Introduction
The journey from concept to market for medical devices presents significant complexity, where precision and compliance are essential. Understanding best practices for manufacturing prototypes not only streamlines this intricate process but also greatly increases the chances of successful product development. Given the numerous stages involved - from concept development to regulatory compliance - how can manufacturers ensure that their prototypes effectively meet user needs while adhering to stringent industry standards?
Understand the Prototype Manufacturing Process
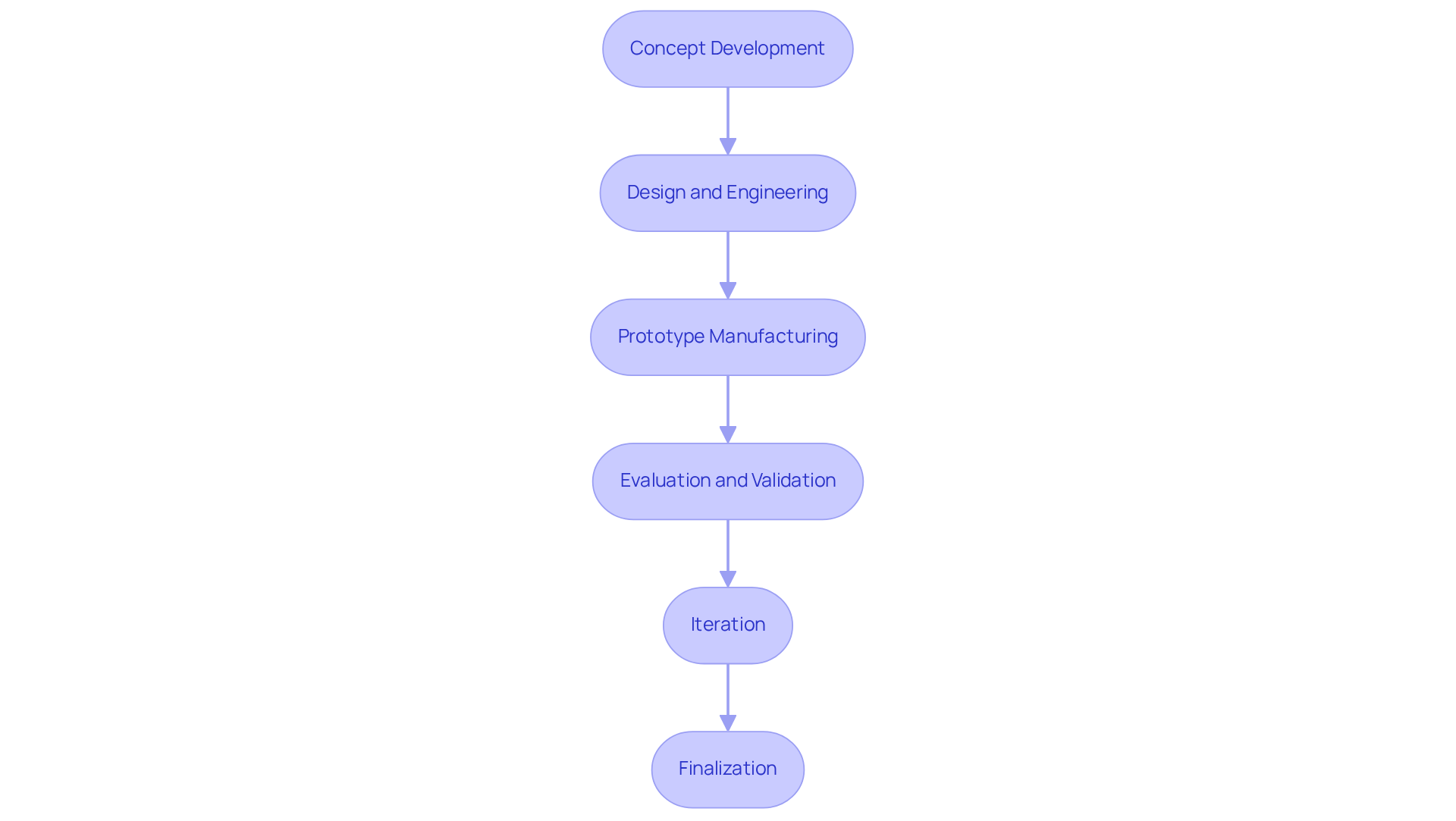
The process for consists of several essential stages:
- : This foundational phase focuses on defining the purpose of the device, identifying target users, and outlining essential features. Early engagement with stakeholders is crucial for refining the concept and ensuring alignment with market needs, as it lays the groundwork for successful development.
- : After establishing the concept, detailed design work begins. This includes creating CAD models and selecting appropriate materials. Collaboration with engineers is vital to ensure that the design is both feasible and manufacturable, addressing potential challenges early in the process.
- Prototype Manufacturing: The process involves manufacturing a prototype to test the design. This stage involves creating physical models of the product. Techniques such as , CNC machining, or injection molding are employed based on the complexity and specifications of the item. facilitates quick iterations, allowing teams to produce multiple design versions within days, which is essential for validating performance.
- : Prototypes undergo rigorous examination to assess functionality, safety, and compliance with regulatory standards. This phase is critical, as informs necessary design adjustments, ensuring that the device meets all required specifications.
- Iteration: Based on evaluation outcomes, models are refined through several iterations. This iterative process is crucial for optimizing the design and ensuring that the final product aligns with user needs and regulatory expectations. It is noteworthy that a significant percentage of medical device models fail during initial testing, highlighting the importance of thorough validation and iteration.
- Finalization: Once the model meets all requirements, it is prepared for production. This includes finalizing manufacturing processes and ensuring that all measures are in place, paving the way for a successful market launch.
By adhering to these best practices in medical equipment development, manufacturers can enhance their chances of success and deliver innovative solutions that address the needs of the healthcare sector.
Select Appropriate Materials and Manufacturing Methods
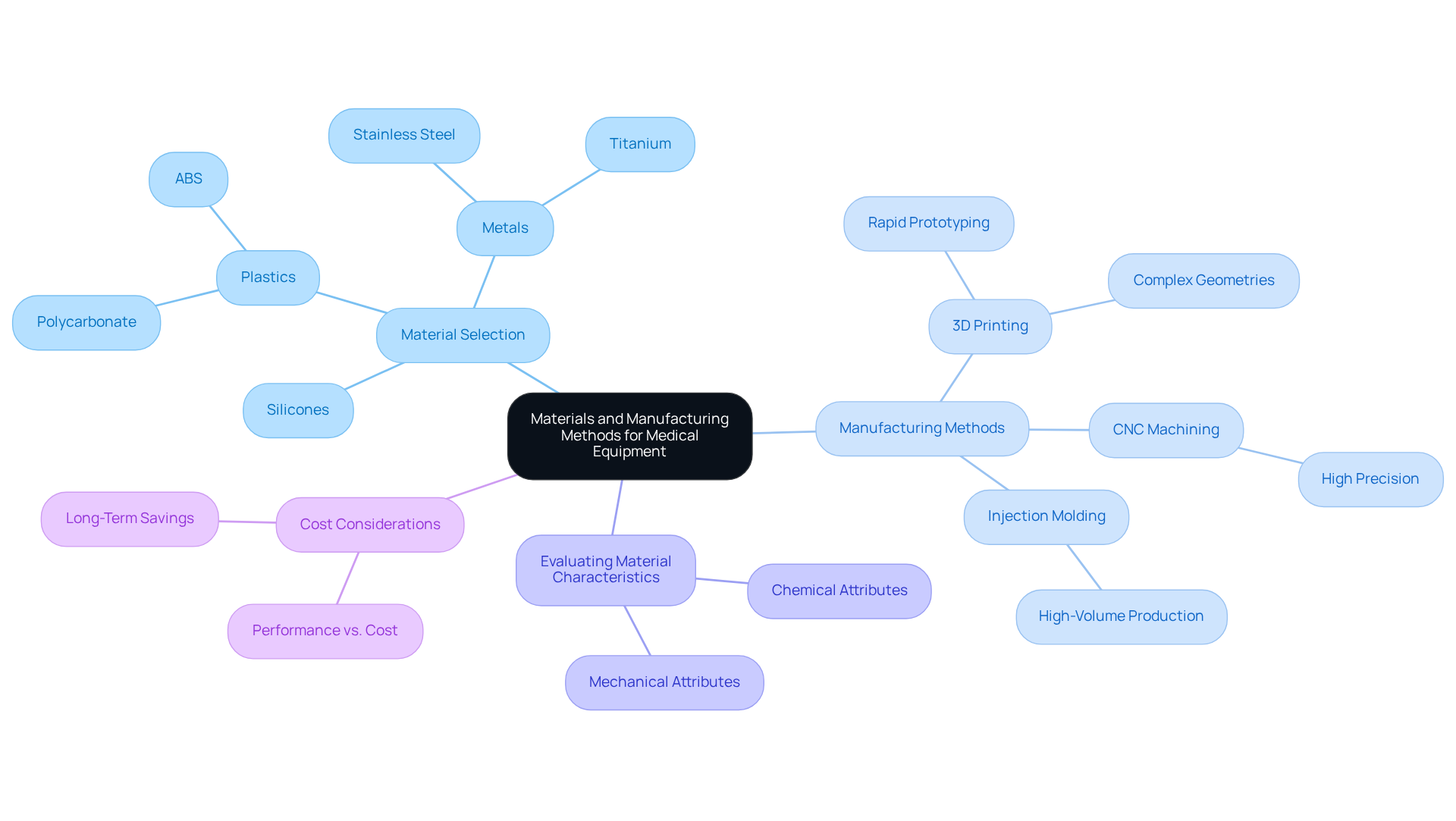
Choosing suitable materials and manufacturing techniques is essential for of successfully. Key best practices to consider include:
-
: It is crucial to prioritize when selecting materials. Commonly used options include:
- Plastics: Materials such as polycarbonate and ABS are favored for their lightweight and versatile properties.
- Metals: Stainless steel and titanium are preferred for their strength and biocompatibility, making them suitable for various medical applications.
- Silicones: These materials are ideal for flexible components and items requiring sterilization, ensuring both safety and functionality.
-
The chosen manufacturing method for manufacturing a prototype should align with the design and intended application of the prototype. Effective options include:
- : This method excels in rapid prototyping, allowing for the creation of complex geometries and significantly reducing development time.
- : Known for its high precision, CNC machining is particularly effective for producing metal components that require exact specifications.
- Injection Molding: Once the design is finalized, injection molding becomes suitable for high-volume production, ensuring consistency and efficiency.
-
Evaluating Material Characteristics: Comprehensive assessments are crucial to verify that the selected materials meet the required mechanical and chemical attributes essential for the equipment's performance and safety. This step is vital to prevent , which can lead to costly setbacks.
-
Cost Considerations: Striking a -effectiveness is critical. While premium materials may involve higher upfront expenses, they can ultimately reduce long-term costs by decreasing the likelihood of failures and recalls, which are particularly significant in the medical equipment sector.
Ensure Regulatory Compliance and Quality Control
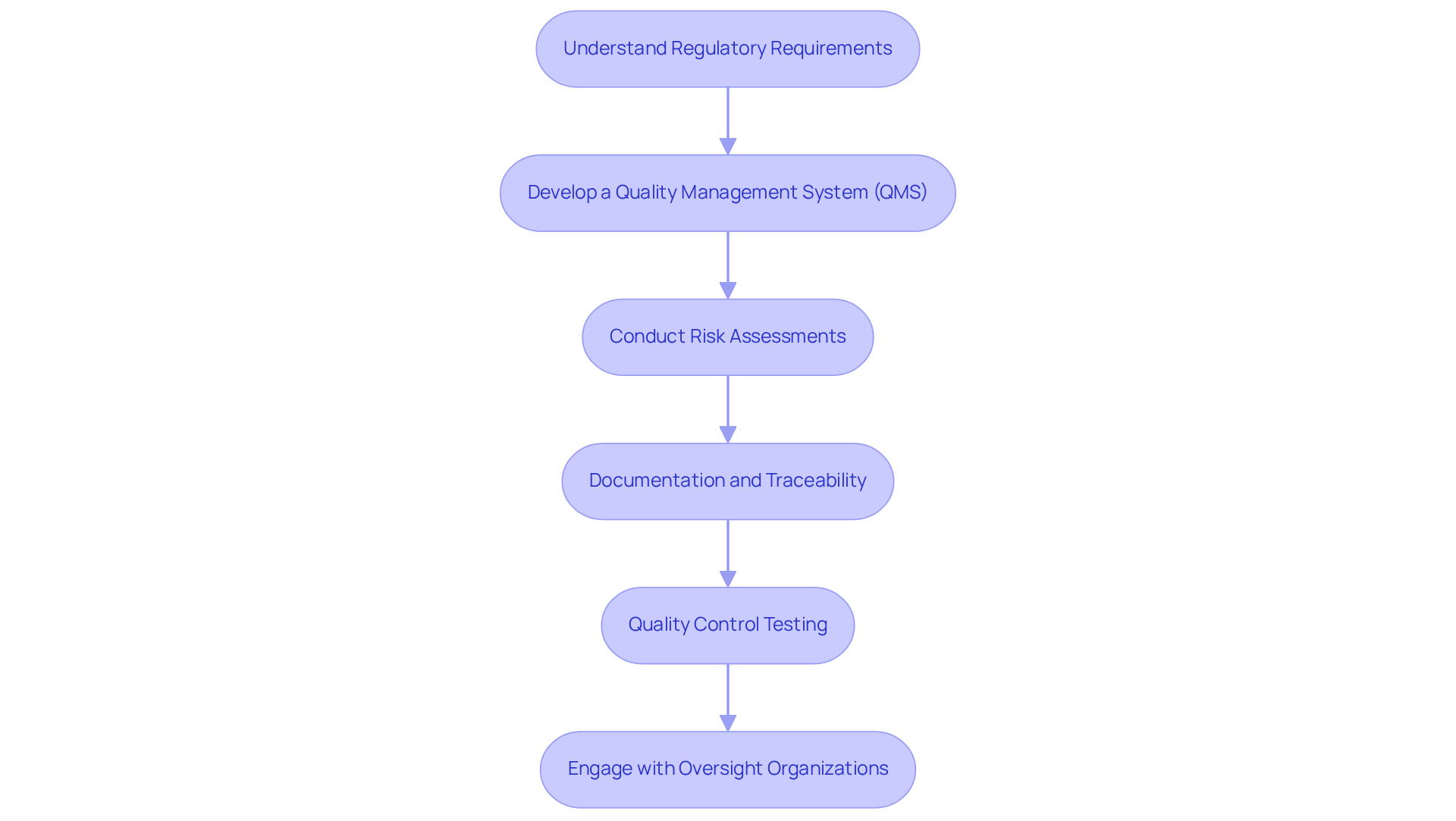
Regulatory compliance and quality control are crucial in of medical instruments. Adhering to established practices can significantly improve the likelihood of and market entry.
- Understand : It is essential to familiarize yourself with key regulations, including and . This foundational knowledge is vital for manufacturing a prototype that meets safety and efficacy standards, ultimately facilitating smoother approvals.
- Develop a (QMS): Implement a comprehensive QMS that outlines processes for design control, , and documentation. A well-structured QMS guarantees consistency and quality throughout the development lifecycle, which is critical for manufacturing a prototype and ensuring compliance.
- Conduct Risk Assessments: Proactively identify potential risks associated with the equipment and devise strategies to mitigate them. This approach not only enhances user safety but also aligns with compliance standards in the process of manufacturing a prototype, thereby reducing the likelihood of adherence issues.
- Documentation and Traceability: Maintain meticulous and testing processes, including design history files, device master records, and validation reports. This thorough record-keeping is essential for manufacturing a prototype, as it ensures compliance submissions and demonstrates adherence to quality standards.
- : Establish rigorous quality control measures to evaluate prototypes at various stages of development. This includes functional testing, biocompatibility assessments, and performance evaluations during the process of manufacturing a prototype to ensure that the apparatus meets all specifications and compliance requirements.
- Engage with Oversight Organizations: Consult with compliance experts or interact with oversight organizations early in the development process. This proactive engagement can clarify requirements and streamline the approval process, ultimately enhancing the chances of successful market entry.
Implementing these best practices not only improves the quality of medical products but also increases the percentage of items that achieve regulatory compliance on the first submission, which is essential in a highly regulated industry.
Implement Iterative Testing and Refinement Strategies
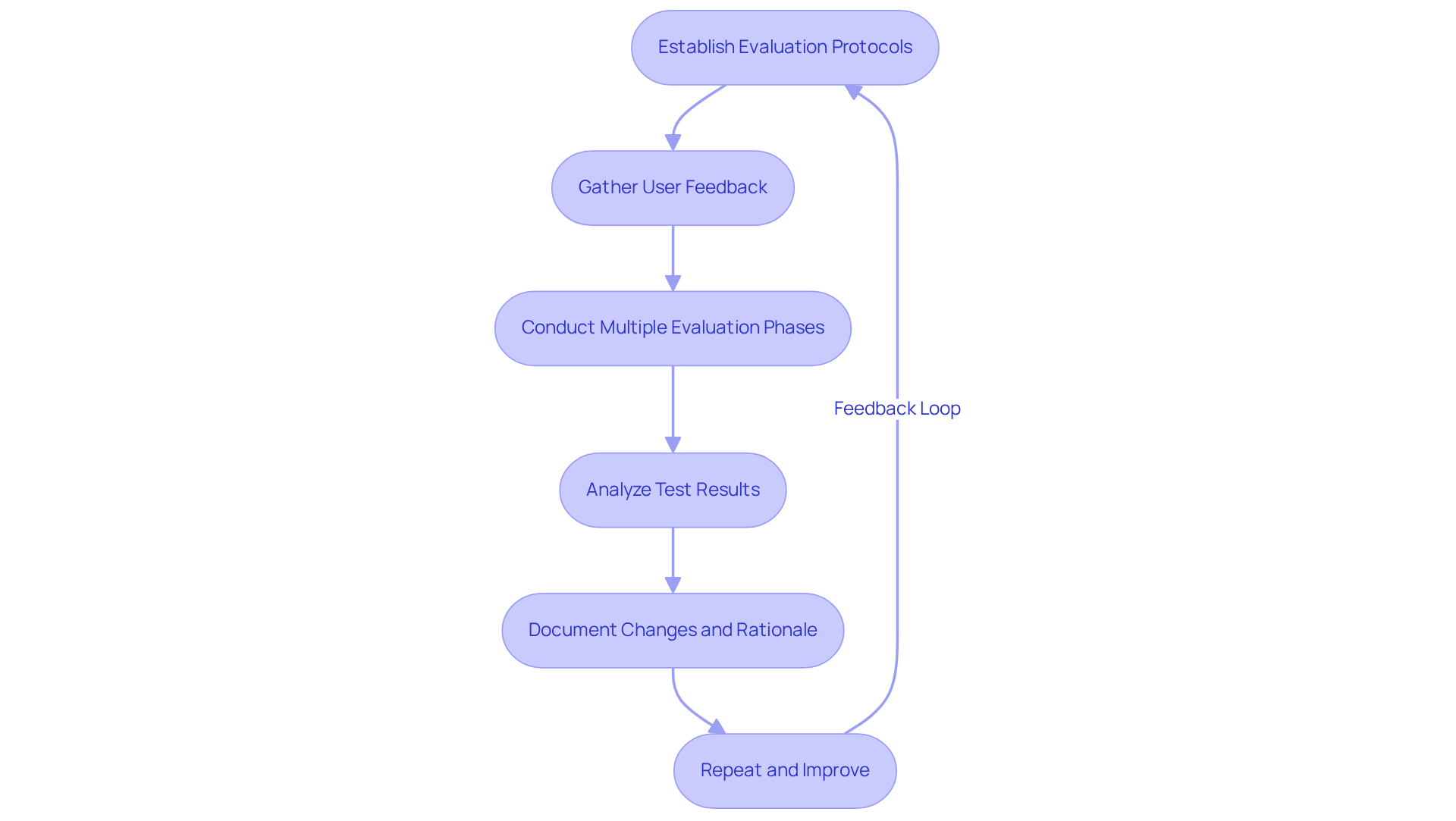
Iterative evaluation and refinement are essential for of effective . Here are :
- Establish : Define clear evaluation protocols that outline objectives, methodologies, and success criteria. This systematic approach ensures reliable results and facilitates compliance with regulatory standards.
- Gather : Involve end-users in the evaluation process to collect valuable insights on usability and functionality. User feedback is critical, as it can uncover that may not be apparent through technical evaluation alone. Engaging with users fosters collaboration and trust, resulting in better-designed products.
- Conduct Multiple Evaluation Phases: Implement a , beginning with basic functionality examinations and advancing to more complex evaluations. This strategy allows for early identification of issues, significantly reducing the risk of costly changes later in the development process. Typically, successful medical instrument development includes manufacturing a prototype that undergoes multiple evaluation stages to ensure comprehensive validation.
- Analyze Test Results: After each , analyze the results to identify trends, issues, and areas for improvement. This data-focused method guides design adjustments and emphasizes changes according to user needs and compliance requirements, enhancing the overall efficiency of the device.
- Document Changes and Rationale: Maintain made to the model, including the reasoning behind each modification. This documentation is vital for and serves as a reference for future development efforts.
- Repeat and Improve: Utilize insights gained from evaluation to continuously enhance the model. This process may involve multiple cycles of testing and modification during the manufacturing of a prototype until it meets all specifications and user expectations, ultimately leading to a more successful product launch.
Conclusion
The process of manufacturing prototypes in the medical device sector is complex and necessitates a strategic approach to ensure successful outcomes. By comprehending the various stages - from concept development to finalization - manufacturers can create devices that not only fulfill market demands but also comply with stringent regulatory standards.
Key insights include the significance of selecting suitable materials and manufacturing methods tailored to the device's intended use. Prioritizing biocompatibility and durability in material selection, along with effective manufacturing techniques such as 3D printing and CNC machining, can greatly influence the prototype's performance and safety. Moreover, implementing rigorous quality control measures and a robust quality management system ensures adherence to regulations, ultimately facilitating smoother market entry.
In light of these best practices, it is essential for manufacturers to embrace an iterative approach to testing and refinement. Engaging with end-users, collecting feedback, and conducting thorough evaluations at each development stage not only enhances the product's functionality but also promotes innovation within the healthcare sector. By prioritizing these strategies, manufacturers can improve their chances of success and contribute to the advancement of medical technology that better serves patients and healthcare providers alike.
Frequently Asked Questions
What are the main stages of the prototype manufacturing process for medical devices?
The main stages include Concept Development, Design and Engineering, Prototype Manufacturing, Evaluation and Validation, Iteration, and Finalization.
What is the purpose of the Concept Development phase?
The purpose of the Concept Development phase is to define the device's purpose, identify target users, and outline essential features, while engaging with stakeholders to refine the concept and align it with market needs.
What activities are involved in the Design and Engineering stage?
In the Design and Engineering stage, detailed design work is conducted, which includes creating CAD models and selecting appropriate materials, while collaborating with engineers to ensure feasibility and manufacturability.
How is a prototype manufactured in the Prototype Manufacturing stage?
A prototype is manufactured using techniques such as 3D printing, CNC machining, or injection molding, depending on the complexity and specifications of the item. Rapid prototyping allows for quick iterations to produce multiple design versions.
What happens during the Evaluation and Validation phase?
During the Evaluation and Validation phase, prototypes undergo rigorous examination to assess functionality, safety, and compliance with regulatory standards, with feedback informing necessary design adjustments.
Why is the Iteration stage important in the prototype manufacturing process?
The Iteration stage is important because it involves refining models based on evaluation outcomes, optimizing the design to ensure alignment with user needs and regulatory expectations, especially considering that many medical device models fail during initial testing.
What does the Finalization phase entail?
The Finalization phase involves preparing the model for production by finalizing manufacturing processes and ensuring all regulatory compliance measures are in place, which is essential for a successful market launch.
How can manufacturers enhance their chances of success in medical equipment development?
Manufacturers can enhance their chances of success by adhering to best practices throughout the prototype manufacturing process, ensuring they deliver innovative solutions that meet the needs of the healthcare sector.
List of Sources
- Understand the Prototype Manufacturing Process
- Future of 3D Printing for Medical Device OEMs (https://stratasys.com/en/resources/blog/3d-printing-medical-device-oem-trends)
- Micro-precision 3D printing: Trends and breakthroughs in medical device manufacturing (https://todaysmedicaldevelopments.com/news/micro-precision-3d-printing-trends-breakthroughs-medical-device-manufacturing)
- Scaling Medical Device Production: From Prototype to 100,000+ Units | Quasar Medical (https://quasarmedical.com/education/scaling-medical-device-production-prototype-to-volume)
- Medical Device Manufacturers Accelerate R&D and Speed to Market via Live Prototyping (https://prnewswire.com/news-releases/medical-device-manufacturers-accelerate-rd-and-speed-to-market-via-live-prototyping-302501846.html)
- The Role of Rapid Prototyping in Fast-Tracking New Medical Technologies (https://gcmiatl.org/the-role-of-rapid-prototyping-in-fast-tracking-new-medical-technologies)
- Select Appropriate Materials and Manufacturing Methods
- Engineering Reality Into Additive for Next Gen Medical Devices (https://designnews.com/3d-printing/engineering-reality-into-additive-manufacturing-for-next-gen-medical-devices)
- Medical Device Manufacturing News Archives - Med-Tech Insights (https://med-techinsights.com/category/news/medical-device-manufacturing-news)
- Medical Device Plastics Market 2026 Latest Innovations, Trends, Growth Factors and Opportunities by 2030 | Expert Review (https://barchart.com/story/news/376059/medical-device-plastics-market-2026-latest-innovations-trends-growth-factors-and-opportunities-by-2030-expert-review)
- New Medical Materials for Device Makers at MD&M West 2026 | Americhem (https://americhem.com/news/medical-materials-mdm-west-2026)
- Micro-precision 3D printing: Trends and breakthroughs in medical device manufacturing (https://todaysmedicaldevelopments.com/news/micro-precision-3d-printing-trends-breakthroughs-medical-device-manufacturing)
- Ensure Regulatory Compliance and Quality Control
- FDA Continues to Ease Regulatory Hurdles for Wearable Health Products | Insights | Squire Patton Boggs (https://squirepattonboggs.com/insights/publications/fda-continues-to-ease-regulatory-hurdles-for-wearable-health-products)
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- FDA’s 2026 Guidance on General Wellness Devices: Policy for Low-Risk Devices— Key Compliance and Regulatory Insights for Digital Health Companies - Kendall PC (https://kendallpc.com/fdas-2026-guidance-on-general-wellness-devices-policy-for-low-risk-devices-key-compliance-and-regulatory-insights-for-digital-health-companies)
- How Will FDA's New Rules Impact Your Health Gadgets? (https://spectrum.ieee.org/fda-medical-device-rules)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- Implement Iterative Testing and Refinement Strategies
- Medical Device Design Trends 2026 | Startups & OEMs | IDP (https://innovativedesignproducts.com/medical-device-design-trends-2026)
- MedTech Product Development Trends in 2026 (https://enlil.com/blog/medtech-product-development-trends-in-2026)
- The Role of Rapid Prototyping in Medical Device Development (https://photofabrication.com/the-role-of-rapid-prototyping-in-medical-device-development)
- Importance Of Customer Feedback In Developing Reliable Medical Devices - Manufacturers Of Ophthalmic Devices And Fluorescein Strips | Madhu Instruments (https://madhuinstruments.com/importance-of-customer-feedback-in-developing-reliable-medical-devices)
- Medical Device Testing in Transition (https://mpo-mag.com/medical-device-testing-in-transition)





