Introduction
Identifying potential failure modes in medical devices is essential for safeguarding patient safety and ensuring product efficacy. The user Failure Mode and Effects Analysis (FMEA) provides a structured approach for manufacturers to identify and mitigate risks before products reach the market, significantly enhancing compliance and trust.
Organizations face significant challenges in effectively implementing user FMEA amidst evolving medical technologies and regulatory complexities. Navigating these complexities is essential for organizations aiming to uphold the highest safety standards in an evolving regulatory landscape.
Understand FMEA in the Context of Medical Devices
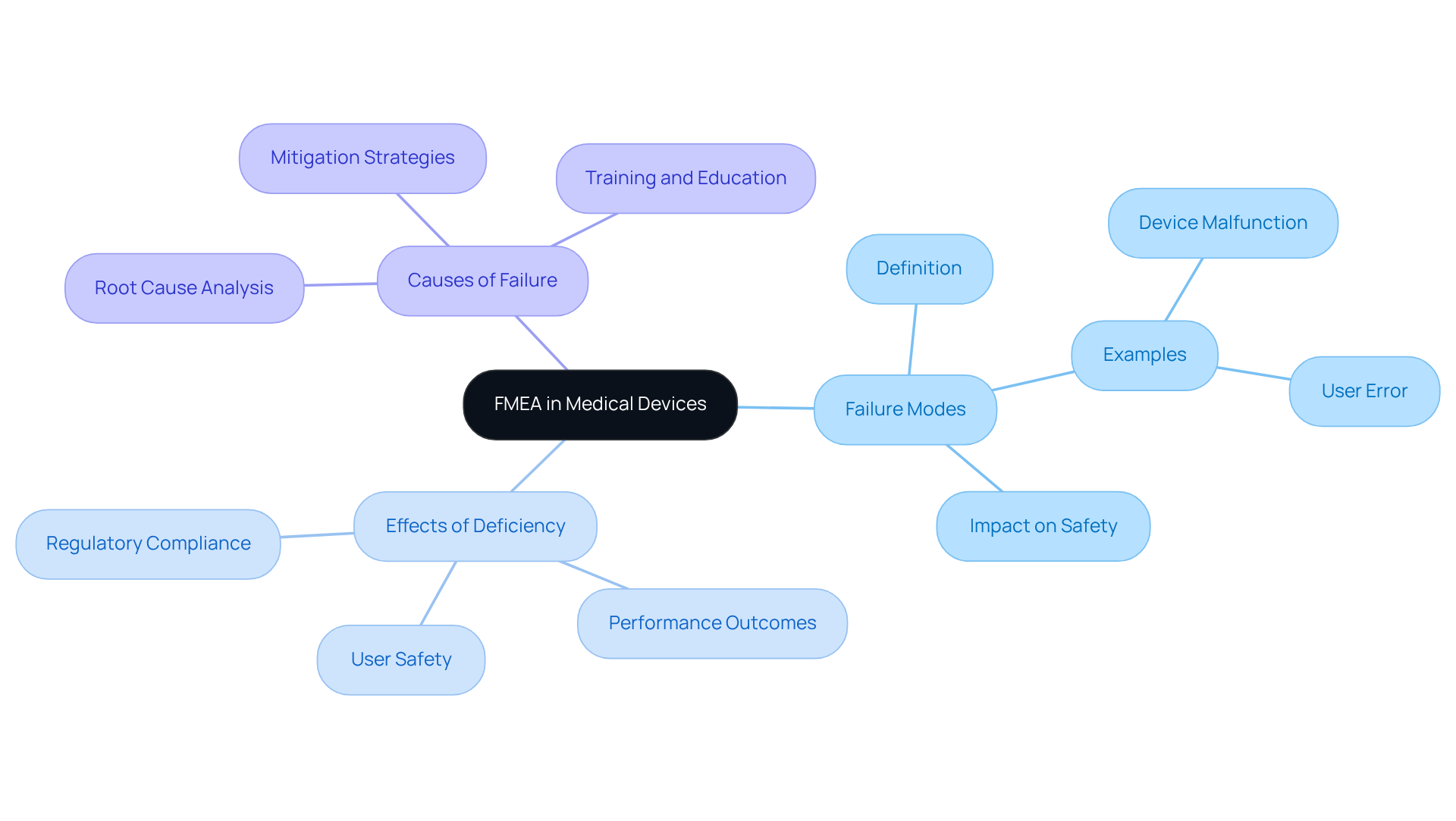
Recognizing potential failure modes in healthcare equipment is crucial for ensuring patient safety and regulatory compliance. User FMEA is a structured method aimed at identifying possible failure modes within a product or process, evaluating their effects, and ranking them according to seriousness and probability. In the field of healthcare equipment, the implementation of user FMEA is essential for guaranteeing that products are both safe and effective before entering the market. This proactive methodology enables manufacturers to identify risks associated with functionality and user interactions. As a result, it enhances patient safety and ensures compliance with regulatory standards.
Key components of FMEA include:
- Failure Modes: These represent the various ways a device can fail to fulfill its intended function.
- Effects of Deficiency: This relates to the outcomes of each malfunction type on the apparatus's performance and user safety.
- Causes of Failure: Identifying the root causes of potential failures is essential for formulating effective mitigation strategies.
The implementation of user FMEA has demonstrated measurable improvements in patient safety outcomes throughout the medical equipment sector. For example, research has demonstrated that applying FMEA can result in a considerable decrease in negative occurrences, with one study indicating a reduction in multi-drug-resistant bacteria outbreaks in ICU environments after the use of this method. Additionally, the user FMEA is recognized as the most commonly used risk assessment tool in the medical equipment sector, with 79.14% of studies implementing it in healthcare processes.
Establishing robust testing systems is essential for ensuring quality and reliability in electronic products during development. By establishing effective testing protocols, manufacturers can identify common errors in manufacturing tests early, thus enhancing efficiency and product quality. This proactive approach not only aids in compliance with stringent regulatory standards but also fosters a culture of safety and quality within the industry.
In 2026, the significance of user FMEA continues to increase as medical technologies advance, and the need for robust risk management strategies becomes increasingly critical. By utilizing user FMEA, medical equipment professionals can proactively tackle risks, resulting in improved product reliability and heightened user confidence. This structured approach not only aids in compliance with stringent regulatory standards but also fosters a culture of safety and quality within the industry. As medical technologies evolve, the absence of FMEA could lead to significant risks that undermine patient trust and safety.
Follow the Step-by-Step FMEA Process
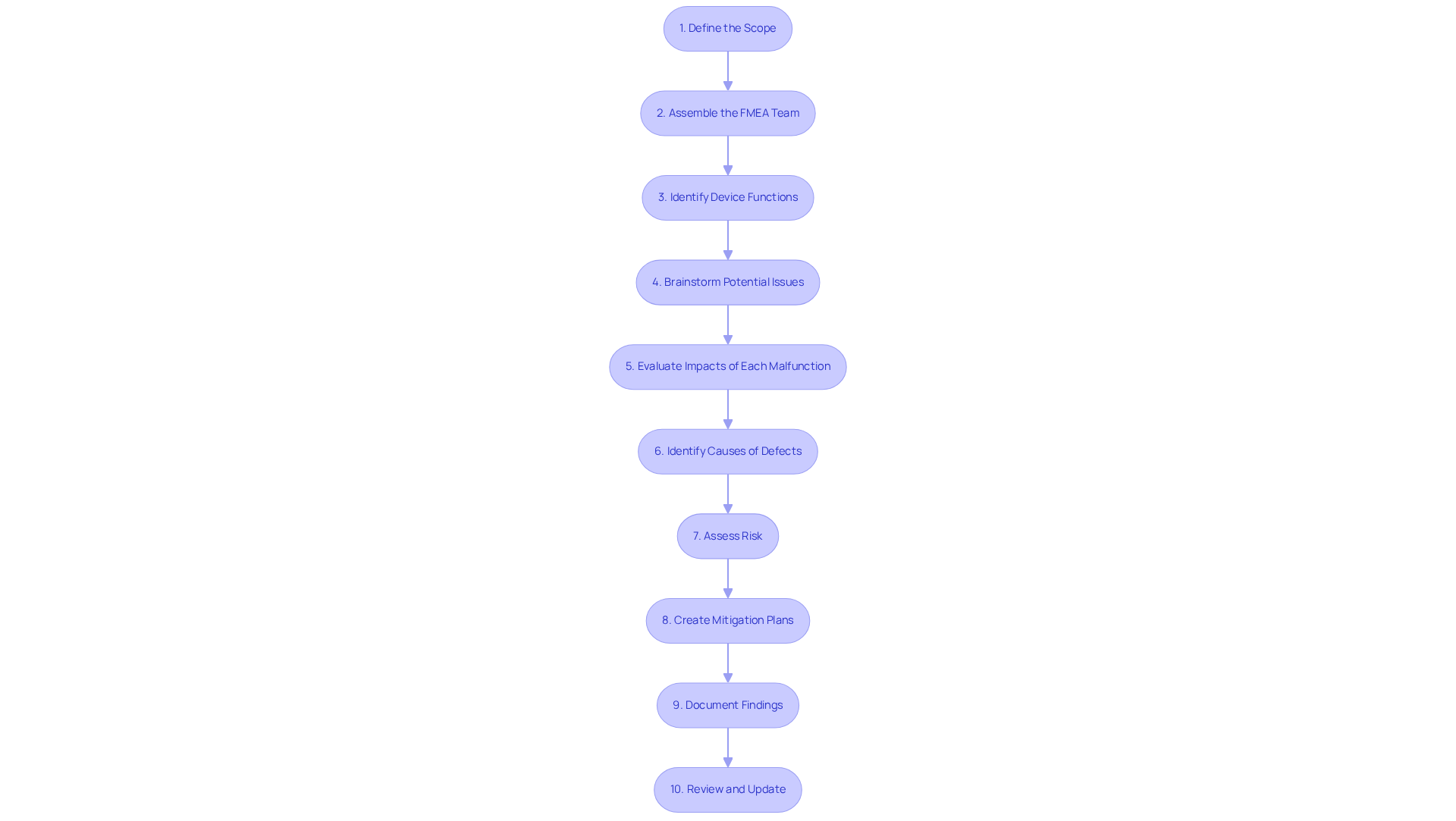
To ensure the safety and efficacy of medical devices, a systematic approach to user FMEA is essential. Follow these steps to conduct an effective FMEA:
- Define the Scope: Clearly outline the system or process to be analyzed, identifying the specific unit or component and its intended use. This step lays the groundwork for a focused and relevant analysis.
- Assemble the FMEA Team: Gather a cross-functional team with diverse expertise, including engineering, quality assurance, and regulatory affairs. This diversity enhances the analysis by incorporating various perspectives, leading to a more comprehensive understanding of potential risks.
- Identify Device Functions: List all functions of the device, focusing on what it is supposed to do under normal operating conditions. Grasping these functions is essential for identifying possible breakdown scenarios.
- Brainstorm Potential Issues: For each function, identify ways it could malfunction, considering both technical problems and user mistakes. Engaging the team in brainstorming sessions can uncover a wide range of potential issues.
- Evaluate Impacts of Each Malfunction: Identify the repercussions of each malfunction type on device performance and patient safety. This evaluation assists in prioritizing which issues require immediate attention.
- Identify Causes of Defects: Analyze why each defect type could occur, considering design flaws, manufacturing issues, and user interactions. Grasping the root causes is crucial for crafting effective strategies to mitigate risks.
- Assess Risk: Allocate a Risk Priority Number (RPN) to each defect type by multiplying its severity, occurrence, and detection ratings. This quantification assists in prioritizing which issues to tackle first, guiding resource allocation effectively.
- Create Mitigation Plans: For high-priority risk types, outline actions to minimize or remove the threat. This may encompass design modifications, extra testing, or user training, ensuring that the product meets safety and performance standards.
- Document Findings: Keep thorough records of the failure mode and effects analysis process, including identified failure modes, their effects, causes, and mitigation strategies. This documentation acts as a dynamic record that helps ensure compliance and fosters continuous improvement.
- Review and Update: Regularly review the failure mode and effects analysis as product designs evolve or new information emerges, ensuring that it remains pertinent and effective. Continuous assessment is vital for adapting to changes in technology and regulatory requirements. By continuously updating the FMEA, organizations can adapt to evolving challenges and maintain high standards of patient safety.
Engage Cross-Functional Teams for Effective Analysis
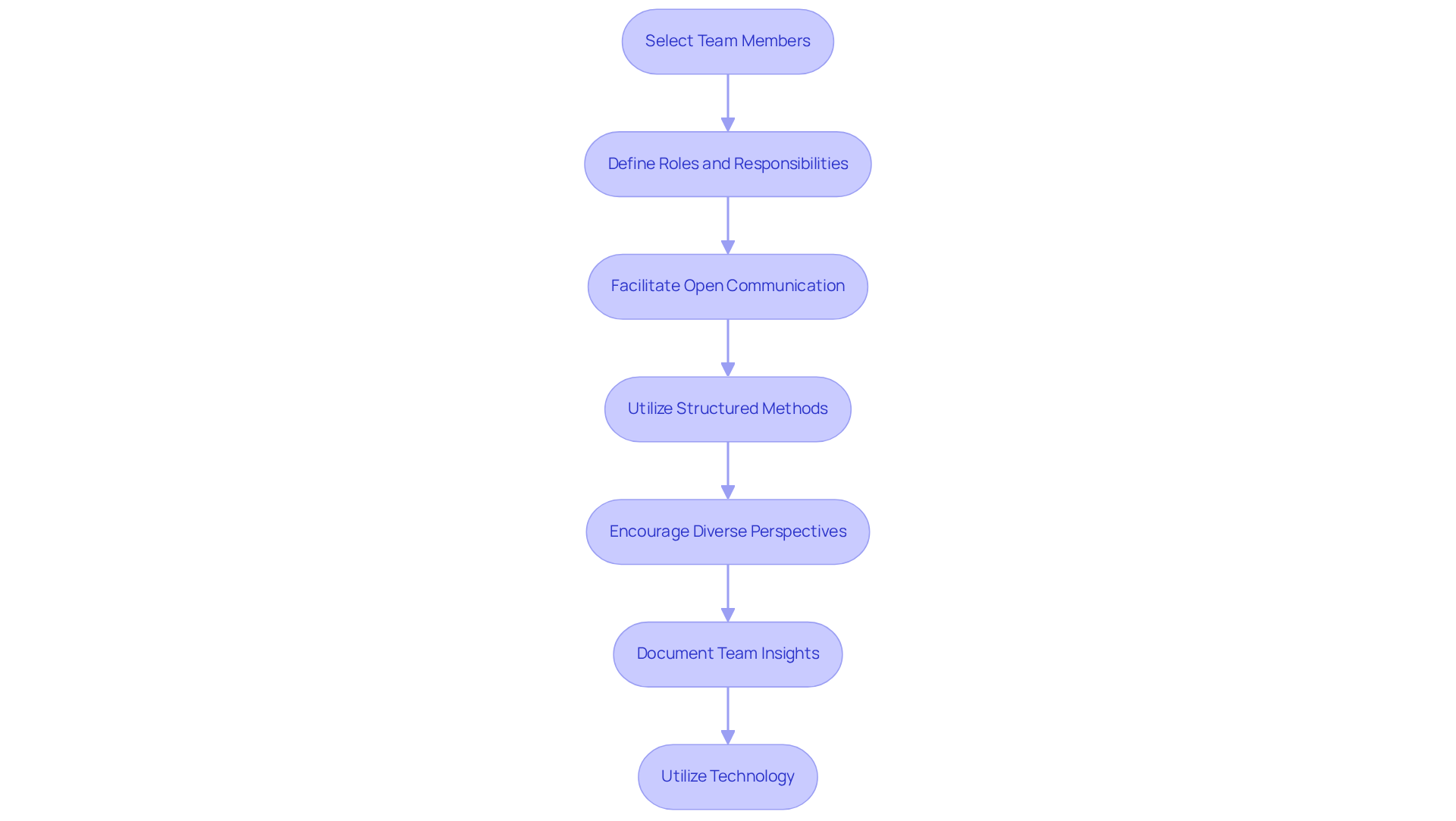
Engaging a cross-functional team is crucial for maximizing the effectiveness of your user FMEA. Here’s how to effectively assemble and utilize such a team:
- Select Team Members: Choose individuals from diverse departments, including engineering, quality assurance, regulatory affairs, and manufacturing. Each member should contribute unique insights relevant to the item under analysis, particularly in light of increasing regulatory complexity and adherence to standards such as ISO 14971. Voler Systems brings extensive experience in supporting medical device firms with legacy test equipment, providing valuable insights through user FMEA case studies of successful failure mode and effects analysis.
- Define Roles and Responsibilities: Clearly outline each team member's role in the failure mode and effects analysis process to ensure accountability and encourage active participation. This clarity helps avoid overlap and confusion, enhancing team efficiency.
- Facilitate Open Communication: Foster an environment where team members feel comfortable sharing their perspectives and concerns. Regular meetings and updates can help maintain engagement and collaboration, ensuring that all voices are heard.
- Utilize Structured Methods: Implement structured brainstorming techniques to ensure comprehensive consideration of all potential failure modes. Techniques such as the Delphi method or fishbone diagrams are highly effective in steering discussions.
- Encourage Diverse Perspectives: Promote discussions that challenge assumptions and explore different viewpoints. This diversity can result in more comprehensive risk identification and improve the overall quality of the analysis outcomes.
- Document Team Insights: Maintain detailed records of discussions and decisions made during team meetings. This documentation is essential for traceability and serves as a reference for future analyses as medical device manufacturing evolves with technology and cybersecurity challenges.
- Utilize Technology: Reflect on the impact of automated testing solutions and robotics in improving accuracy and consistency throughout the failure mode and effects analysis process. Voler Systems' commitment to innovative medical technology development ensures that these technologies can support effective risk management and compliance efforts.
Ultimately, leveraging diverse insights not only strengthens the analysis but also fortifies compliance in an evolving regulatory landscape.
Document Findings and Ensure Traceability
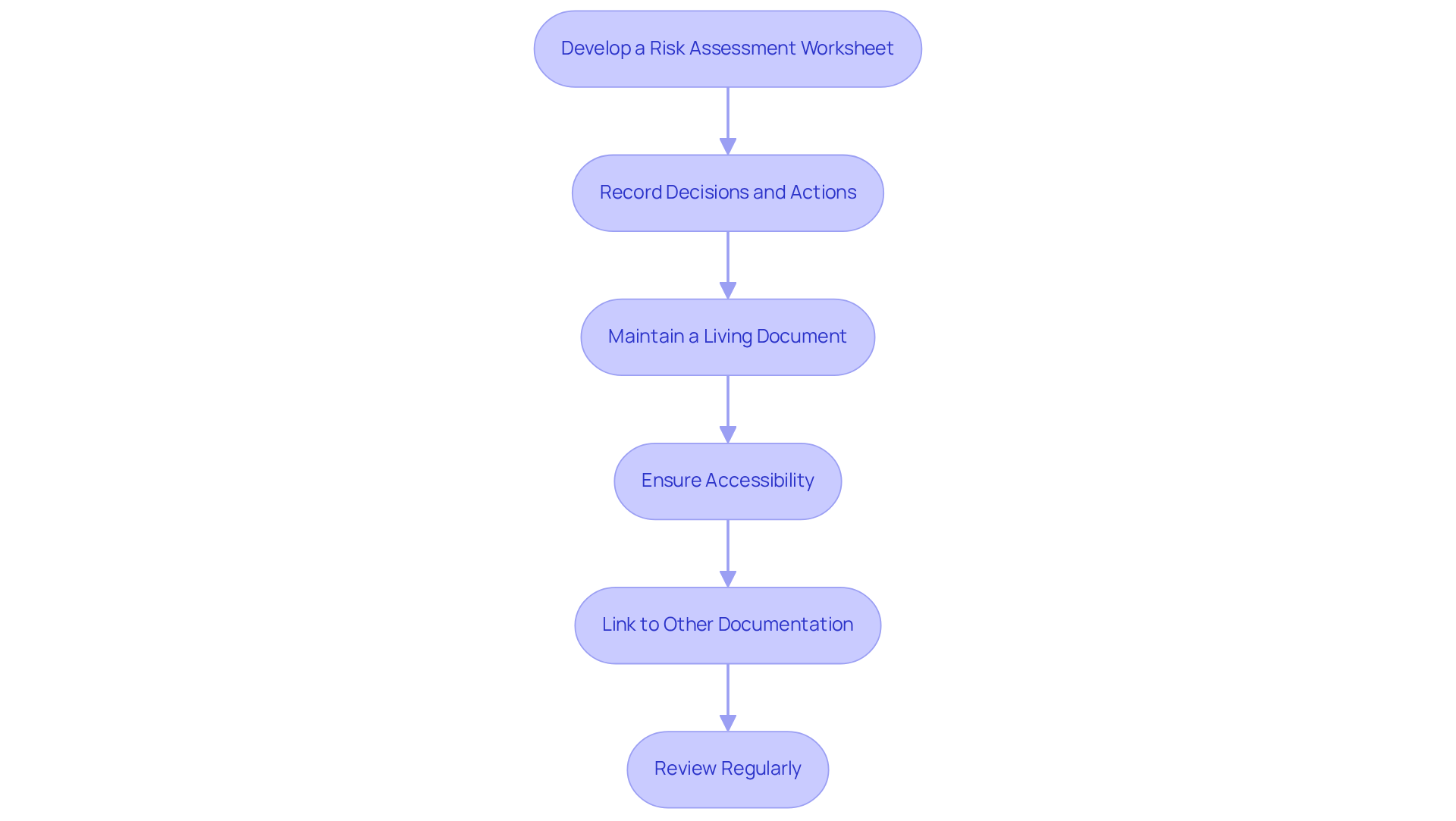
Effective documentation is a cornerstone of successful user FMEA in the medical device industry. Follow these guidelines to ensure thorough record-keeping:
- Develop a Risk Assessment Worksheet: Utilize a standardized template to record all components of the risk assessment procedure, including recognized issues, impacts, causes, and prevention strategies. For example, a thorough worksheet for analyzing potential issues can assist in recognizing possible problems early in the design stage, as shown in a case study related to a wearable health monitor.
- Record Decisions and Actions: Document the reasoning behind decisions made during the failure mode and effects analysis, including the prioritization of failure modes and the selected mitigation strategies. This practice is essential, particularly with the user FMEA in the context of the FDA's Quality Management System Regulation (QMSR) effective February 2, 2026. It underscores the need for thorough documentation and compliance support, an area where Voler Systems excels in assisting startups.
- Maintain a Living Document: Treat the failure mode and effects analysis as a dynamic document that is regularly updated as new information becomes available or as the device design evolves. Regular updates are crucial for maintaining user FMEA compliance. Neglecting follow-up actions on identified threats can undermine the analysis's significance.
- Ensure Accessibility: Store failure mode and effects analysis documentation in a centralized location where all team members can easily access it. This promotes transparency and facilitates collaboration, which is essential for effective risk management.
- Link to Other Documentation: Ensure that the Failure Mode and Effects Analysis is connected to other relevant documents, such as design specifications, risk management files, and regulatory submissions. This traceability is crucial for compliance and audits, particularly with the new QMSR requirements, where Voler Systems' expertise can guide startups in implementing user FMEA to maintain proper documentation practices.
- Review Regularly: Schedule periodic reviews of the FMEA documentation to ensure it remains current and reflects any changes in the device or its intended use. Regular reviews help maintain compliance and improve overall product safety, as highlighted by industry experts like Mike Wolf, who emphasize the importance of continuous improvement in documentation practices. Failure to maintain proper documentation can result in regulatory penalties and jeopardize patient safety.
Conclusion
The absence of a structured approach to risk management in medical device development can lead to significant safety concerns. Implementing a user Failure Mode and Effects Analysis (FMEA) is essential for the success of medical devices, as it serves as a proactive approach to identifying and mitigating potential risks. By recognizing possible failure modes, evaluating their effects, and prioritizing them effectively, manufacturers can enhance patient safety and ensure compliance with regulatory standards. The structured methodology of user FMEA fosters safety and quality while enhancing user confidence in medical technologies.
Throughout this guide, the importance of a systematic step-by-step FMEA process has been emphasized, highlighting key aspects such as:
- Defining the scope
- Engaging cross-functional teams
- Maintaining thorough documentation
Each step is critical for identifying and addressing potential risks, thereby improving product reliability. Furthermore, the integration of diverse perspectives within teams enhances the quality of the analysis, ensuring a more comprehensive understanding of potential issues.
In conclusion, the application of user FMEA in medical device development is not just a regulatory requirement; it is a vital practice that safeguards patient safety and promotes innovation in healthcare technology. Manufacturers that prioritize FMEA will not only enhance patient safety but also position themselves as leaders in innovation within the healthcare sector.
Frequently Asked Questions
What is FMEA in the context of medical devices?
FMEA, or Failure Mode and Effects Analysis, is a structured method used to identify potential failure modes in healthcare equipment, evaluate their effects, and rank them based on seriousness and probability to ensure patient safety and regulatory compliance.
Why is user FMEA important for medical devices?
User FMEA is essential for guaranteeing that medical products are safe and effective before entering the market. It helps manufacturers identify risks associated with functionality and user interactions, enhancing patient safety and ensuring compliance with regulatory standards.
What are the key components of FMEA?
The key components of FMEA include: - Failure Modes: Various ways a device can fail to fulfill its intended function. - Effects of Deficiency: Outcomes of each malfunction type on the device's performance and user safety. - Causes of Failure: Root causes of potential failures, which are essential for developing effective mitigation strategies.
How has user FMEA impacted patient safety outcomes in the medical equipment sector?
The implementation of user FMEA has led to measurable improvements in patient safety outcomes, including a significant decrease in negative occurrences, such as a reduction in multi-drug-resistant bacteria outbreaks in ICU environments.
How commonly is user FMEA used in the medical equipment sector?
User FMEA is recognized as the most commonly used risk assessment tool in the medical equipment sector, with 79.14% of studies implementing it in healthcare processes.
Why is establishing robust testing systems important during the development of electronic products?
Establishing robust testing systems is crucial for ensuring quality and reliability in electronic products. Effective testing protocols help identify common errors early, enhancing efficiency and product quality while aiding compliance with regulatory standards.
What is the future significance of user FMEA as medical technologies advance?
As medical technologies evolve, the significance of user FMEA continues to increase, becoming critical for robust risk management strategies. It helps tackle risks proactively, resulting in improved product reliability and heightened user confidence.
What could happen if FMEA is not implemented in medical device development?
The absence of FMEA could lead to significant risks that undermine patient trust and safety, as it is essential for identifying and mitigating potential failures in medical devices.
List of Sources
- Understand FMEA in the Context of Medical Devices
- Failure mode effect analysis use and limitations in medical device risk management (https://sciencedirect.com/science/article/pii/S2199853124002336)
- Data-driven FMEA approach for hazard identification and risk evaluation in digital health - Scientific Reports (https://nature.com/articles/s41598-025-11929-4)
- 23 - 24 June 2026: Failure Mode and Effects Analysis (FMEA) in Medical Device Manufacturing - AMMI (https://ammi.com.my/fmea-2026)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC11719677)
- Follow the Step-by-Step FMEA Process
- How to do FMEA for Medical Devices: Step-by-Step Guide to Risk Management - SoftComply (https://softcomply.com/fmea-medical-devices-risk-management-guide)
- FMEA Explained: Failure Mode & Effects Analysis Steps & Examples (https://sologic.com/en-us/resources/learning/what-is-fmea)
- meddeviceguide.com (https://meddeviceguide.com/blog/fmea-medical-devices-guide)
- Implementing System FMEA: A Step-by-Step Example for Medical Devices (https://volersystems.com/blog/implementing-system-fmea-a-step-by-step-example-for-medical-devices)
- Master FMEA Failure: Steps to Enhance Medical Device Safety (https://volersystems.com/blog/master-fmea-failure-steps-to-enhance-medical-device-safety)
- Engage Cross-Functional Teams for Effective Analysis
- Master FMEA Failure: Steps to Enhance Medical Device Safety (https://volersystems.com/blog/master-fmea-failure-steps-to-enhance-medical-device-safety)
- Future of Medical Device Manufacturing: Trends & Challenges 2026 (https://insight.averna.com/en/resources/blog/navigating-the-future-of-medical-device-manufacturing)
- FMEA: Why You're Missing Critical Risks in Medical Device Lifecycle Management (https://propharmagroup.com/thought-leadership/fmea-missing-critical-risks-medical-device-lifecycle-management)
- Document Findings and Ensure Traceability
- Master FMEA Documents for Medical Device Success and Compliance (https://volersystems.com/blog/master-fmea-documents-for-medical-device-success-and-compliance)
- iqvia.com (https://iqvia.com/blogs/2026/01/fda-updates-guidance-on-real-world-evidence-for-medical-devices)
- CDRH Proposed Guidances for Fiscal Year 2026 (FY2026) (https://fda.gov/medical-devices/guidance-documents-medical-devices-and-radiation-emitting-products/cdrh-proposed-guidances-fiscal-year-2026-fy2026)
- FDA’s New QMSR Rule for 2026: What Medical Device Manufacturers Must Do Now (https://theoris.com/fdas-new-qmsr-rule-for-2026-what-medical-device-manufacturers-must-do-now)
- Medical Device Compliance: 2025 Insights & 2026 Priorities (https://medenvoyglobal.com/blog/medical-device-compliance-2025-insights-2026-priorities)





