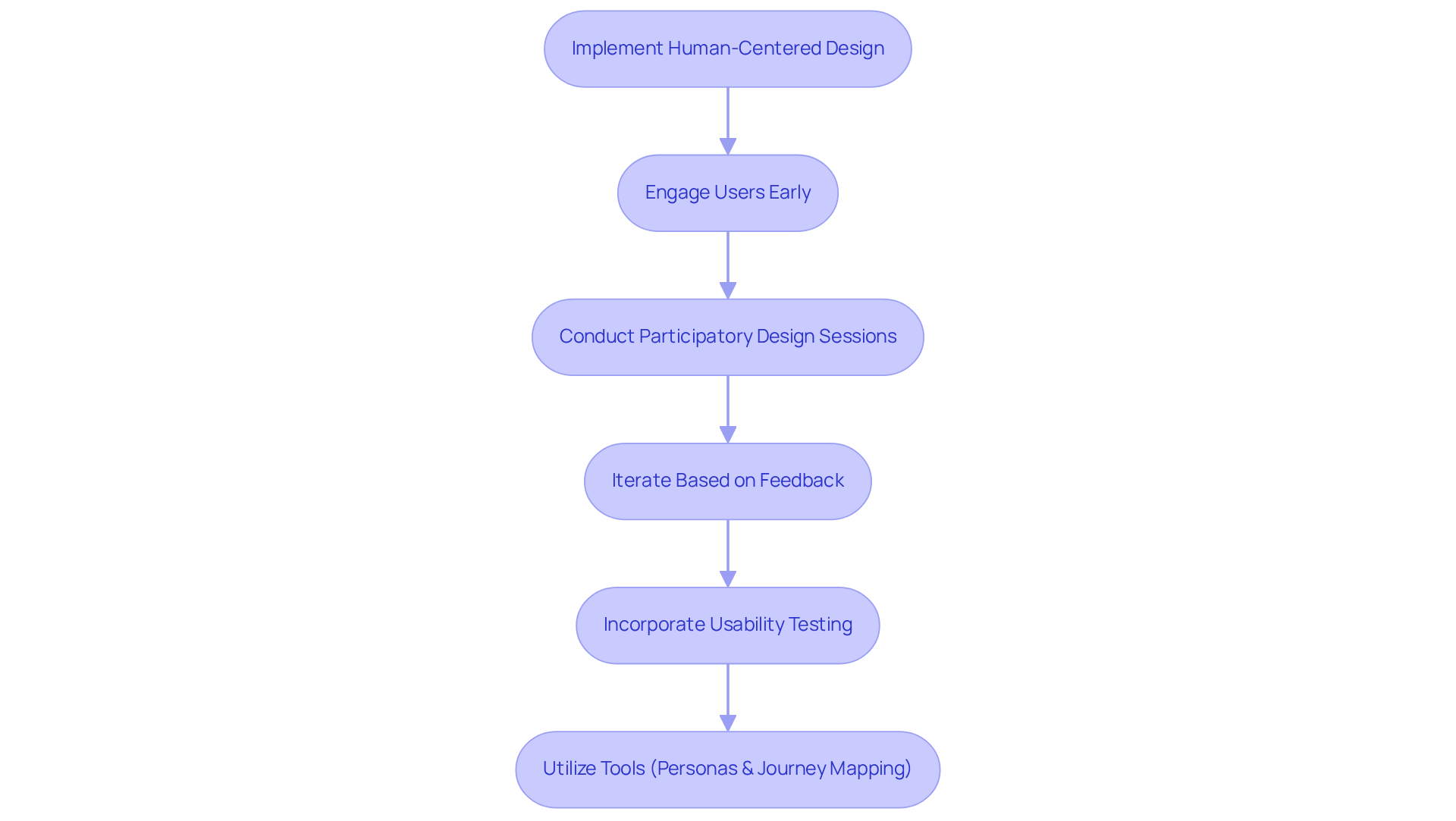
Human-centered design (HCD) is essential for medical equipment development that prioritizes user experience. At Voler Systems, we engage users early in the design process through participatory design sessions, allowing for valuable feedback on prototypes. This iterative method allows for modifications based on real-world usage scenarios, effectively enhancing functionality and usability.
Incorporating usability testing at various stages of development is vital for identifying potential issues before product launch. For example, research shows that products created with HCD principles can result in a 30% decrease in participant errors during clinical trials, demonstrating the method's effectiveness and Voler Systems' commitment to optimizing medical equipment development and ensuring compliance.
To implement HCD effectively, tools such as personas and journey mapping should be utilized to visualize interactions with the system. This practice enhances usability and deepens our understanding of user needs, leading to more successful product outcomes. By prioritizing HCD, Voler Systems not only enhances product usability but also fosters trust in medical technology.
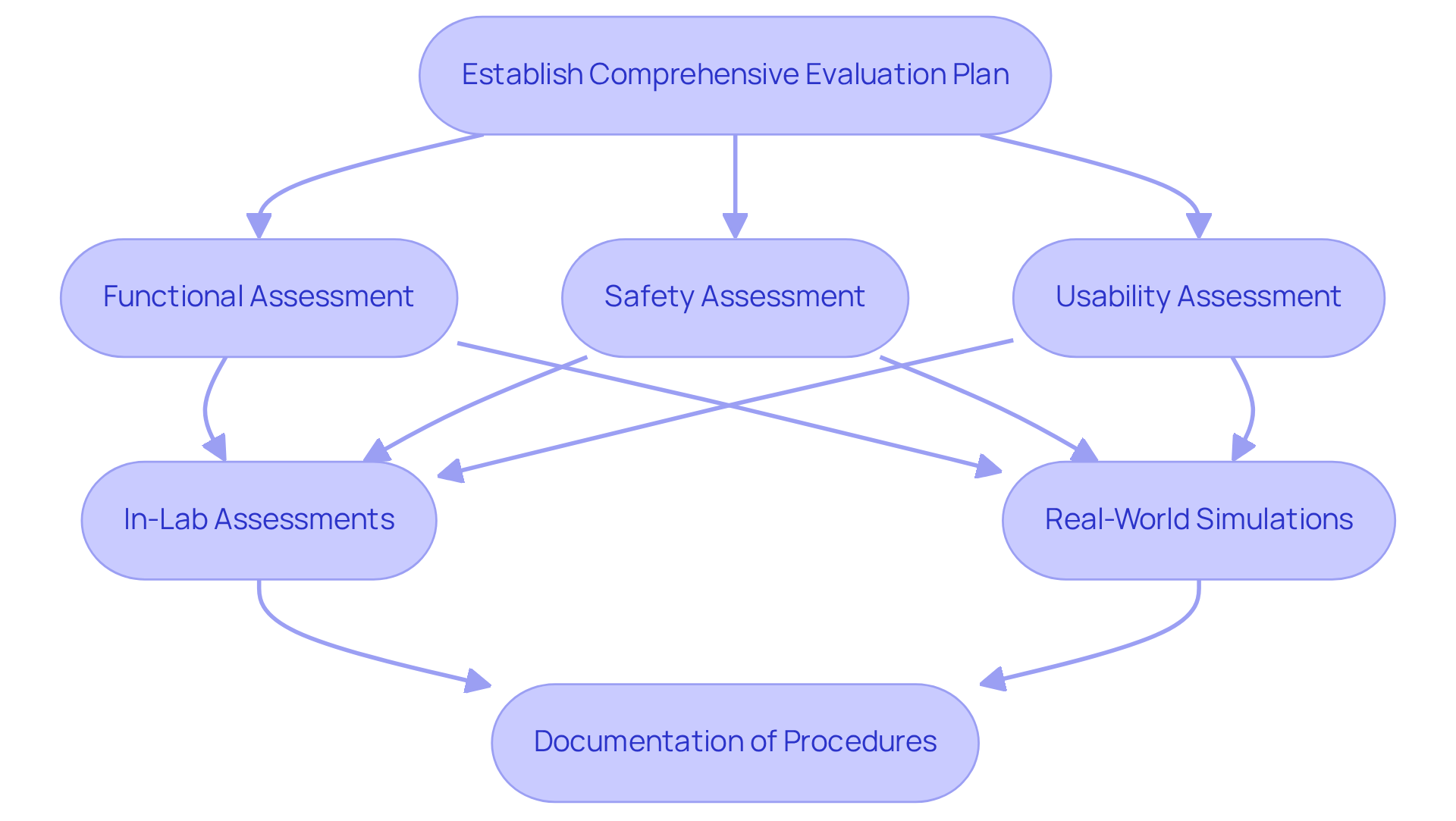
Testing and validation are foundational elements in medical equipment development, yet many overlook their complexity. A comprehensive evaluation plan must be established, focusing on functional, safety, and usability assessments. This plan should include both in-lab assessments and real-world simulations to evaluate equipment performance across diverse conditions.
For instance, research indicates that usability evaluation with real participants is critical for uncovering potential issues that may not surface in controlled environments. This evaluation is critical for driving significant enhancements in device design and boosting overall customer satisfaction. Organizations that prioritize usability evaluations often experience improved compliance and user acceptance rates.
Furthermore, meticulous documentation of all evaluation procedures is vital to demonstrate compliance with legal standards. This documentation should encompass test protocols, results, and any corrective actions implemented. Regular evaluations and updates to testing protocols are imperative to integrate emerging technologies and methodologies effectively, ensuring that the product consistently meets the highest standards of safety and efficacy. Voler Systems provides thorough documentation compliance assistance, which is crucial for managing the intricacies of regulatory issues, as highlighted in several case studies and manuals that showcase successful results in medical equipment development. Ultimately, neglecting thorough evaluation processes can lead to costly setbacks in regulatory compliance and user trust.
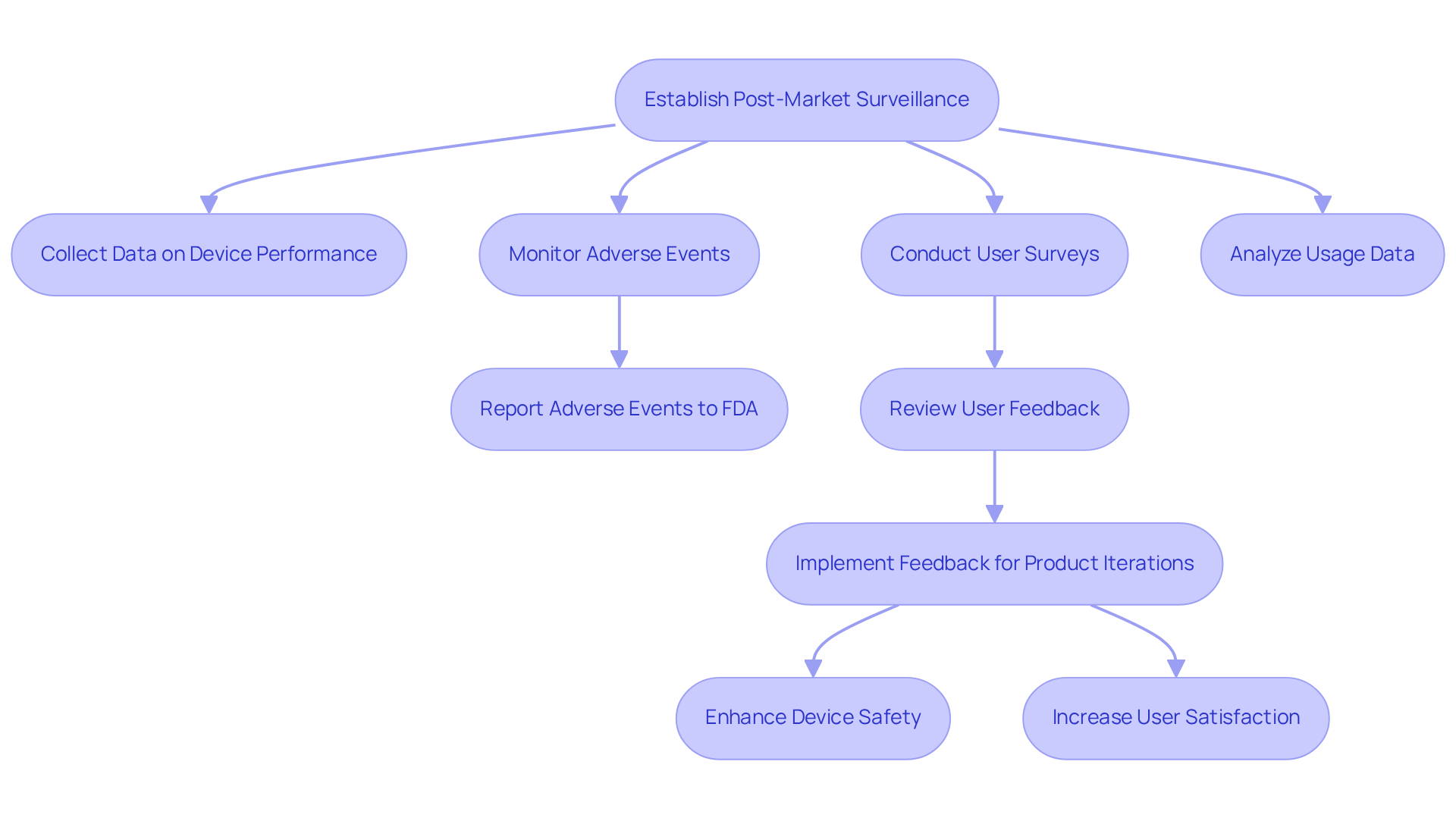
Effective post-market surveillance is essential for ensuring the safety and efficacy of medical devices throughout their lifecycle. Establish a comprehensive system for collecting and analyzing data on device performance and user feedback following product launch. This includes monitoring adverse events, conducting user surveys, and analyzing usage data to identify performance trends.
For instance, the FDA mandates that manufacturers report any device-related adverse events, providing valuable insights into potential safety issues. A proactive approach to post-market surveillance enables early identification of issues, facilitating prompt corrective measures to enhance device safety.
To establish effective feedback mechanisms, implement dedicated channels, such as hotlines and online platforms, for users to report issues efficiently. Regularly review this feedback to inform future product iterations and enhancements. By cultivating a culture of continuous improvement, manufacturers can systematically enhance device safety and increase user satisfaction through iterative feedback and innovation.
In medical equipment development, balancing regulatory compliance with user-centric design presents significant challenges. By prioritizing safety standards and user needs, developers can create devices that meet legal requirements while enhancing patient outcomes and satisfaction.
Throughout this article, we have examined critical practices for effective medical equipment development. These include:
Each of these practices plays a vital role in ensuring that medical devices are safe, effective, and user-friendly, ultimately contributing to improved healthcare delivery.
Medical device development requires ongoing commitment to quality and user feedback beyond the launch phase. By fostering a culture of continuous improvement and actively engaging with users, manufacturers can adapt and refine their products over time. Ultimately, the commitment to continuous improvement and user engagement will determine the success of medical devices in meeting evolving healthcare demands.
What are the key regulatory bodies involved in medical equipment development?
The key regulatory bodies involved are the FDA (Food and Drug Administration) and the EMA (European Medicines Agency), which govern the safety and efficacy of medical devices.
What is the importance of identifying regulatory requirements in medical equipment development?
Identifying regulatory requirements is critical to ensure compliance with safety and efficacy standards, which includes guidelines for premarket submissions, quality management systems, and post-market surveillance.
What recent proposals aim to improve compliance processes in medical equipment development?
Recent proposals published on December 16, 2025, aim to streamline compliance processes, enhancing predictability while maintaining patient safety.
When is the feedback period for the recent proposals related to regulatory requirements?
The feedback period for these proposals extends from January 7, 2026, to March 5, 2026.
How can developers collect insights into user needs during medical equipment development?
Developers can collect insights through surveys, interviews, and observational studies with prospective participants to understand their needs and challenges.
What is the significance of involving users in the medical equipment development process?
Involving users significantly lowers the chance of usability mistakes, thereby improving patient safety and satisfaction.
What should a requirements document for medical equipment development include?
A requirements document should detail both compliance standards and client needs, and it should be routinely revised to incorporate new regulations and feedback.
What role do regulatory sandboxes play in medical equipment development?
Regulatory sandboxes support evidence generation for innovative technologies, allowing for more flexible compliance pathways.
How does aligning compliance with user feedback impact medical devices?
Aligning compliance with user feedback can significantly enhance the effectiveness of devices and improve patient safety.
