What Are the Essential Parts for Constructing a Product?
Explore what do you call the parts you need to construct a product and their vital roles...

The world of medical devices is fundamentally shaped by the concept of product architecture, a framework that defines how components interact and function together. Grasping this architecture is crucial for designing effective and compliant healthcare solutions that not only adhere to regulatory standards but also improve patient outcomes. As the industry progresses, manufacturers encounter the challenge of integrating advanced technologies, such as artificial intelligence, while ensuring that their designs remain safe and efficient. Organizations must navigate these complexities to develop innovative medical devices that distinguish themselves in a competitive market.
A showcases the design of a system, which involves the organized layout of its functional components and the interactions among these components to achieve the intended purpose. In healthcare equipment, the design structure is crucial as it directly influences performance, manufacturability, and compliance with regulatory standards. A well-defined [product architecture example](https://performa-code.com/blog/medtech-compliance-architecture-why-compliant-devices-still-fail-in-the-field) ensures that all elements operate cohesively, which is essential for maintaining the safety and efficacy of medical instruments. This product architecture example encompasses both the physical arrangement of components and their logical relationships, impacting every aspect from design to production and user experience.
For instance, the evolution of MedTech portfolios from single-function tools to multi-modal systems is a product architecture example that illustrates the necessity for . These systems, which encompass diagnosis, monitoring, and therapy, are increasingly sought after by for their capacity to facilitate coordinated care throughout the patient journey. The integration of AI and machine learning into equipment further underscores the importance of a strong product architecture example, as these technologies require meticulous structuring to ensure compliance and reliability. has developed a range of , including wearable devices and heart pumps, leveraging AI-assisted engineering to enhance the design and development of these complex systems, ensuring they are delivered on time, within budget, and are easy to manufacture.
Moreover, the regulatory landscape is evolving, with the FDA intensifying its evaluation processes for software-driven and AI-enabled healthcare tools. This shift necessitates that manufacturers prioritize item design to meet . In 2024, equipment failure emerged as the leading cause of recalls, surpassing manufacturing defects, underscoring the critical need for a product architecture example that can effectively mitigate risks associated with design and development.
As noted by industry experts, not only enhances compliance but also significantly impacts the overall safety and efficacy of . Organizations that adeptly navigate the complexities of their offerings can accelerate their time to market while maintaining a solid technical roadmap, ultimately leading to improved patient outcomes and satisfaction.
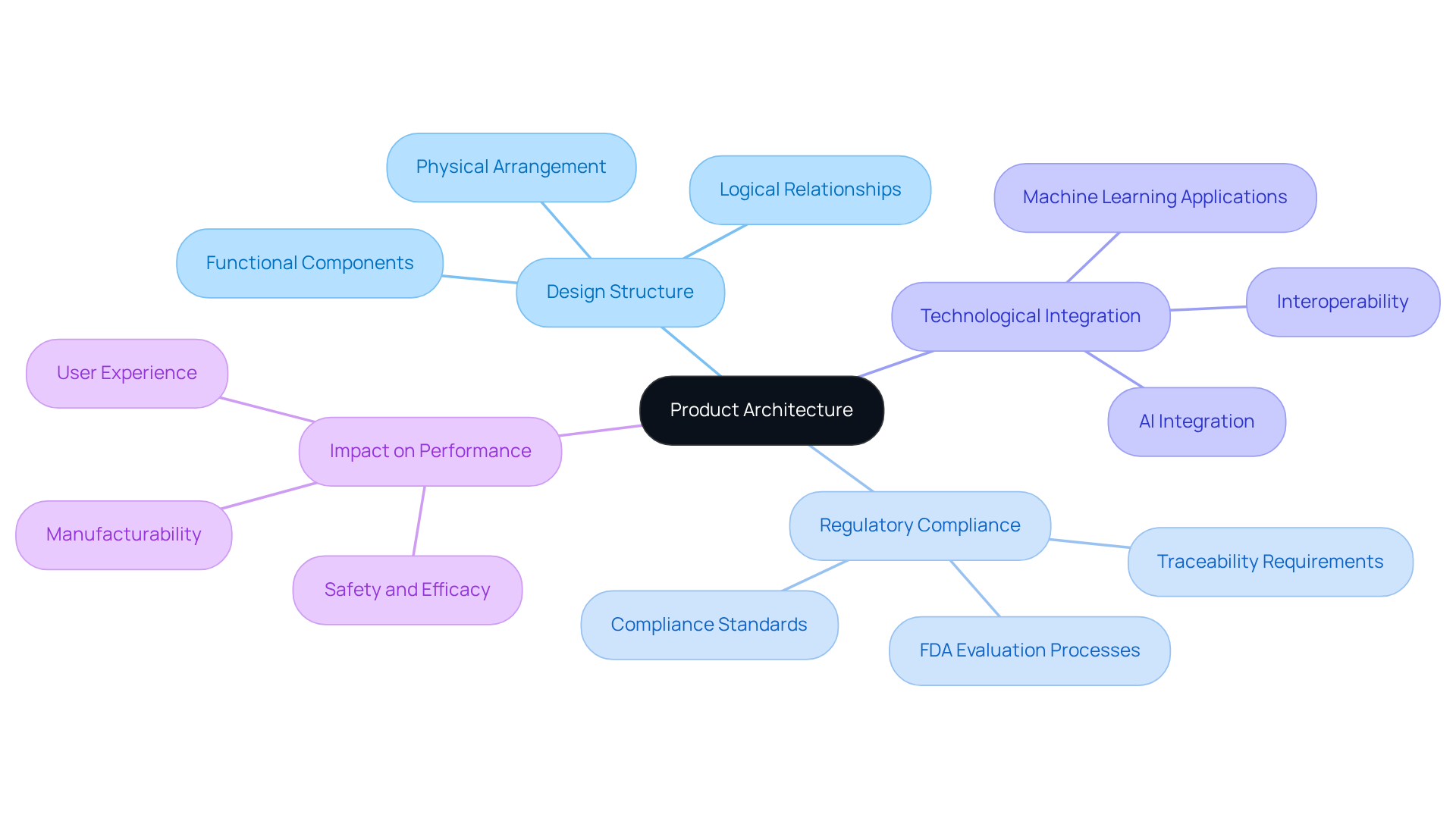
The evolution of item structure has its roots in the early days of industrial design, where the primary focus was on functionality and efficiency. Over the decades, advancements in technology, materials, and manufacturing processes have elevated product design into a more sophisticated discipline. In the healthcare equipment sector, the integration of digital technologies and artificial intelligence has revolutionized design practices, serving as a of more complex and integrated architectures.
For instance, has been instrumental in advancing , contributing to aimed at neurological pain management. The shift from analog to digital systems has enabled the development of tools that are not only more efficient but also capable of -essential features for modern healthcare applications.
Moreover, innovations in electronic have significantly enhanced the capabilities of medical devices. This is exemplified by the creation of during .
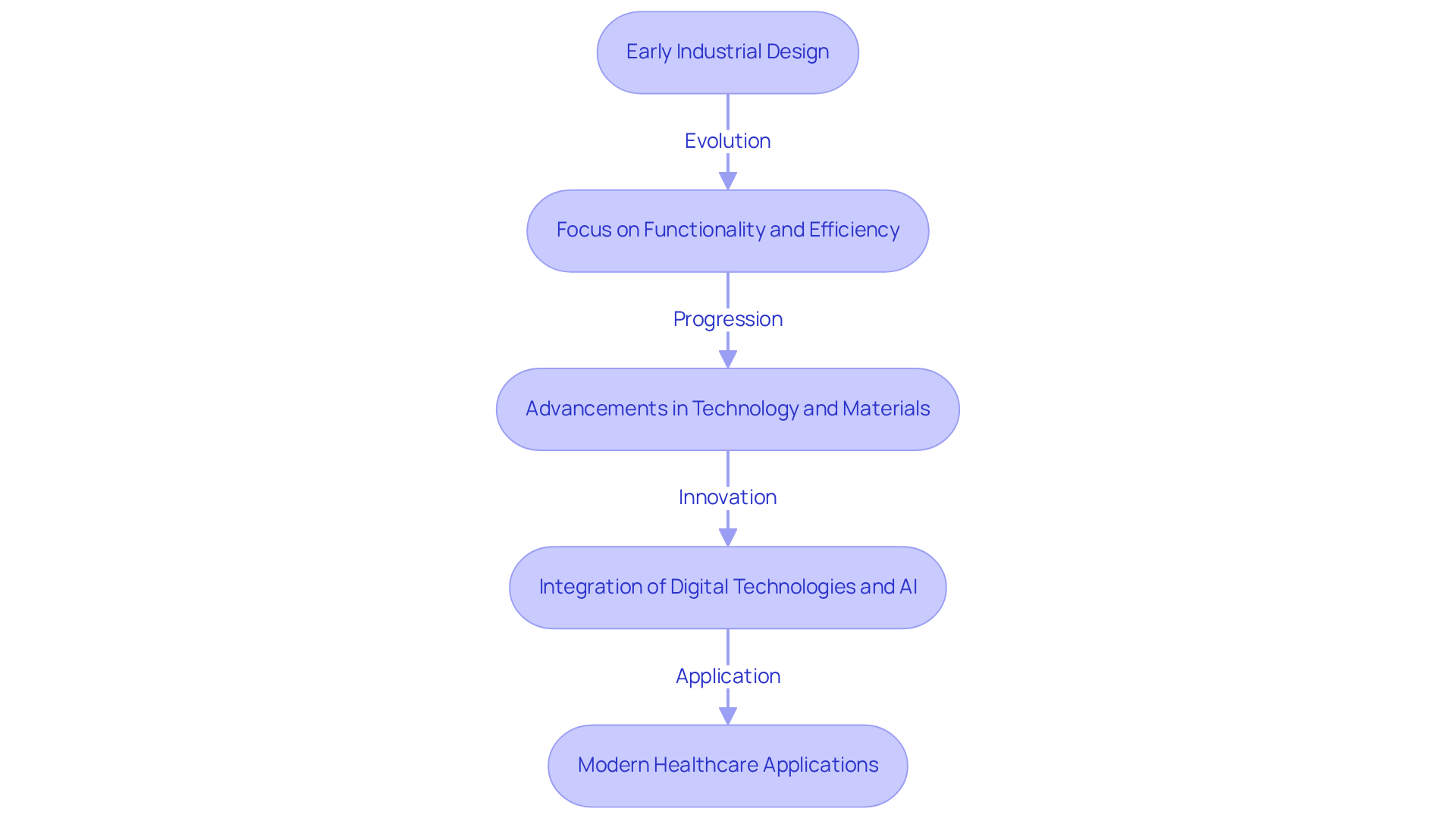
can be categorized into two main types: modular and integral.
In contrast, , enhancing performance but complicating repairs and upgrades.
Understanding these architectural types provides a that enables designers to make that meet regulatory requirements and user needs, ultimately supporting the development of innovative and reliable .
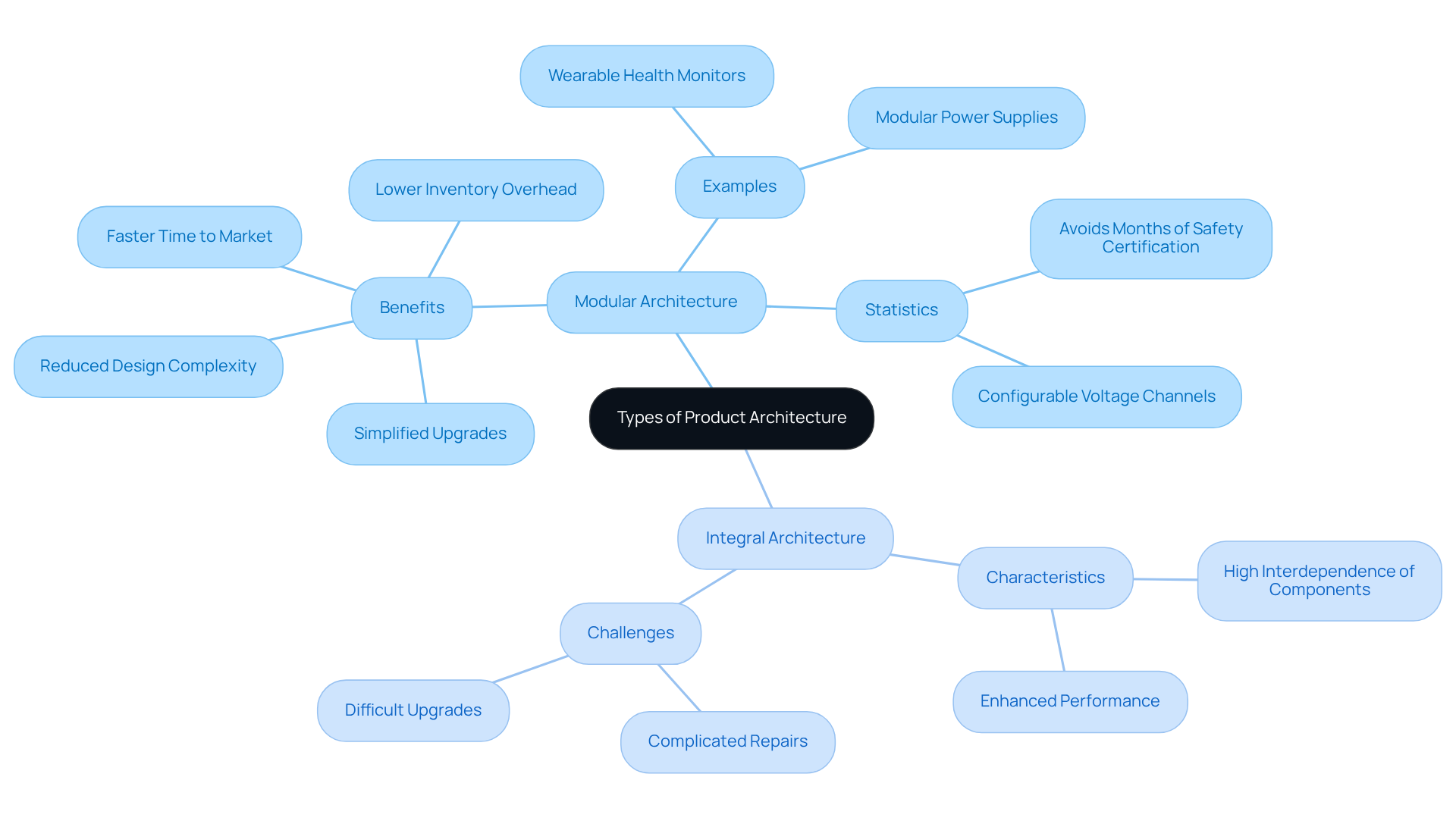
The structure of goods is pivotal in the development lifecycle, influencing aspects from initial concept design to final production. A well-defined structure streamlines the development process by clarifying the relationships between components, which can significantly . In the realm of , adhering to a robust design framework is essential for compliance with , a critical factor for securing market approval. Moreover, effective among cross-functional teams, allowing engineers, designers, and regulatory experts to work together efficiently. This collaborative approach is vital for creating that meet the evolving needs of providers and patients.
At Voler Systems, we leverage cutting-edge , incorporating FPGA and AI technologies to enhance the performance and efficiency of . Our commitment to optimizing battery life through ensures that our devices not only comply with regulatory standards but also deliver reliable performance in demanding healthcare environments.
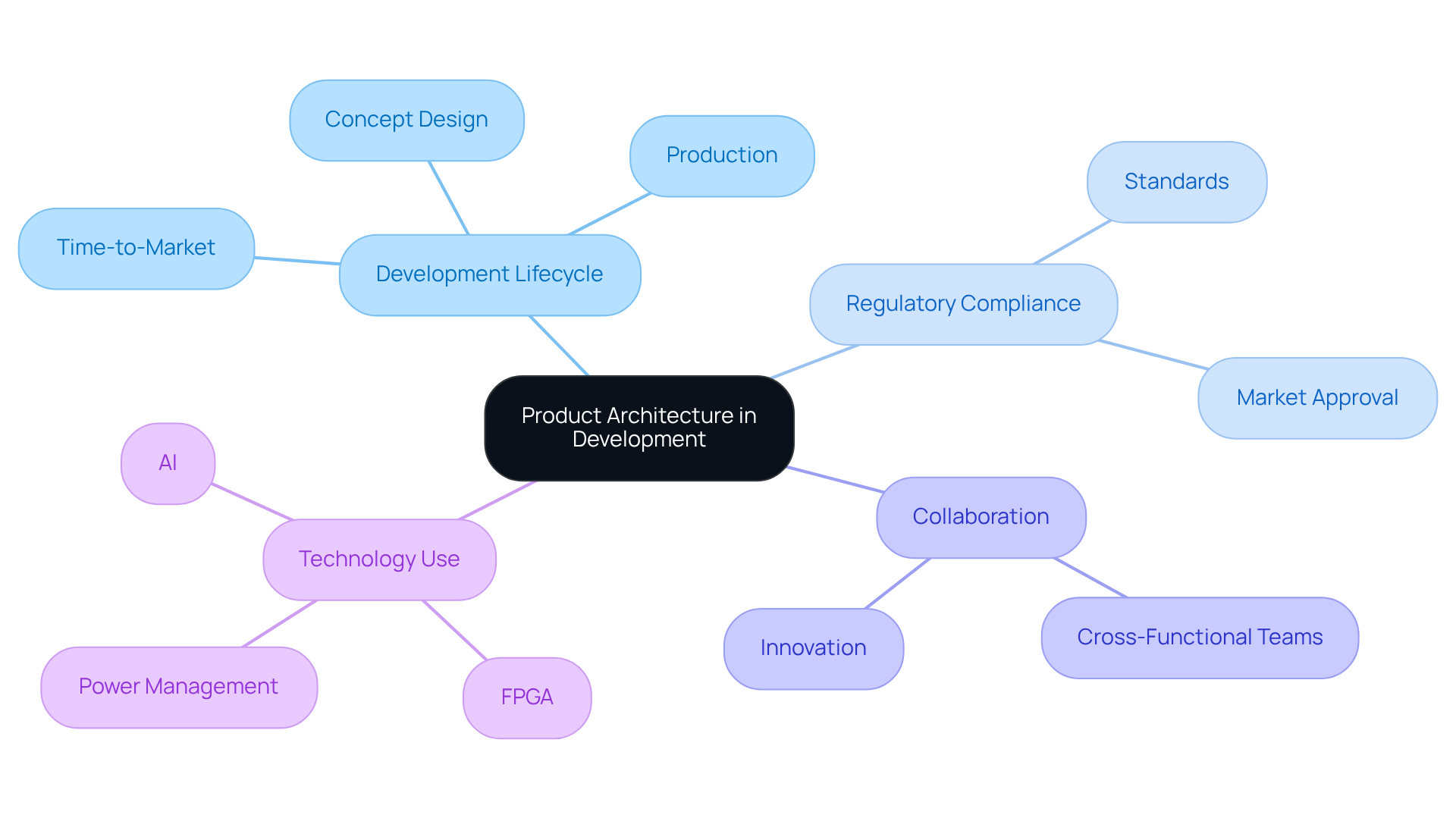
A comprehensive understanding of product architecture is crucial for the successful design and development of medical devices. This structured approach to organizing functional components and their interactions enhances performance and manufacturability while ensuring compliance with regulatory standards. The evolution of product architecture, particularly within healthcare, underscores the necessity for integrated designs that address the complexities of modern medical applications, ultimately leading to improved patient outcomes.
Key insights from the discussion reveal a transition from traditional single-function tools to advanced multi-modal systems that incorporate AI and digital technologies. The significance of modular and integral architectures is emphasized, demonstrating how these approaches can enhance the efficiency of upgrades, repairs, and overall compliance with evolving regulations. Effective product architecture plays a pivotal role in streamlining the development process and fostering collaboration among teams, which is essential for creating innovative healthcare solutions.
In conclusion, the importance of product architecture extends beyond mere design; it shapes the future of medical technology by ensuring safety, efficacy, and adaptability in a rapidly changing landscape. As the industry continues to evolve, embracing robust architectural principles will be vital for manufacturers aiming to deliver reliable, cutting-edge medical devices that meet the needs of both healthcare providers and patients. Engaging with the complexities of product architecture is not just beneficial - it is imperative for success in the healthcare technology sector.
What is product architecture in the context of healthcare equipment?
Product architecture refers to the organized layout of functional components and their interactions within a system, which is crucial for performance, manufacturability, and compliance with regulatory standards in healthcare equipment.
Why is a well-defined product architecture important for medical instruments?
A well-defined product architecture ensures that all elements operate cohesively, which is essential for maintaining the safety and efficacy of medical instruments.
How does product architecture influence the design and production process?
Product architecture impacts every aspect from design to production and user experience by defining both the physical arrangement of components and their logical relationships.
Can you provide an example of product architecture evolution in healthcare?
An example is the evolution of MedTech portfolios from single-function tools to multi-modal systems that integrate diagnosis, monitoring, and therapy, facilitating coordinated care throughout the patient journey.
How does the integration of AI and machine learning relate to product architecture?
The integration of AI and machine learning into healthcare equipment requires meticulous structuring within the product architecture to ensure compliance and reliability.
What role does Voler Systems play in healthcare product architecture?
Voler Systems develops a range of healthcare tools, including wearable devices and heart pumps, utilizing AI-assisted engineering to enhance the design and development of complex systems.
What recent changes have occurred in the regulatory landscape for healthcare tools?
The FDA has intensified its evaluation processes for software-driven and AI-enabled healthcare tools, necessitating manufacturers to prioritize design to meet stringent compliance standards.
What was the leading cause of recalls in 2024, and what does it imply for product architecture?
Equipment failure emerged as the leading cause of recalls, surpassing manufacturing defects, highlighting the critical need for a product architecture that effectively mitigates risks associated with design and development.
How does effective item design impact medical device compliance and safety?
Effective item design enhances compliance and significantly impacts the overall safety and efficacy of medical devices, leading to improved patient outcomes and satisfaction.
What benefits do organizations gain by navigating product architecture complexities?
Organizations that effectively manage the complexities of their offerings can accelerate their time to market while maintaining a solid technical roadmap, ultimately improving patient outcomes and satisfaction.
