4 Essential Features of Product Development for Medical Devices
Introduction Understanding the complex landscape of medical device development...

The medical device industry is at a critical juncture, with projections indicating that the healthcare equipment market will exceed $623 billion by 2026. In this context, the importance of a well-structured new product introduction (NPI) process becomes increasingly evident. This article explores best practices that not only facilitate the transition from concept to market-ready product but also address the regulatory complexities that frequently impede success.
How can organizations effectively navigate these challenges to ensure their products meet compliance standards while also aligning with market needs?
The (NPI) process is crucial in the , facilitating the transition from concept to market-ready product. This process not only fosters innovation but also ensures , which are vital for patient safety and market acceptance. A well-executed new product introduction process can significantly shorten time to market, enhance , and boost overall customer satisfaction. Companies that prioritize new product introduction frequently report a higher because they are better prepared to navigate the complexities of regulatory requirements and market demands.
In 2026, the is projected to exceed $623 billion, underscoring the importance of in capitalizing on market opportunities. Furthermore, the integration of advanced biomaterials and AI technologies into workflows for new product introduction is expected to enhance product performance and , making it imperative for manufacturers to adopt . As the landscape evolves, the focus on real-world evidence creation and lifecycle management will further solidify the role of NPI in ensuring the safety and efficacy of healthcare products.
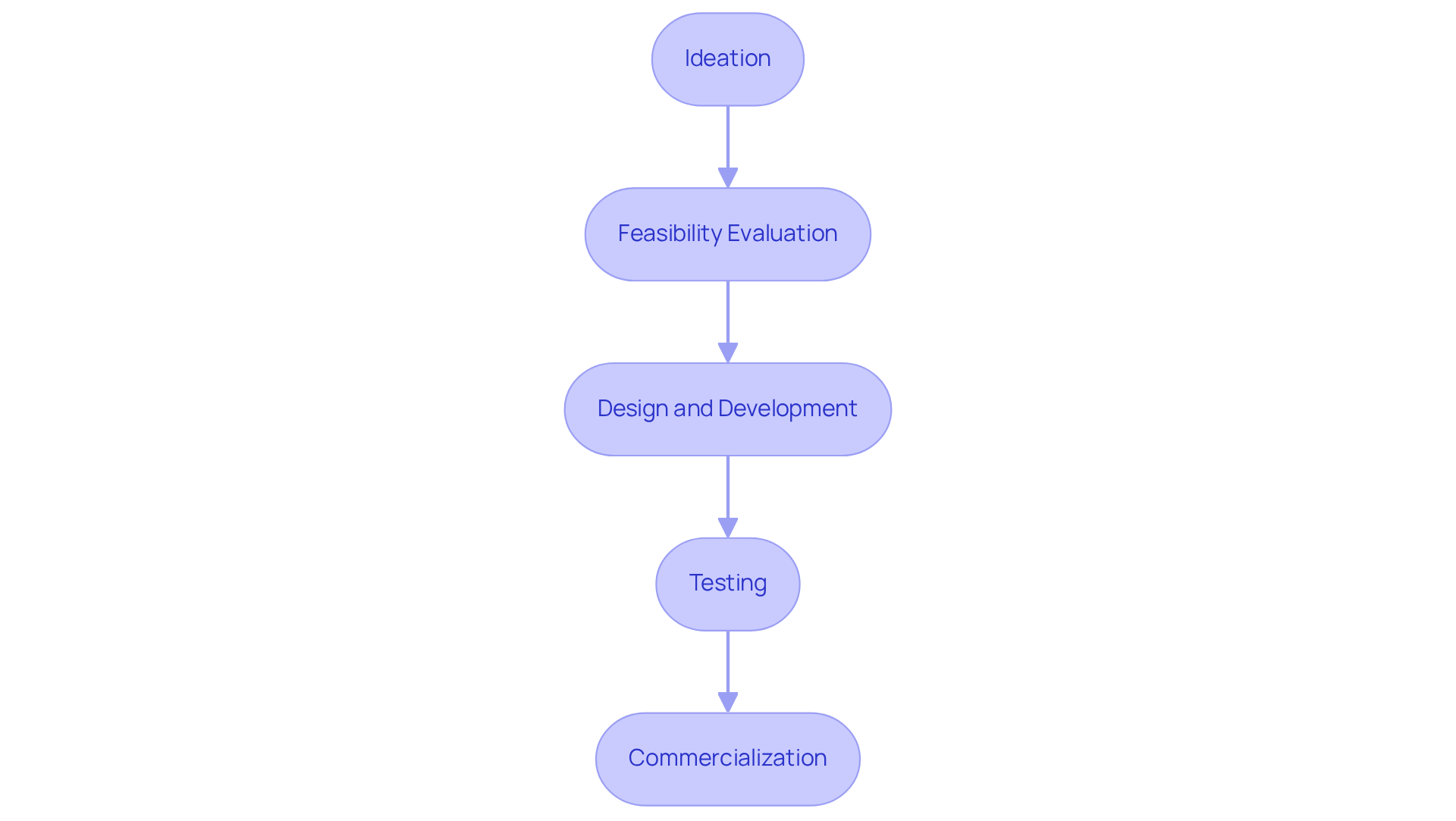
An organized is essential for the effective advancement of . This process encompasses several key phases:
Each stage must be meticulously planned and executed to ensure compliance with and alignment with market demands.
During the ideation phase, comprehensive is vital for identifying unmet needs and validating product concepts. Following this, a assesses both technical and financial viability, confirming that the proposed solution is practical and sustainable. Successful healthcare equipment teams have demonstrated that adhering to a structured new product introduction process significantly enhances their chances of achieving prompt and efficient .
Organizations that implement disciplined execution across these stages often experience higher success rates, as they are better equipped to navigate the complexities of regulatory compliance and market integration. provides essential , supporting startups in the healthcare equipment sector to effectively manage these regulatory challenges. By emphasizing a systematic approach and leveraging [Voler Systems](https://volersystems.com)' expertise, companies can adeptly address the challenges of healthcare product development and position themselves for sustained success.
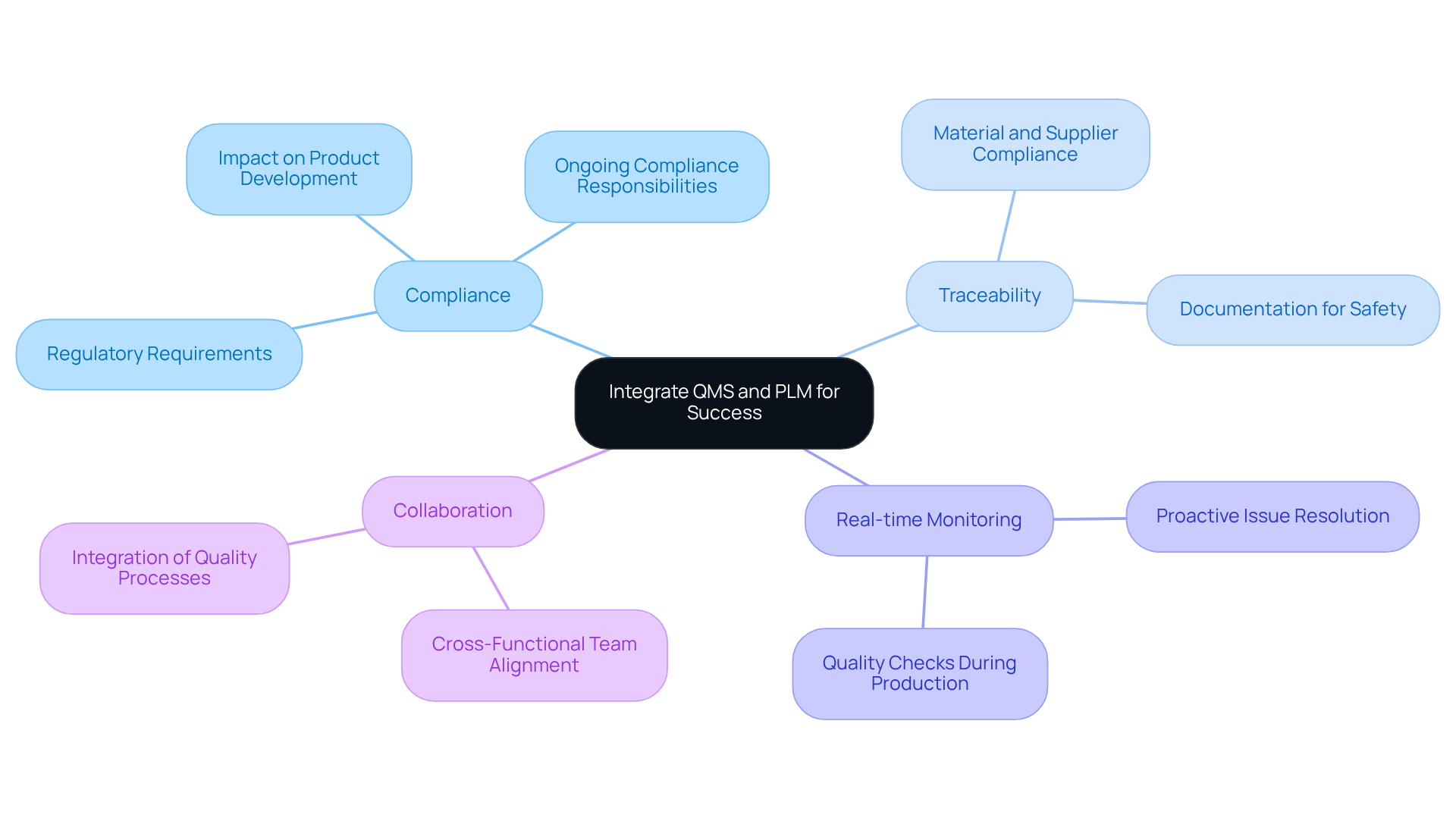
Integrating with is essential for ensuring that are developed and manufactured to the . A robust QMS enables organizations to maintain , while PLM offers a comprehensive view of the product's lifecycle, from conception to post-market surveillance. By aligning these two systems, companies can enhance traceability, streamline operations, and mitigate the risk of non-compliance.
Implementing a QMS integrated with PLM facilitates real-time monitoring of , allowing teams to proactively address issues and ensure that the final product meets all . Furthermore, incorporating efficient is crucial for verifying the reliability and functionality of electronic items, particularly in the healthcare and wearable sectors. This approach not only but also fosters collaboration among teams, ensuring that all aspects of adhere to established best practices in engineering design.
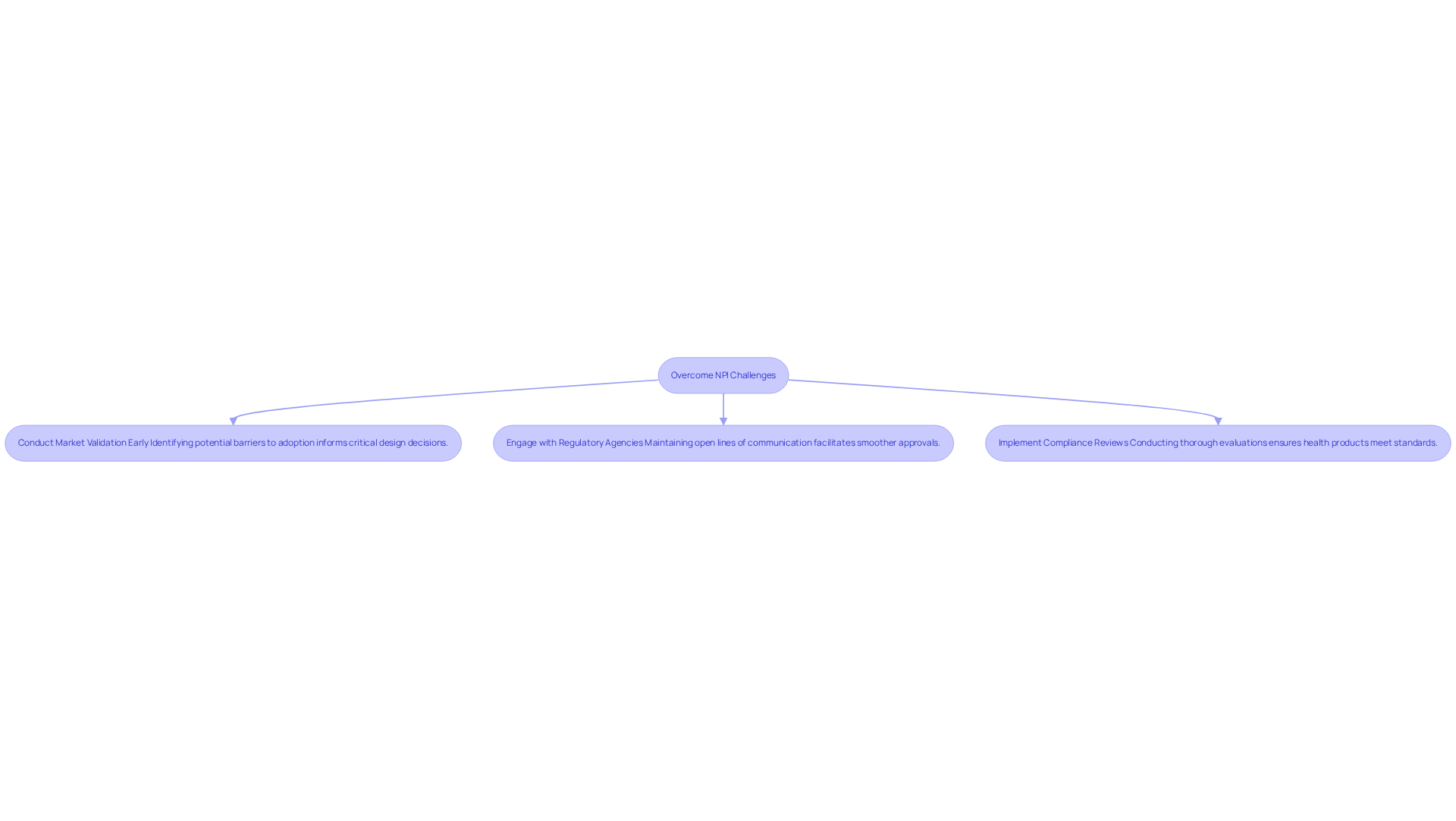
The (NPI) process for healthcare products presents several challenges, notably , , and resource limitations. Approximately 15% of data breaches in the healthcare equipment sector are linked to external vendors, underscoring the critical role of . To effectively navigate these challenges, organizations should adopt a that encompasses comprehensive market research, early engagement with regulatory bodies, and the establishment of .
Key Steps to Overcome NPI Challenges:
By anticipating and addressing these challenges, companies can significantly enhance their new product introduction efforts, which leads to improved success rates in bringing innovative medical devices to market. Client testimonials highlight the effectiveness of Voler Systems' , demonstrating how early engagement and thorough validation have contributed to successful product launches.
The new product introduction (NPI) process is a crucial element in the medical device industry, acting as the link between innovative ideas and market-ready products. By effectively managing this process, organizations can ensure compliance with stringent regulatory standards while also enhancing product quality and customer satisfaction. A structured NPI framework is essential for navigating market complexities and achieving successful product launches.
This article has highlighted key strategies for mastering NPI in medical devices. Understanding the significance of a well-defined introduction process, integrating quality management, and managing the product lifecycle are all critical in overcoming common challenges. Through thorough market research, engagement with regulatory agencies, and implementation of compliance reviews, companies can significantly boost their chances of success in a competitive landscape.
The importance of effective new product introduction in the medical device sector cannot be overstated. As the healthcare market evolves, adopting best practices in NPI will drive innovation and ensure that products meet the highest standards of safety and efficacy. Organizations are encouraged to take a proactive approach to NPI, utilizing structured processes and collaboration to pave the way for successful and impactful healthcare solutions.
What is the new product introduction (NPI) process in the medical device industry?
The NPI process is crucial for transitioning a medical device from concept to market-ready product, fostering innovation and ensuring compliance with regulatory standards for patient safety and market acceptance.
Why is the NPI process important for medical device companies?
A well-executed NPI process can shorten time to market, enhance product quality, and boost overall customer satisfaction, leading to a higher success rate in product launches.
What market trends highlight the importance of effective NPI strategies?
The healthcare equipment market is projected to exceed $623 billion by 2026, emphasizing the need for effective NPI strategies to capitalize on market opportunities.
How do advanced technologies impact the NPI process?
The integration of advanced biomaterials and AI technologies into NPI workflows is expected to enhance product performance and patient outcomes, making robust NPI frameworks imperative for manufacturers.
What future trends are expected to influence the NPI process in healthcare?
The focus on real-world evidence creation and lifecycle management will further solidify the role of NPI in ensuring the safety and efficacy of healthcare products as the landscape evolves.
