Introduction
Bringing a new medical device to market presents numerous challenges, yet it also offers significant opportunities for innovation and enhanced patient care. The New Product Introduction (NPI) framework is essential, guiding stakeholders through the intricate stages of ideation, design, testing, and regulatory compliance. However, the journey toward successful NPI is fraught with obstacles.
How can organizations effectively navigate this complex landscape to ensure a smooth transition from concept to commercialization? By exploring best practices in NPI, we can uncover strategies that foster collaboration, maintain quality standards, and ultimately facilitate successful product launches within the medical device sector.
Define New Product Introduction (NPI) in Medical Devices
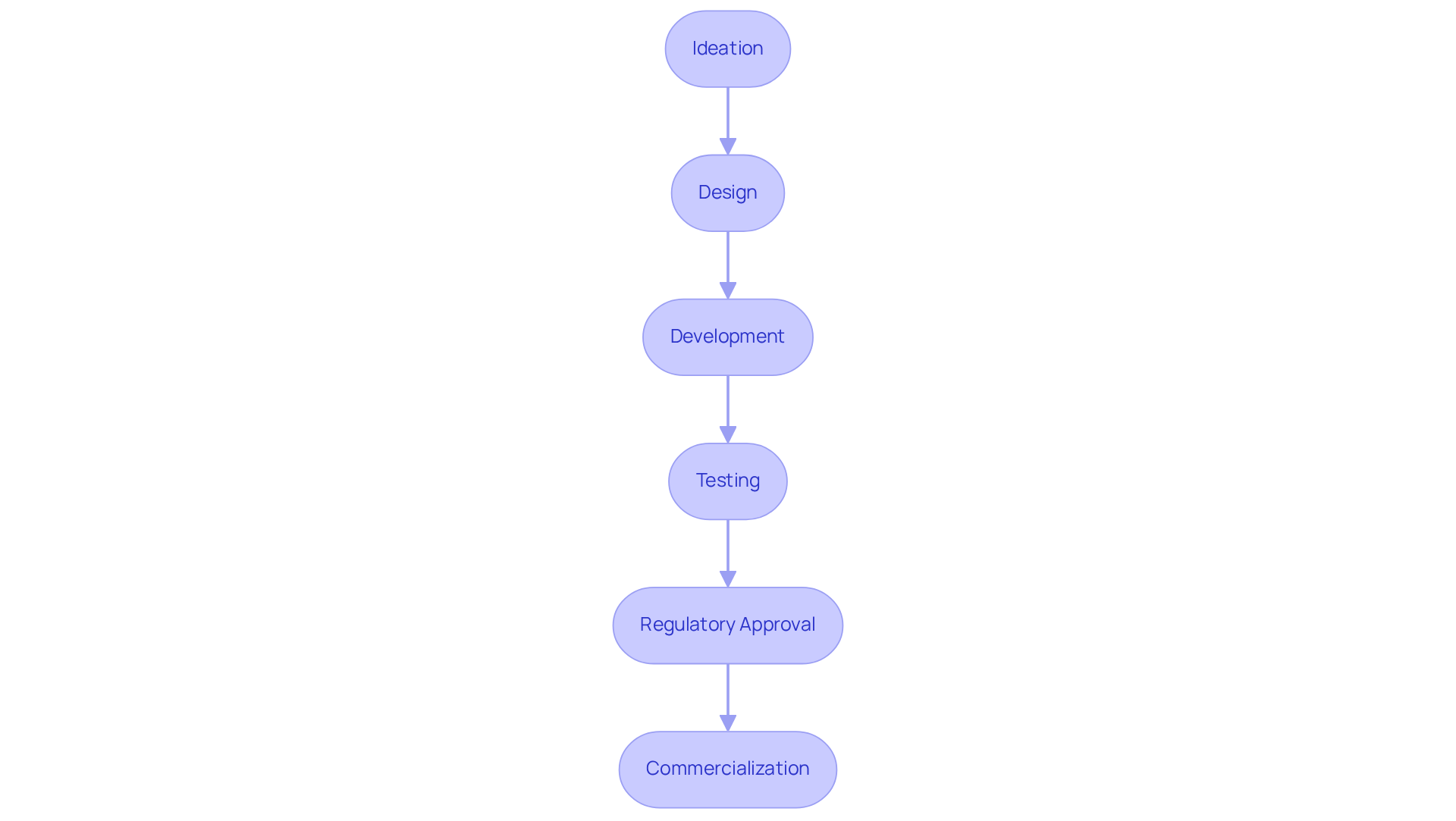
The process of in . This process includes several critical stages:
- Ideation
- Design
- Development
- Testing
- Regulatory approval
- Commercialization
The is essential in the due to that ensure patient safety and equipment efficacy. Understanding (NPI) is essential for stakeholders involved in , as it outlines the necessary steps to effectively launch a product that meets both regulatory compliance and market demands.
Additionally, is crucial for ensuring . By leveraging , such as those offered by Voler Systems, manufacturers can enhance their processes for , particularly in areas like AI-driven engineering and tailored for medical applications.
Outline Key Steps in the NPI Process for Medical Devices
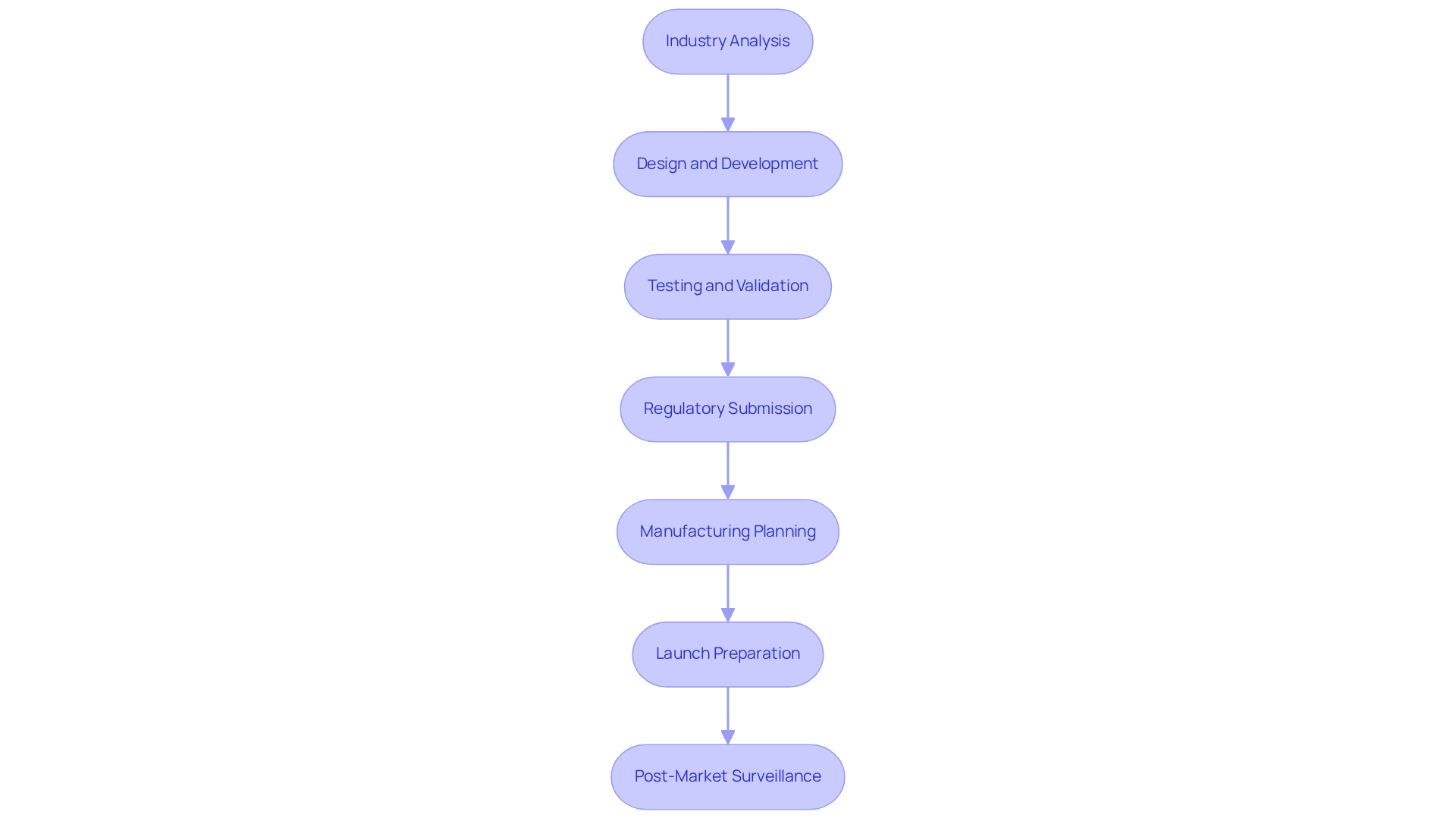
The process for medical devices consists of several essential steps that facilitate a successful market entry:
- Industry Analysis: This initial phase involves identifying industry needs and validating the product concept through user feedback and competitive evaluation. Effective is crucial, as it informs design decisions and aligns the product with user expectations.
- : During this stage, initial designs and prototypes are created, emphasizing functionality, usability, and adherence to regulatory standards. A well-structured design process can significantly reduce time-to-market and enhance product viability.
- : Rigorous testing is conducted to ensure the equipment meets safety and performance criteria. This includes preclinical and clinical trials, which are vital for demonstrating the device's efficacy and safety to regulatory bodies.
- Regulatory Submission: Preparing and submitting the necessary documentation to , such as the FDA, is a critical step. Adherence to relevant regulations is essential to prevent delays in approval and entry into the industry.
- ning: Developing a comprehensive involves sourcing materials, selecting suppliers, and establishing efficient production processes. This planning phase is crucial for ensuring that the product can be manufactured at scale without compromising quality.
- : A robust marketing strategy is created, alongside finalizing packaging and distribution plans. This preparation is essential to ensuring a and maximizing product visibility.
- : Following release, ongoing observation of the product's performance in the commercial sector is crucial. Gathering data for ongoing improvement and ensuring compliance with regulatory requirements helps maintain product quality and user satisfaction.
By meticulously following these steps, organizations can significantly enhance their chances of a successful while minimizing risks associated with and market acceptance.
Foster Cross-Functional Collaboration for Successful NPI
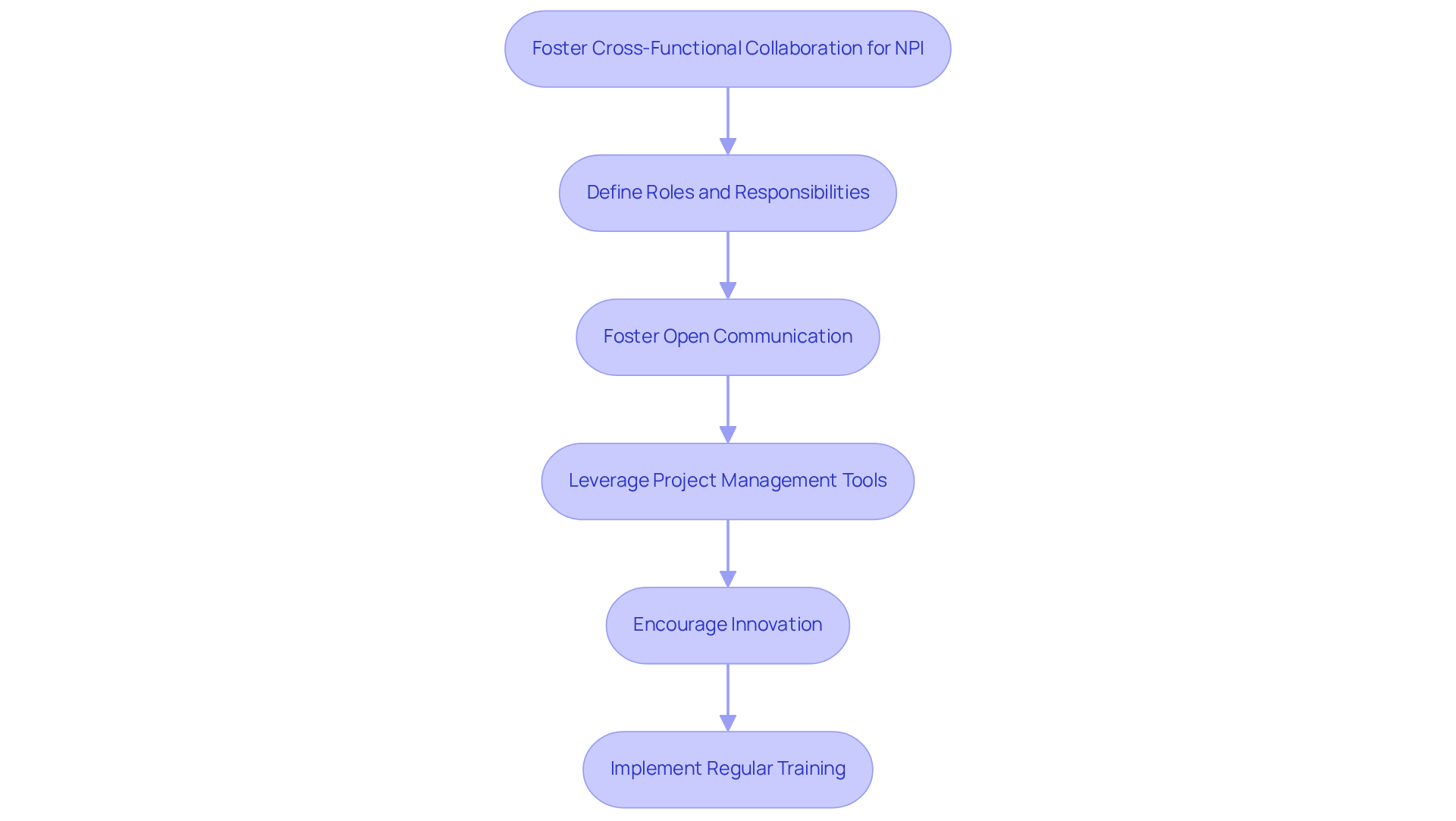
is essential for in the . It necessitates the integration of diverse teams from engineering, regulatory affairs, quality assurance, marketing, and manufacturing, all striving towards a unified objective. The following key practices can enhance collaboration:
- : Clearly delineate each team member's role to promote accountability and streamline communication.
- : Create an environment where team members feel empowered to share ideas, feedback, and concerns. Regular meetings and collaborative tools can facilitate this exchange.
- Leverage : Employ project management software to monitor progress, manage tasks, and ensure alignment across teams, thereby enhancing overall efficiency.
- : Inspire team members to think creatively and propose inventive solutions to challenges encountered during the NPI stage, fostering continuous improvement.
- : Offer training sessions that enhance team members' understanding of each other's roles and the NPI process, fostering empathy and cooperation.
By prioritizing these collaborative practices, organizations can significantly enhance their efforts, which will lead to successful and improved market performance.
Ensure Regulatory Compliance and Quality Assurance in NPI
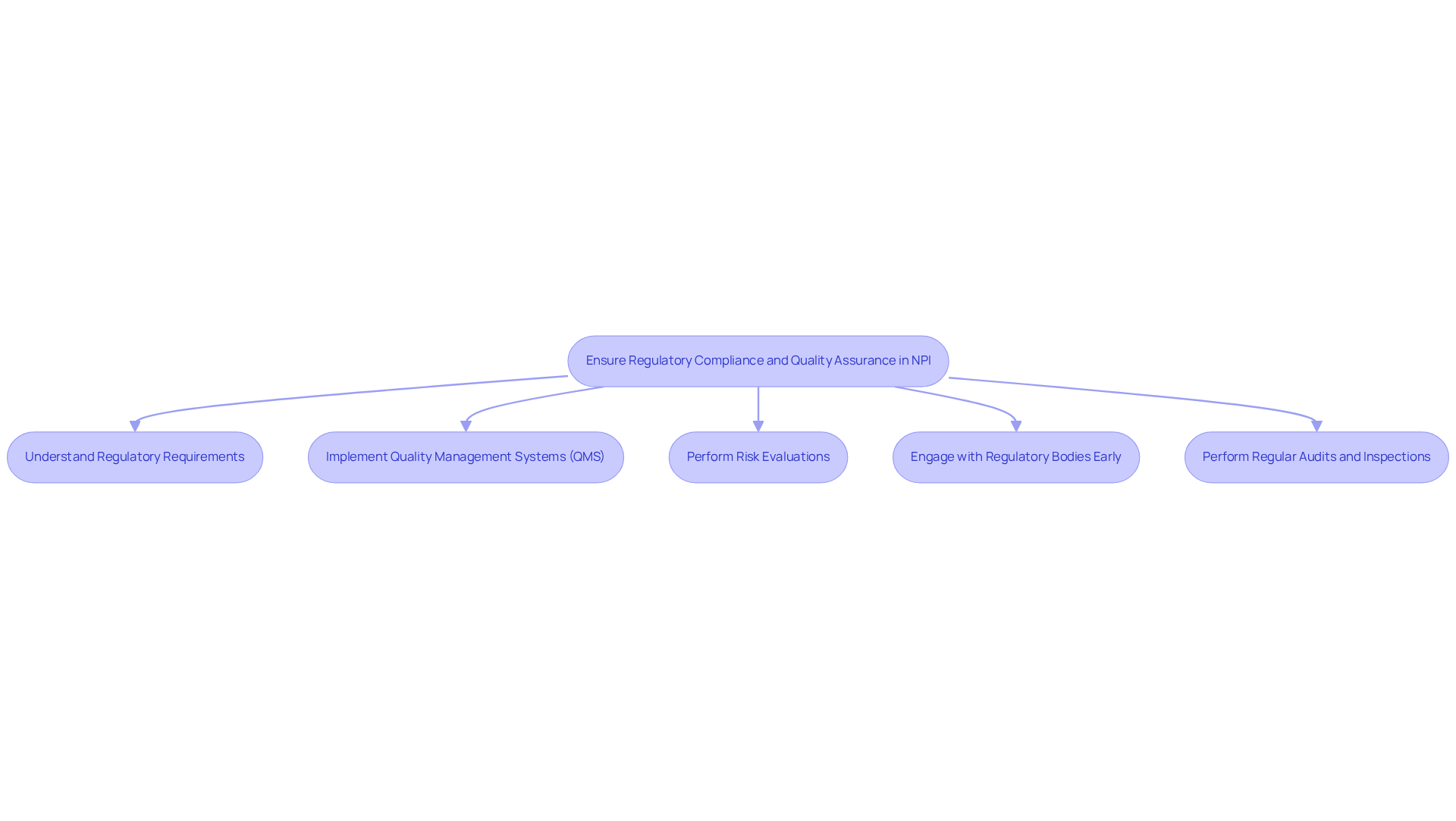
are essential elements of the framework for medical instruments. To ensure adherence to regulations and uphold high-quality standards, organizations should consider the following key practices:
- Understand : It is essential to familiarize oneself with the specific regulations applicable to the product, including FDA guidelines and ISO standards. This foundational knowledge is crucial for successful submissions and compliance.
- Implement (QMS): Establishing a robust QMS that aligns with regulatory requirements is vital. This ensures that all procedures are documented, monitored, and continuously improved. A well-structured QMS can significantly enhance success rates for by fostering a culture of quality.
- : Regular evaluations of potential hazards related to the equipment and its production methods are necessary. Proactively implementing mitigation strategies for identified risks can prevent costly recalls and enhance patient safety, as device failure accounted for 11.1% of all recall events in recent years.
- : Maintaining open communication with regulatory agencies throughout the development phase is important. Early involvement can provide valuable insights and assist in addressing potential compliance issues before they arise, thereby lowering the risk of disruption.
- : Conducting internal audits to ensure compliance with established processes and standards is crucial. Preparing for external audits by maintaining thorough documentation and promptly addressing any non-conformities is essential for sustaining compliance and quality assurance.
By prioritizing these practices, organizations can , and improve their chances of a successful market launch through , ultimately leading to better patient outcomes and satisfaction.
Conclusion
The successful introduction of new medical devices relies on a well-structured New Product Introduction (NPI) process that effectively guides products from concept to market. By comprehensively understanding the critical stages involved - from ideation and design to regulatory approval and commercialization - stakeholders can ensure that their products meet both market demands and stringent regulatory standards. The significance of NPI in the medical device sector is paramount, as it not only safeguards patient safety but also enhances overall product efficacy.
Key insights explored in this article include:
- The necessity of cross-functional collaboration
- The establishment of robust quality management systems
- The implementation of effective risk evaluation strategies
Each step of the NPI process, including thorough testing, regulatory submission, and post-market surveillance, plays a pivotal role in minimizing risks and maximizing the chances of a successful product launch. By fostering an environment of open communication and teamwork among diverse teams, organizations can significantly enhance their NPI efforts, leading to improved market performance and better patient outcomes.
Ultimately, prioritizing best practices in new product introduction is essential for navigating the complexities of the medical device industry. Embracing these strategies not only facilitates compliance with regulatory requirements but also promotes innovation and continuous improvement. As the landscape of medical technology evolves, a commitment to excellence in NPI will remain a cornerstone for organizations aiming to make a meaningful impact in healthcare.
Frequently Asked Questions
What is New Product Introduction (NPI) in medical devices?
New Product Introduction (NPI) in medical devices is the process that systematically transitions a new medical instrument from concept to market, encompassing several critical stages.
What are the critical stages of the NPI process?
The critical stages of the NPI process include ideation, design, development, testing, regulatory approval, and commercialization.
Why is NPI important in the medical equipment sector?
NPI is essential in the medical equipment sector due to stringent regulatory requirements and the necessity for high-quality standards that ensure patient safety and equipment efficacy.
Who needs to understand the NPI process?
Stakeholders involved in medical product development need to understand the NPI process, as it outlines the necessary steps to effectively launch a product that meets regulatory compliance and market demands.
What common issues should be recognized in the NPI process?
Recognizing common pitfalls in manufacturing tests is crucial for ensuring quality and efficiency in electronic product design during the NPI process.
How can manufacturers enhance their NPI processes?
Manufacturers can enhance their NPI processes by leveraging innovative electronic design solutions, such as AI-driven engineering and ultra-low power solutions tailored for medical applications, as offered by companies like Voler Systems.
List of Sources
- Define New Product Introduction (NPI) in Medical Devices
- Quality 4.0: What Defines Successful New Product Introduction? (https://qualitydigest.com/inside/innovation-article/quality-40-what-defines-successful-new-product-introduction-040918.html)
- Medical Device Product Development Case Study (https://greenlight.guru/blog/medical-device-product-development-case-study)
- Medical Device NPI Case Study (https://sanmina.com/media-center/case-studies/medical-device-npi-case-study)
- Quality 4.0 and Manufacturing New Product Introduction (NPI) (https://blog.lnsresearch.com/quality-4.0-and-new-product-introductions)
- birlasoft.com (https://birlasoft.com/articles/how-to-boost-npi-in-medical-device-industry-with-mdm)
- Outline Key Steps in the NPI Process for Medical Devices
- Medical Device Industry Facts, Trends and Statistics 2025 (https://arterexmedical.com/medical-device-industry-statistics)
- Complete guide to the new product introduction process - NPI Guide | Ricardo (https://ricardo.com/en/news-and-insights/industry-insights/guide-to-the-new-product-introduction-npi-process)
- Medical Device Regulatory News and Updates (https://pureglobal.com/resources/regulatory-updates)
- grandviewresearch.com (https://grandviewresearch.com/industry-analysis/medical-device-clinical-trials-market-report)
- Foster Cross-Functional Collaboration for Successful NPI
- Poor Communication Drives 70% of Device Reg Problems (https://mddionline.com/medical-device-regulations/communication-breakdowns-not-complexity-drive-70-of-costly-medical-device-regulatory-problems)
- 2025 NPI Award for AI-driven Automated Downtime Labeling (https://archsys.io/hub/news/arch-systems-wins-2025-npi-award-for-ai-driven-revolution-in-manufacturing-process-control)
- The Role of Collaboration in Medical Device Innovation: Partnering with Experts for Breakthrough Solutions (https://occamdesign.com/the-role-of-collaboration-in-medical-device-innovation-partnering-with-experts-for-breakthrough-solutions)
- The Winning Formula: Cross-Functional Collaboratio... (https://medicalaffairsspecialist.org/blog/the-winning-formula-cross-functional-collaboration-as-a-catalyst)
- Collaborate, Compete, or Coexist? Navigating the Landscape of Medical Device Companies | (https://frontlinemedtech.com/collaborate-compete-or-coexist-navigating-the-landscape-of-medical-device-companies)
- Ensure Regulatory Compliance and Quality Assurance in NPI
- c2a-sec.com (https://c2a-sec.com/60-healthcare-and-medical-device-cybersecurity-risk-statistics-for-2025)
- Medical Device Compliance: 2025 Insights & 2026 Priorities (https://medenvoyglobal.com/blog/medical-device-compliance-2025-insights-2026-priorities)
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)





