Introduction
Achieving compliance with ISO 62304 represents a critical milestone for developers of medical device software, as it guarantees the safety and effectiveness of their applications. This international standard delineates the lifecycle processes essential for regulatory adherence and enhances the overall quality and reliability of software products.
However, the path to compliance is often fraught with challenges, particularly as the healthcare technology landscape evolves with the integration of AI and machine learning.
How can developers effectively navigate these complexities to ensure their software meets both market demands and stringent safety regulations?
Understand IEC 62304 and Its Importance in Medical Device Software
The international standard that governs the lifecycle processes for is . It outlines and maintenance to ensure both safety and effectiveness. For developers, a is crucial, as it not only facilitates adherence to regulatory requirements but also significantly enhances quality and reliability.
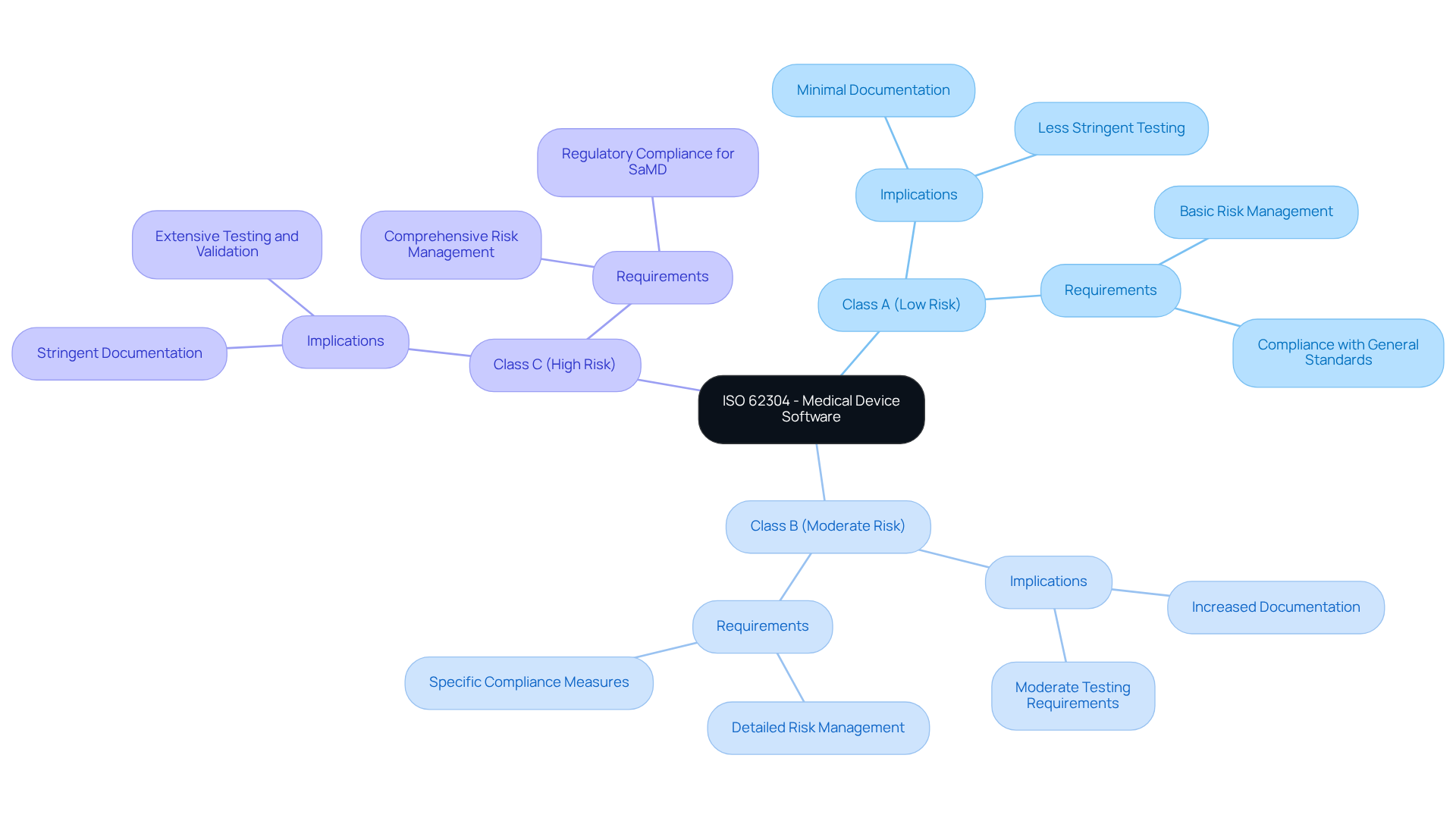
The standard categorizes applications into three safety groups:
- Class A (low risk)
- Class B (moderate risk)
- Class C (high risk)
Among these, Class C necessitates the most stringent controls. Grasping these classifications is fundamental for achieving compliance and .
As healthcare technology continues to evolve, particularly with the integration of AI and machine learning, compliance with becomes increasingly vital. This compliance ensures that applications . medical device software have demonstrated its effectiveness in mitigating risks and improving reliability, thereby reinforcing its status as a cornerstone of medical device development.
Identify Key Requirements for IEC 62304 Compliance
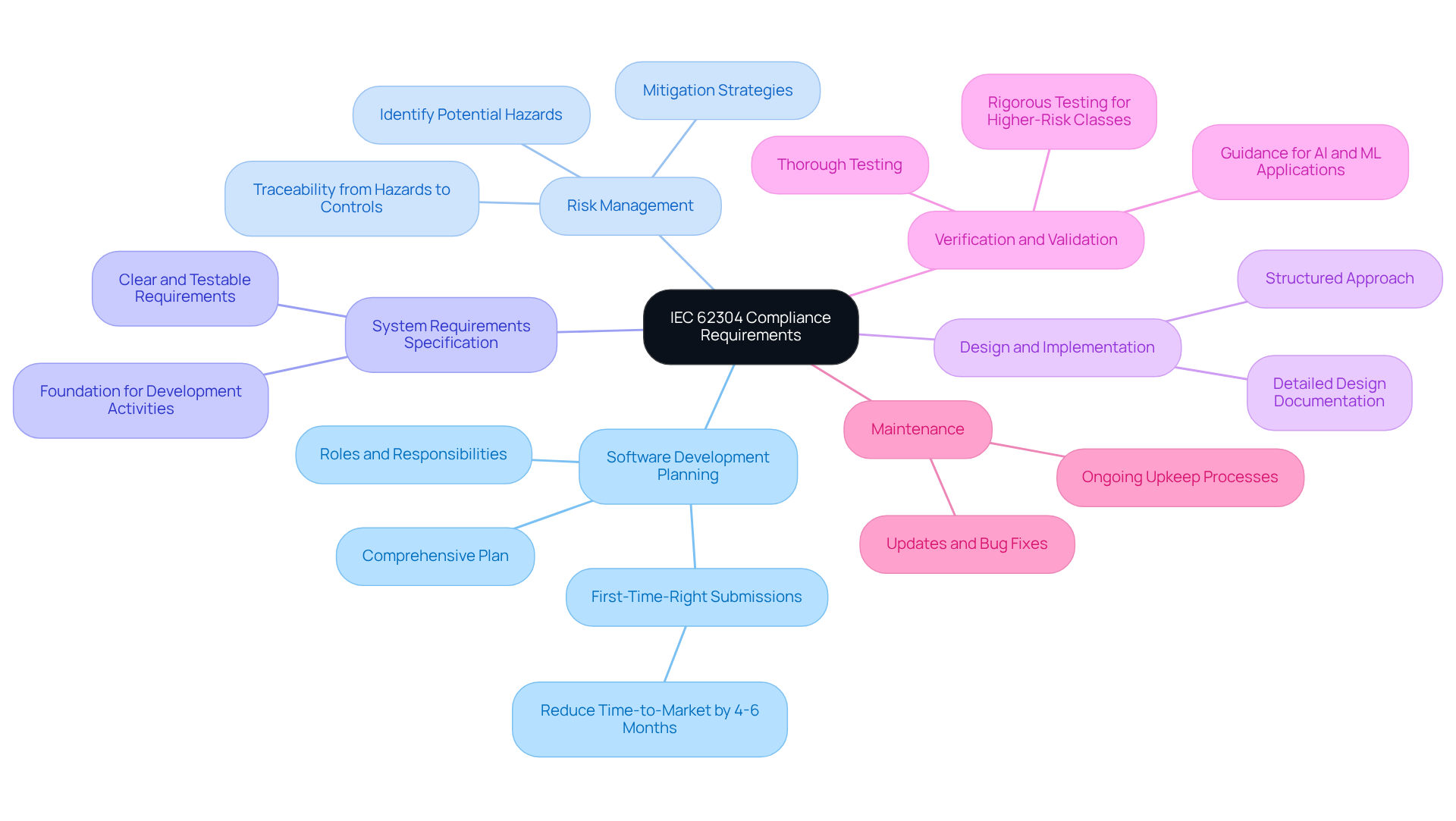
requires a focus on several essential requirements:
- Software Development Planning: A comprehensive plan must be established, detailing the software lifecycle, roles, and responsibilities. Organizations that implement IEC 62304 early in their development processes often achieve first-time-right submissions, reducing time-to-market by an average of 4 to 6 months. This proactive planning is crucial for effective user manual development, where provides invaluable documentation compliance support.
- : A robust process is essential. This involves identifying potential hazards associated with the software and implementing effective mitigation strategies. by emphasizing software-specific risks, focusing on consequences rather than just the probability of harm. Maintaining traceability from hazards to controls is critical for compliance, an area where can guide startups through the complexities of regulatory requirements.
- (SRS): Clear and testable requirements must be documented to define the application's objectives. This specification serves as a foundation for subsequent development activities and is essential for creating that accurately represent application capabilities.
- Design and Implementation: A structured approach to design and execution is vital to ensure that the system meets the specified requirements. This includes detailed design documentation that breaks down system components into manageable units for implementation and testing, which is essential for developing comprehensive .
- : is essential to confirm that the system meets its requirements and validate its intended use. For higher-risk application classes, such as Class B and C, rigorous testing and documentation are mandated to ensure safety and effectiveness. The standard now includes specific guidance for AI and machine learning applications, requiring rigorous testing, validation, and risk assessment to ensure safe, reliable performance in clinical settings. supports startups in navigating these rigorous requirements.
- Maintenance: Establishing processes for ongoing upkeep throughout the system lifecycle is critical. This includes planning for updates and bug fixes to ensure ongoing adherence and performance, which is vital for maintaining accurate and up-to-date .
By focusing on these requirements, developers can establish a strong basis for IEC standards adherence, ensuring that their medical device software is safe, effective, and aligned with regulatory expectations. It is also important to stay informed about the , to ensure that all processes align with the latest regulatory expectations.
Implement Effective Processes for Compliance
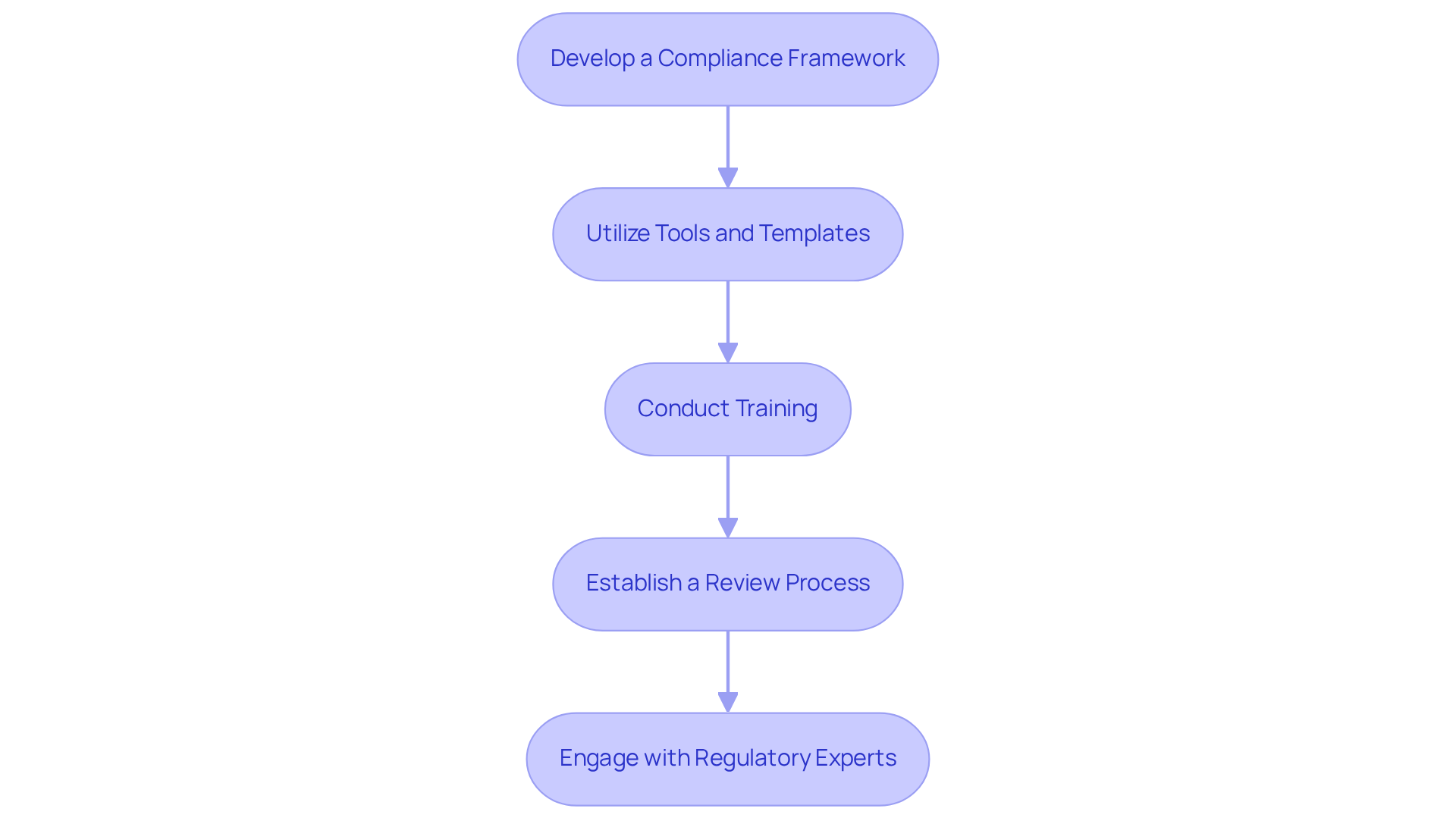
To implement effective processes for , the development of ISO 62304 is crucial for ensuring compliance and safety in healthcare applications. Consider the following steps:
- Develop a : Establish a framework that integrates IEC requirements into your existing software development processes. This framework should encompass documentation practices, , and testing protocols, ensuring a thorough approach to adherence. It is essential to record and follow each of the five fundamental processes described in IEC to satisfy regulatory requirements.
- Utilize Tools and Templates: Leverage and templates designed for . These resources can streamline documentation efforts and ensure that all necessary elements are thoroughly addressed, thereby reducing the risk of oversight. Incorporating statistics or examples of effective tools used in the industry can further enhance adherence efforts.
- Conduct Training: Provide targeted training for your development team on IEC standards and best practices. This training should encompass all roles involved in the development process, including developers, product managers, QA testers, and designers, ensuring that everyone understands their responsibilities in achieving adherence and fostering a culture of quality and accountability.
- Establish a Review Process: Implement a for the development lifecycle to ensure adherence to . Regular peer evaluations, audits, and adherence checks can help identify potential gaps and strengthen compliance throughout the development phases. Additionally, incorporate continuous monitoring, bug tracking, and user feedback post-deployment to identify new risks in medical software.
- Engage with Regulatory Experts: Collaborate with regulatory specialists or consultants who can provide valuable guidance and insights into upholding standards throughout the development lifecycle. Their expertise can assist in navigating complex regulatory environments and ensure that your processes conform to current standards, including the integration of established frameworks such as .
By adhering to these steps, organizations can establish a robust compliance process for ISO 62304 that not only meets IEC standards but also enhances the overall .
Maintain Documentation and Traceability for Compliance
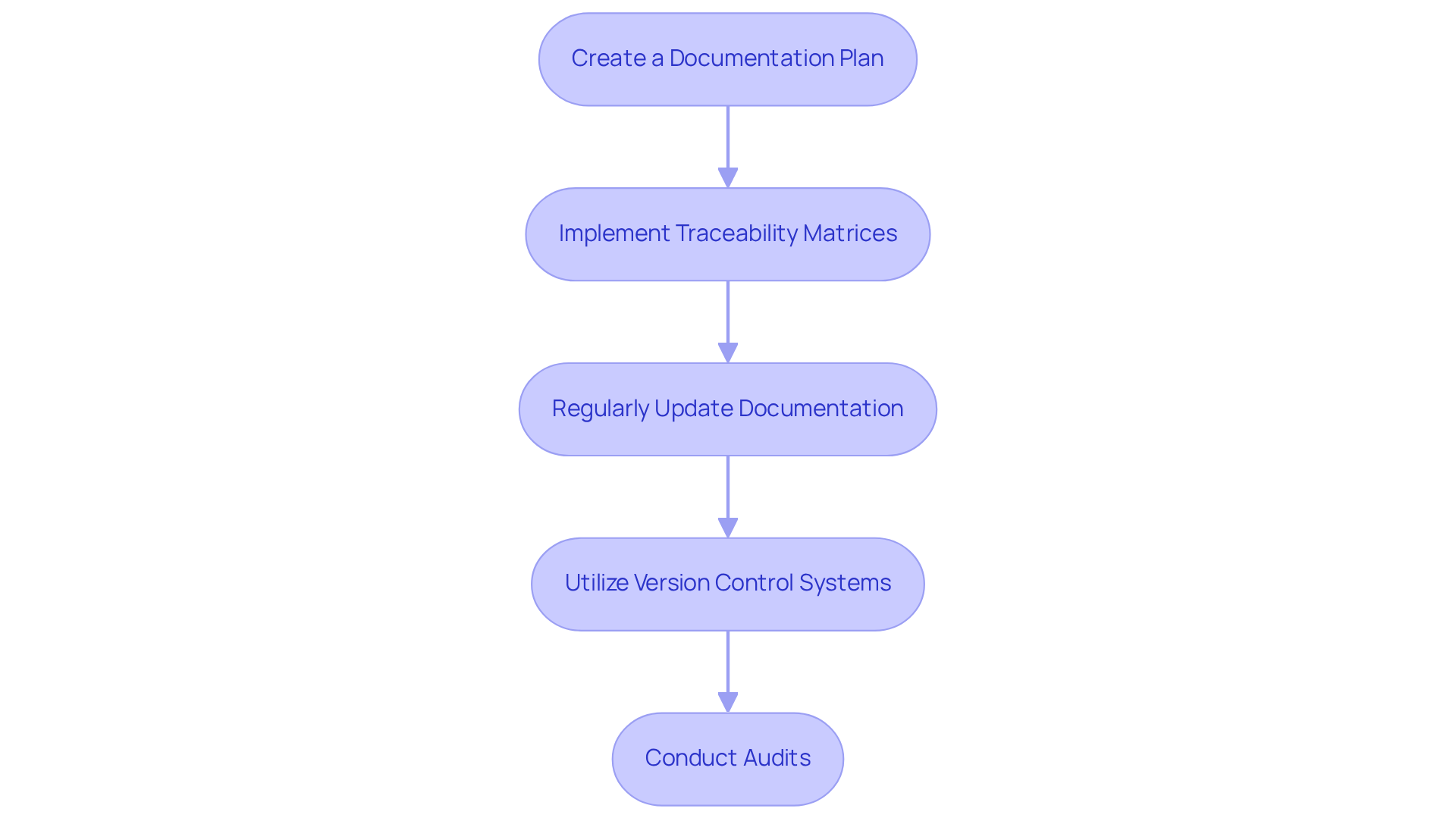
Effective are essential components for compliance with . The following key practices should be adhered to:
- Create a : Develop a comprehensive plan that specifies the required documentation at each phase of the system lifecycle. This plan should encompass Software Requirements Specifications (SRS), design documents, test plans, and maintenance records.
- Implement : Utilize to establish connections between requirements, design, implementation, and testing. This practice ensures that all requirements are adequately addressed and provides a clear audit trail.
- : Ensure that documentation remains current by conducting frequent reviews and revisions as the application evolves. This includes documenting changes, updates, and any deviations from the original plan.
- Utilize : Employ to manage modifications to documentation and code. This approach helps maintain a historical record of changes and guarantees that all team members are working with the most recent versions.
- : Schedule regular evaluations of methods to ensure compliance with IEC standards. These audits can help identify gaps and areas for improvement.
By maintaining thorough in , organizations can demonstrate their commitment to compliance and enhance the reliability of their .
Ensure Continuous Improvement and Stay Updated with IEC 62304
To ensure and remain compliant with , organizations should consider the following strategies:
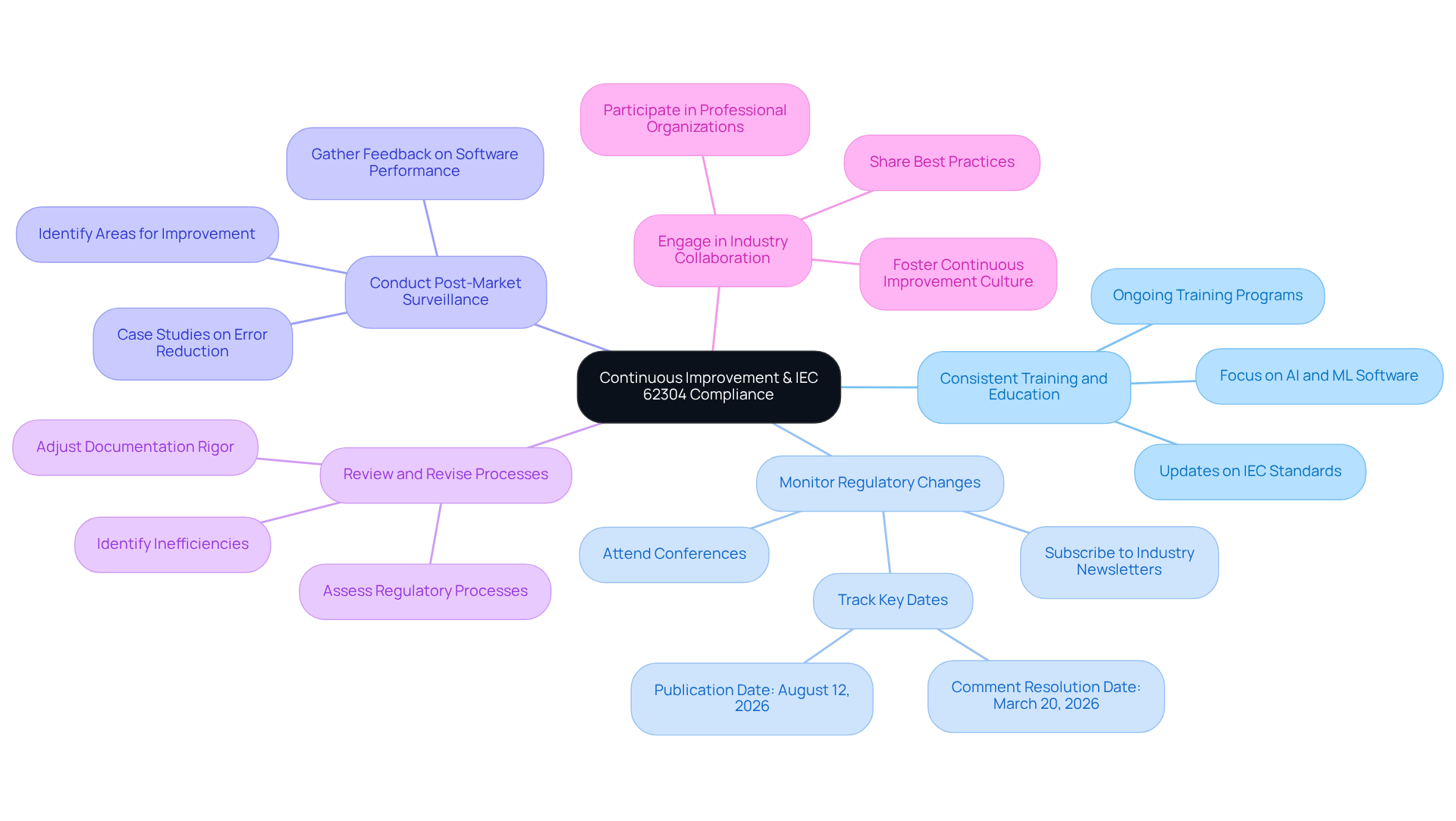
- Consistent Training and Education: Investing in ongoing training for your team is essential to keep them informed about revisions to IEC standards and optimal application development methods. The updated standard underscores the importance of on new requirements, particularly concerning AI and machine learning software.
- : It is crucial to stay updated on changes to ISO 62304 and related regulations. Subscribing to industry newsletters, attending conferences, and participating in professional organizations can facilitate this. Key dates for adherence preparation, such as the comment resolution starting March 20, 2026, should be closely monitored to ensure alignment with forthcoming updates.
- : Implementing post-market surveillance practices allows organizations to gather and identify areas for improvement. This proactive approach can inform future development cycles and adherence efforts, as evidenced by case studies where manufacturers achieved significant reductions in record errors through continuous monitoring.
- Review and Revise Processes: Regular assessments of and documentation are necessary to identify inefficiencies or gaps. Making the required adjustments enhances effectiveness and ensures alignment with current standards, especially as the new Process Rigor Levels will necessitate varying documentation rigor based on classification.
- : Collaborating with other organizations and industry groups to share insights and best practices related to adherence fosters a culture of and innovation. This collaboration is vital for adapting to evolving regulatory requirements and maintaining high standards of product safety.
By adopting these strategies, organizations can maintain a proactive approach to compliance with ISO 62304 , ensuring that their software remains safe and effective.
Conclusion
Achieving compliance with ISO 62304 is crucial for the development of medical device software, as it ensures that products are safe, effective, and meet regulatory standards. This guide delineates the necessary steps and best practices for navigating the complexities of IEC 62304 compliance, highlighting the importance of thorough planning, risk management, and continuous improvement.
Key insights include:
- The classification of software applications into safety groups
- The necessity of a robust documentation process
- The emphasis on training and collaboration
By adhering to these requirements and maintaining traceability throughout the development lifecycle, organizations can significantly enhance the quality and reliability of their medical devices. Furthermore, staying informed about upcoming regulatory changes is essential for ensuring ongoing compliance and adapting to new industry standards.
In a rapidly evolving healthcare landscape, the significance of ISO 62304 compliance is paramount. Organizations are encouraged to adopt a proactive approach, engaging in continuous education and industry collaboration to foster innovation and uphold safety standards. By prioritizing these strategies, developers can not only achieve compliance but also contribute to the advancement of safe and effective medical technologies that ultimately benefit patient care.
Frequently Asked Questions
What is IEC 62304 and why is it important in medical device software?
IEC 62304 is an international standard that governs the lifecycle processes for medical device applications. It outlines essential requirements for development and maintenance to ensure safety and effectiveness, which is crucial for developers to adhere to regulatory requirements and enhance quality and reliability.
How are medical device applications categorized under IEC 62304?
Medical device applications are categorized into three safety groups: Class A (low risk), Class B (moderate risk), and Class C (high risk). Class C requires the most stringent controls to ensure compliance and safeguard patient health.
Why is compliance with IEC standards becoming increasingly vital?
As healthcare technology evolves, particularly with AI and machine learning integration, compliance with IEC standards ensures that applications meet market demands and safety regulations, thereby mitigating risks and improving reliability.
What are the key requirements for achieving compliance with IEC 62304?
Key requirements include: 1. Software Development Planning 2. Risk Management 3. System Requirements Specification (SRS) 4. Design and Implementation 5. Verification and Validation 6. Maintenance
What role does Software Development Planning play in IEC 62304 compliance?
A comprehensive software development plan outlines the software lifecycle, roles, and responsibilities. Early implementation of IEC 62304 can lead to first-time-right submissions, reducing time-to-market by an average of 4 to 6 months.
How does Risk Management relate to IEC 62304?
A robust risk management process is essential for identifying potential hazards associated with the software and implementing effective mitigation strategies. IEC 62304 emphasizes software-specific risks and requires maintaining traceability from hazards to controls.
What is the importance of System Requirements Specification (SRS) in IEC 62304?
The SRS documents clear and testable requirements that define the application's objectives, serving as a foundation for development activities and ensuring user manuals accurately represent application capabilities.
What is involved in the Design and Implementation phase for IEC 62304 compliance?
A structured approach to design and execution is vital, including detailed design documentation that breaks down system components for implementation and testing, which is essential for developing comprehensive user manuals.
What is the significance of Verification and Validation in IEC 62304?
Thorough testing is necessary to confirm that the system meets its requirements and validates its intended use. Higher-risk application classes, like Class B and C, require rigorous testing and documentation to ensure safety and effectiveness.
What should developers keep in mind regarding Maintenance for IEC 62304 compliance?
Developers must establish processes for ongoing upkeep throughout the system lifecycle, including planning for updates and bug fixes to maintain adherence and performance, which is crucial for keeping user manuals accurate and up-to-date.
What upcoming changes should developers be aware of regarding IEC standards?
Developers should stay informed about upcoming updates to IEC standards, which are set to begin on 12 August 2026, to ensure that all processes align with the latest regulatory expectations.
List of Sources
- Understand IEC 62304 and Its Importance in Medical Device Software
- lfhregulatory.co.uk (https://lfhregulatory.co.uk/iec-62304-update-2026)
- What is IEC 62304 and why is it important in SaMD Development? (https://htdhealth.com/insights/iec-62304-and-samd-development)
- Probability of software defects: What does IEC 62304 require? (https://blog.johner-institute.com/iec-62304-medical-software/probability-of-software-defects)
- How IEC 62304 Sparked a New Era in Medical Device Software Development (https://linkedin.com/pulse/how-iec-62304-sparked-new-era-medical-device-software-parchetalab-c6jnc)
- intuitionlabs.ai (https://intuitionlabs.ai/articles/iec-62304-medical-device-software-guide)
- Identify Key Requirements for IEC 62304 Compliance
- IEC 62304 Guide: Safe Medical Device Software Development (https://freyrsolutions.com/blog/iec-62304-compliance-the-complete-guide-to-medical-device-software-development-without-the-pitfalls)
- lfhregulatory.co.uk (https://lfhregulatory.co.uk/iec-62304-update-2026)
- Developing IEC 62304 Compliant Software: Proven Tips & Best Practices (https://greenlight.guru/webinar/developing-iec-62304-compliant-software-proven-tips-best-practices)
- ventionteams.com (https://ventionteams.com/blog/medical-device-software-statistics)
- Navigating IEC 62304 Standard for Medical Software (https://cleio.com/insights/blog/navigating-iec-62304-standard-for-medical-software)
- Implement Effective Processes for Compliance
- Ensuring Regulatory Compliance in Custom MedTech Software • The Havok Journal | Military & Veteran News, Commentary, Culture (https://havokjournal.com/fitness/medical/ensuring-regulatory-compliance-in-custom-medtech-software)
- IEC 62304 Compliance Guide: Medical Device Software (https://morulaa.com/iec-62304-medical-device-software-lifecycle-guide)
- lfhregulatory.co.uk (https://lfhregulatory.co.uk/iec-62304-update-2026)
- productcreationstudio.com (https://productcreationstudio.com/blog/building-the-foundation-right-iec-62304-for-medical-device-firmware-development)
- Healthcare Compliance Trends & Statistics (https://ispartnersllc.com/blog/healthcare-compliance-trends)
- Maintain Documentation and Traceability for Compliance
- securitycompass.com (https://securitycompass.com/blog/iec-62304-medical-software-lifecycle)
- intuitionlabs.ai (https://intuitionlabs.ai/articles/iec-62304-medical-device-software-guide)
- ketryx.com (https://ketryx.com/blog/a-comprehensive-guide-to-iec-62304-navigating-the-standard-for-medical-device-software)
- IMDRF Releases Key Guidance Documents for Medical Device Software (https://emergobyul.com/news/imdrf-releases-key-guidance-documents-medical-device-software)
- Ensure Continuous Improvement and Stay Updated with IEC 62304
- Medical Device Manufacturer's Continuous Improvement Approach Reduces Errors in Records | ASQ (https://asq.org/quality-resources/articles/case-studies/medical-device-manufacturers-continuous-improvement-approach-reduces-errors-in-records?id=6f48b3eeb7354aed84acd5f749b1d67d&srsltid=AfmBOopHNnM_jPZC5hUuh-3cUJUNJ2h39rtH-Q6oORbQSM-It4Gba5-l)
- intuitionlabs.ai (https://intuitionlabs.ai/articles/iec-62304-medical-device-software-guide)
- lfhregulatory.co.uk (https://lfhregulatory.co.uk/iec-62304-update-2026)
- cloudtheapp.com (https://cloudtheapp.com/best-practices-for-continuous-improvement-in-medical-device-quality)






