Introduction
The landscape of medical device development is evolving rapidly, influenced by the rising prevalence of chronic diseases and the demand for innovative solutions. Navigating the complex phases of this process - from concept validation to post-market surveillance - necessitates a strategic approach that emphasizes compliance and quality. Manufacturers can enhance their success in this competitive arena by adopting best practices that ensure adherence to stringent regulations while fostering effective collaboration among development teams.
Outline the Key Stages of Medical Device Development
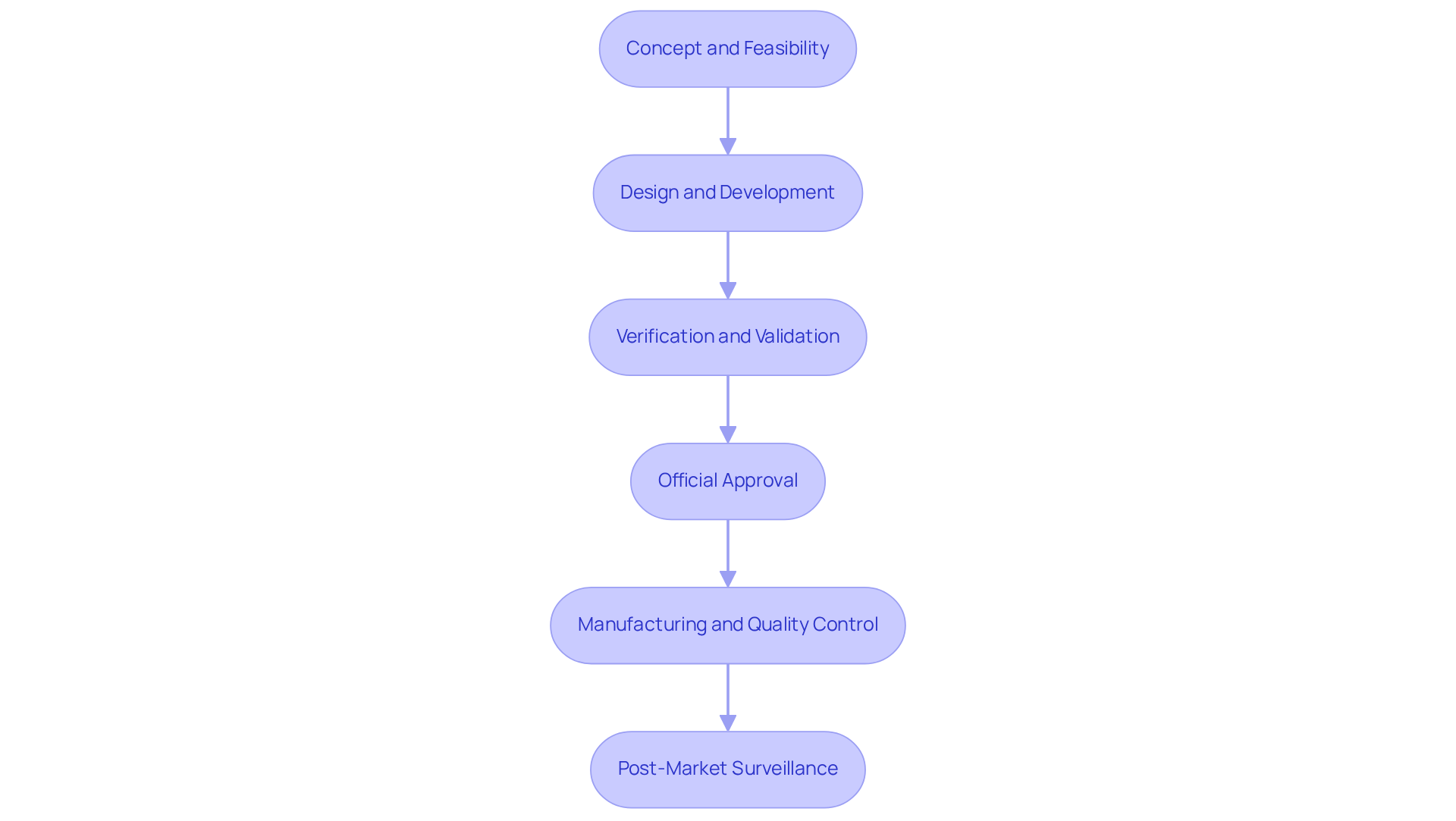
The encompasses several critical stages that ensure the successful creation and launch of innovative products:
- : This foundational stage focuses on identifying a medical need and conducting thorough market research to validate the concept. Defining the intended use and target user population is essential, as it sets the direction for subsequent phases. With the , such as diabetes, which affects approximately 422 million people globally, understanding market demands is more crucial than ever.
- Design and Development: During this phase, detailed design specifications are established, and prototypes are created. Key activities include selecting appropriate materials, designing user interfaces, and integrating necessary technologies. The emergence of personalized medicine and the emphasize the significance of innovative design in addressing specific patient needs.
- : This stage ensures that the apparatus meets its design specifications and user requirements. Verification involves rigorous testing against established specifications, while validation confirms that the apparatus fulfills its intended purpose. As oversight standards change, producers must to uphold adherence and guarantee product safety.
- : Before a device can be marketed, it must undergo a thorough review to ensure . This frequently necessitates providing comprehensive documentation to oversight organizations, such as the FDA. The enforcement of the Quality Management System Regulation (QMSR) starting February 2, 2026, represents a significant shift in regulatory requirements, necessitating immediate adaptation by manufacturers.
- : Once approved, the product enters production. This stage involves establishing efficient manufacturing processes and implementing stringent quality control measures to ensure consistency and reliability. Companies that prioritize digital readiness and integrate advanced quality management systems are better positioned to meet the demands of the evolving market.
- : After launch, continuous monitoring is vital to track the device's performance in real-world settings. This continuous monitoring addresses any arising problems and ensures ongoing adherence to compliance standards. The formalization of remote regulatory assessments (RRAs) has shifted the landscape to an 'inspection-anytime' reality, emphasizing the need for perpetual readiness in quality management.
By comprehending and skillfully maneuvering through these product phases, medical product manufacturers can enhance their opportunities for success in a competitive and swiftly evolving market.
Implement Best Practices for Each Development Phase
To ensure a successful , consider the following best practices for each phase:
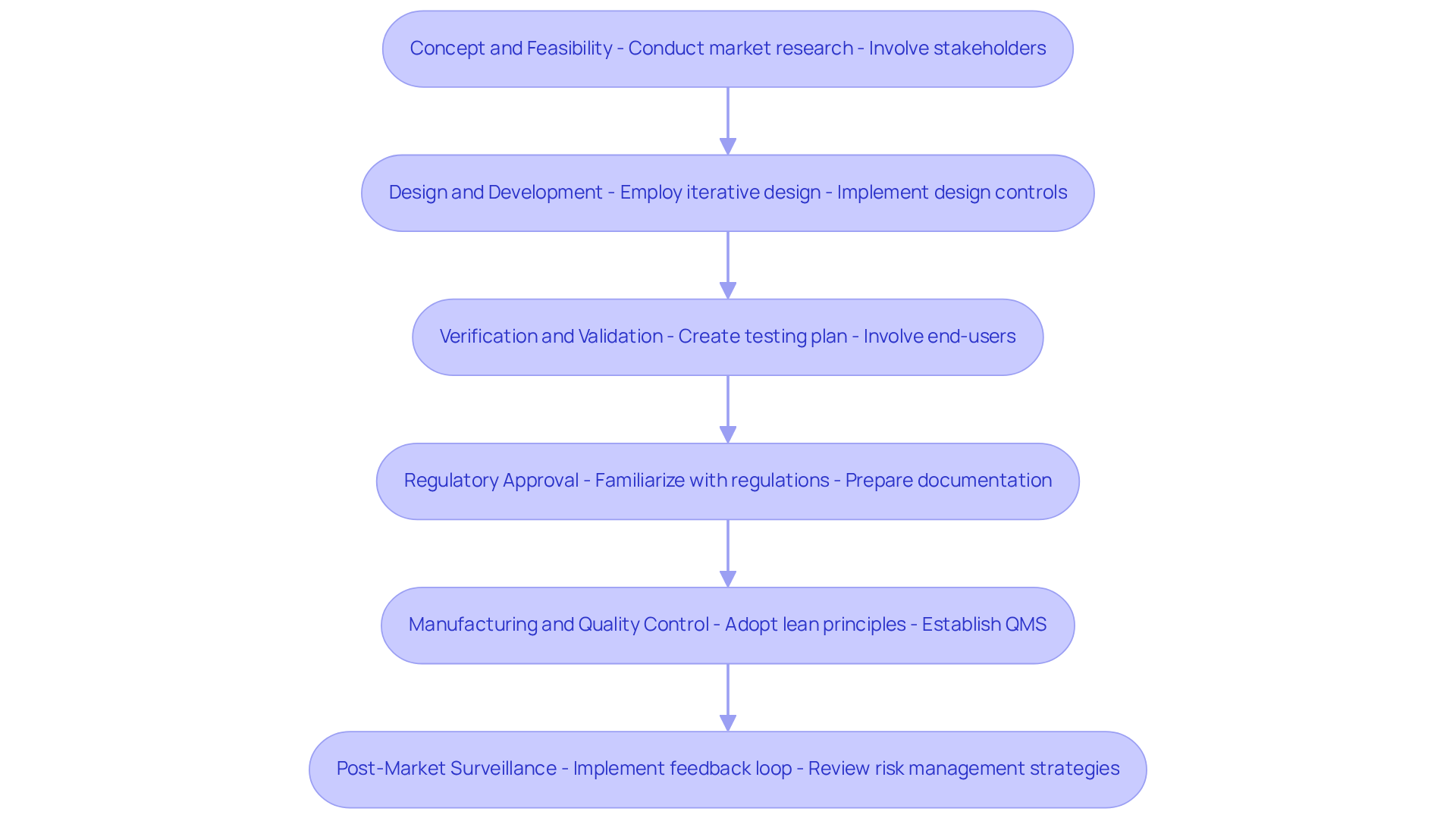
-
Concept and Feasibility:
- Conduct to identify user needs and analyze the competitive landscape. This foundational step is crucial for aligning the with market demands.
- Involve stakeholders early on to collect valuable insights and confirm the concept, fostering collaboration that can enhance the product's relevance and acceptance.
-
Design and Development:
- Employ iterative design processes that facilitate continuous feedback and improvements. This approach allows for adjustments based on real-time insights, ultimately leading to a more refined product. Adhering to can ensure that are completed on time and within budget, thereby enhancing overall project efficiency.
- Implement design controls to meticulously document changes and maintain traceability, ensuring that all modifications are well-recorded and justified throughout the development cycle. This documentation is critical for compliance, particularly in the medical equipment industry, and is an area where Voler Systems provides valuable assistance.
-
Verification and Validation:
- Create a robust testing plan that encompasses both functional and usability testing. This dual focus ensures that the equipment not only functions as intended but is also user-friendly. Identifying common errors in manufacturing tests early can prevent costly revisions later in the workflow, and consulting FAQs can provide insights into these frequent pitfalls.
- Involve end-users in validation testing to confirm that the device meets their needs and expectations, significantly enhancing user satisfaction and product success.
-
Regulatory Approval:
- Familiarize yourself with relevant regulations early in the process to prevent potential delays. Understanding the is essential for smooth navigation through , particularly with the FDA's ongoing efforts to improve transparency in healthcare communications.
- Prepare , as thorough documentation is critical for regulatory success. Voler Systems' expertise in can be invaluable during this phase.
-
Manufacturing and Quality Control:
- Adopt lean manufacturing principles to optimize production efficiency and minimize waste. Streamlining workflows can lead to cost reductions and improved product quality.
- Establish a (QMS) that aligns with ISO 13485 standards, ensuring that quality is embedded in every aspect of the manufacturing process. Note that the begins on February 2, 2026, which is critical for compliance.
-
Post-Market Surveillance:
- Implement a feedback loop to capture user experiences and device performance data. This ongoing collection of information is vital for continuous improvement throughout the and for addressing any emerging issues.
- Regularly review and update risk management strategies based on post-market data, ensuring that the product remains safe and effective throughout its lifecycle. Continuous adaptation is essential, particularly in light of evolving regulatory expectations and the integration of new technologies.
Ensure Regulatory Compliance and Quality Control
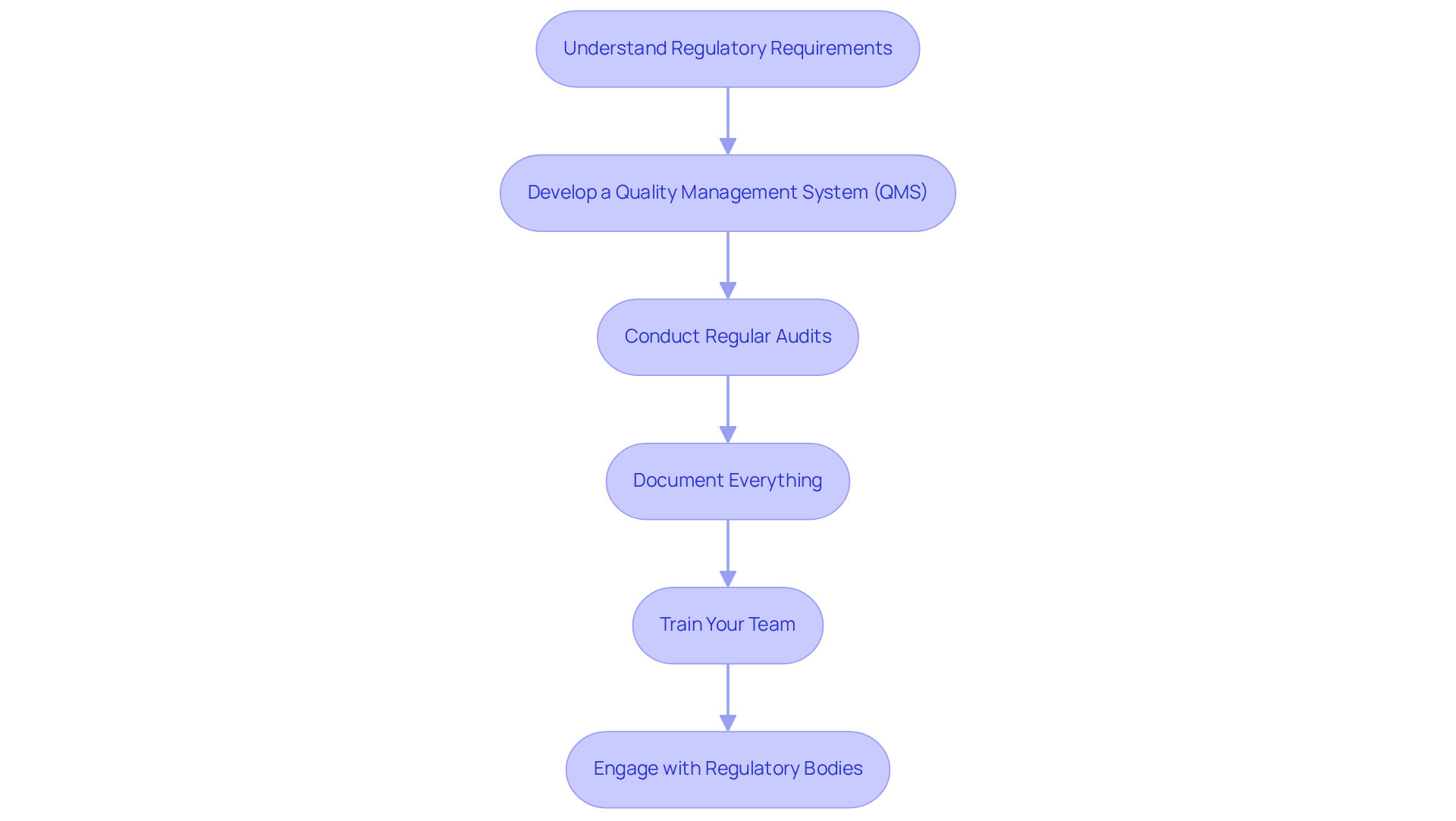
and quality control are essential components of successful . To ensure adherence, consider the following strategies:
- Understand : It is vital to familiarize yourself with the specific regulations applicable to your device, including . This foundational knowledge is crucial for effectively navigating the approval system.
- Develop a (QMS): Implement a QMS that aligns with . This system should encompass all facets of the development cycle, including from design and manufacturing to post-market activities, ensuring a comprehensive approach to quality.
- Conduct Regular Audits: Arrange to assess adherence to established procedures and identify areas for enhancement. This proactive strategy helps maintain high-quality standards and prepares your organization for external assessments.
- Document Everything: Maintain meticulous records of all procedures, decisions, and changes. This documentation is essential for official submissions and serves as proof of adherence during audits, reinforcing your commitment to quality.
- Train Your Team: Ensure that all team members are well-versed in compliance requirements and quality control processes. Regular training sessions can strengthen the significance of adherence and quality in product development, fostering a culture of excellence.
- Engage with : Establish open lines of communication with regulatory agencies. Timely and regular involvement can clarify requirements and simplify the approval process, thereby decreasing the risk of delays and regulatory issues.
As the medical equipment landscape evolves, particularly with the upcoming enforcement of the in February 2026, manufacturers must adapt their strategies to ensure compliance and maintain a competitive edge.
Foster Collaboration Among Development Teams
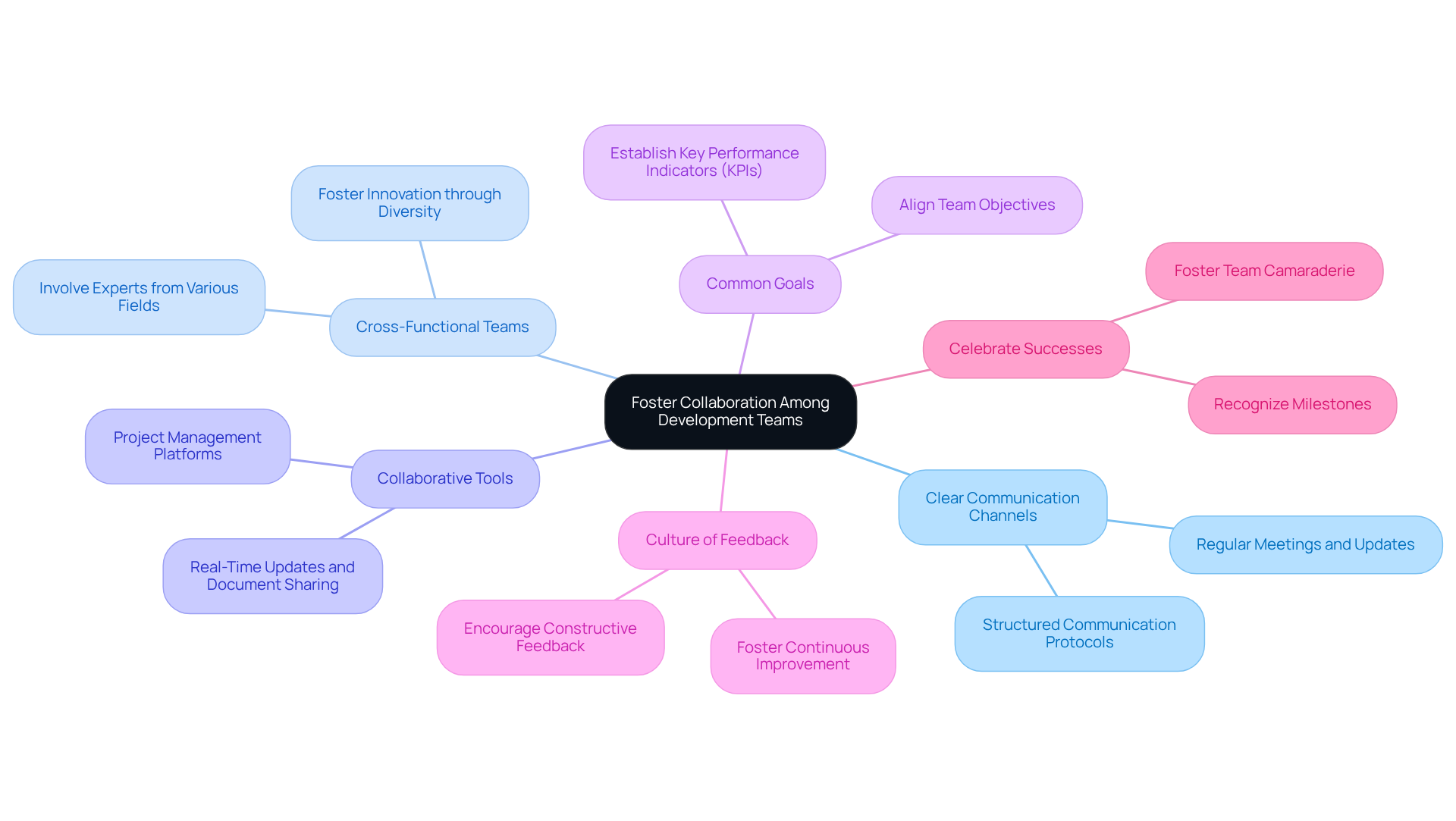
Collaboration is crucial for the successful development of . Here are strategies to enhance teamwork:
- Establish : Implement structured communication protocols to keep all team members informed and aligned. Regular meetings and updates facilitate transparency and ensure that everyone is aligned with project goals.
- Encourage by involving experts from various fields - engineering, regulatory, and marketing - during the . This diversity fosters innovation and ensures that multiple perspectives are integrated into the product phases, thereby enhancing overall effectiveness.
- Utilize Collaborative Tools: Leverage project management and that enable real-time updates and document sharing. Platforms such as Trello, Asana, or Slack can significantly improve coordination among team members, streamlining workflows and enhancing productivity.
- Set Common Goals: Align the team around (KPIs). This alignment helps maintain focus and encourages collective accountability, driving the project toward successful outcomes.
- Promote a : Foster an environment where team members feel comfortable providing constructive feedback throughout the development process. This culture of openness leads to continuous improvement and innovation, which are essential for navigating the complexities of .
- Celebrate Successes Together: Recognize and as a team. This practice fosters camaraderie and motivates team members to continue striving for excellence, reinforcing a positive team dynamic.
Conclusion
Mastering the phases of medical device development is essential for manufacturers aiming to deliver innovative and safe products to the market. By navigating through the critical stages - from concept and feasibility to post-market surveillance - developers can enhance their chances of success in a complex and competitive landscape. Each phase plays a pivotal role in ensuring that the final product not only meets regulatory standards but also effectively addresses real user needs.
This article emphasizes key practices for each stage of development. Conducting thorough market research and involving stakeholders early on can shape a product that resonates with users. Iterative design processes, robust testing plans, and meticulous documentation are crucial for verification, validation, and regulatory approval. Moreover, adopting lean manufacturing principles and establishing strong quality management systems are vital for maintaining product integrity throughout the lifecycle. Continuous collaboration among cross-functional teams fosters innovation and ensures that diverse perspectives are incorporated, ultimately leading to a more successful product.
As the medical device industry evolves, particularly with upcoming regulatory changes like the Quality Management System Regulation in 2026, staying informed and adaptable is imperative. Manufacturers are encouraged to prioritize compliance and quality control while fostering teamwork and open communication. By embracing these best practices, organizations can not only improve their development efficiency but also contribute to the advancement of healthcare solutions that enhance patient outcomes.
Frequently Asked Questions
What are the key stages of medical device development?
The key stages of medical device development include Concept and Feasibility, Design and Development, Verification and Validation, Official Approval, Manufacturing and Quality Control, and Post-Market Surveillance.
What is involved in the Concept and Feasibility stage?
This stage focuses on identifying a medical need, conducting market research to validate the concept, and defining the intended use and target user population.
Why is the Design and Development stage important?
This stage establishes detailed design specifications, creates prototypes, selects materials, designs user interfaces, and integrates technologies, all of which are crucial for meeting specific patient needs.
What does the Verification and Validation stage entail?
Verification involves testing the device against established specifications, while validation confirms that the device fulfills its intended purpose, ensuring compliance with user requirements.
What is required for Official Approval of a medical device?
A device must undergo a thorough review to ensure compliance with safety and efficacy standards, which includes providing comprehensive documentation to oversight organizations like the FDA.
What happens during the Manufacturing and Quality Control stage?
After approval, the product enters production, requiring the establishment of efficient manufacturing processes and stringent quality control measures to ensure consistency and reliability.
What is the purpose of Post-Market Surveillance?
Continuous monitoring is vital to track the device's performance in real-world settings, addressing any arising problems and ensuring ongoing compliance with standards.
How have regulatory requirements changed recently?
The enforcement of the Quality Management System Regulation (QMSR) starting February 2, 2026, represents a significant shift in regulatory requirements, necessitating adaptation by manufacturers.
List of Sources
- Outline the Key Stages of Medical Device Development
- Navigate 2026's Medical Device Manufacturing Trends (https://mddionline.com/medical-device-regulations/navigate-2026-s-medical-device-manufacturing-trends)
- 2026 Medical Device Quality Trends (https://mastercontrol.com/gxp-lifeline/2026-medical-device-trends-quality-management)
- What 2025 reveals about medtech’s next leap in 2026 (https://medicaldevice-network.com/features/what-2025-reveals-about-medtech-next-leap-in-2026)
- Medical Devices Market Size, Share, Global Growth Report 2034 (https://fortunebusinessinsights.com/industry-reports/medical-devices-market-100085)
- FDA Device Guidance Agenda: What to Watch in 2026 (https://hoganlovells.com/en/publications/fda-device-guidance-agenda-what-to-watch-in-2026)
- Implement Best Practices for Each Development Phase
- Top Medical Device Regulations to Watch in 2026 (https://xtalks.com/top-medical-device-regulations-to-watch-in-2026-4549)
- 2026 Medical Device Quality Trends (https://mastercontrol.com/gxp-lifeline/2026-medical-device-trends-quality-management)
- clarkstonconsulting.com (https://clarkstonconsulting.com/insights/2026-medical-device-industry-trends)
- Conversation with Evangeline Loh: Anticipated changes for the Medtech market in 2026 (https://ul.com/insights/conversation-evangeline-loh-anticipated-changes-medtech-market-2026)
- Navigate 2026's Medical Device Manufacturing Trends (https://mddionline.com/medical-device-regulations/navigate-2026-s-medical-device-manufacturing-trends)
- Ensure Regulatory Compliance and Quality Control
- 5 Critical Trends for Compliance and Innovation (https://mddionline.com/medical-device-regulations/2026-medical-device-quality-5-critical-trends-for-compliance-and-innovation)
- 2026 Medical Device Quality Trends (https://mastercontrol.com/gxp-lifeline/2026-medical-device-trends-quality-management)
- Medical Device Compliance: 2025 Insights & 2026 Priorities (https://medenvoyglobal.com/blog/medical-device-compliance-2025-insights-2026-priorities)
- gmp-compliance.org (https://gmp-compliance.org/gmp-news/overview-of-the-fdas-new-quality-management-system-regulation-effective-from-2-february-2026)
- JPM 2026: How to expand internationally while ensuring regulatory compliance (https://hoganlovells.com/en/publications/jpm-2026-how-to-expand-internationally-while-ensuring-regulatory-compliance)
- Foster Collaboration Among Development Teams
- Healthcare-Tech Collaboration Market Share & Industry Trends 2030 (https://realtimedatastats.com/research-report/healthcare-tech-collaboration-market)
- Want to Drive Innovation? You Need Cross-Functional Collaboration (https://americanhealthcareleader.com/2025/drive-innovation-with-cross-functional-collaboration)
- Future of Medical Device Manufacturing: Trends & Challenges 2026 (https://insight.averna.com/en/resources/blog/navigating-the-future-of-medical-device-manufacturing)
- deliberatedirections.com (https://deliberatedirections.com/quotes-future-of-healthcare)
- 37 Quotes on Health Care and Health Tech from 2016 (https://medium.com/@r_scott_munro/37-quotes-on-health-care-and-health-tech-from-2016-e34576edfc1f)





