Best Practices for Manufacturing a Prototype in Medical Devices
Discover best practices for manufacturing a prototype in medical devices to ensure...

Navigating the intricate landscape of medical device quality management systems (QMS) is essential for ensuring compliance and achieving operational excellence within the healthcare sector. Organizations can realize substantial benefits by mastering the fundamental practices that support an effective QMS, which includes understanding regulatory standards and implementing robust risk management strategies.
However, a significant challenge persists: how can manufacturers not only fulfill compliance requirements but also cultivate a culture of continuous improvement and innovation in their processes?
This article explores the critical components and best practices that can empower medical device companies to excel in a highly regulated environment.
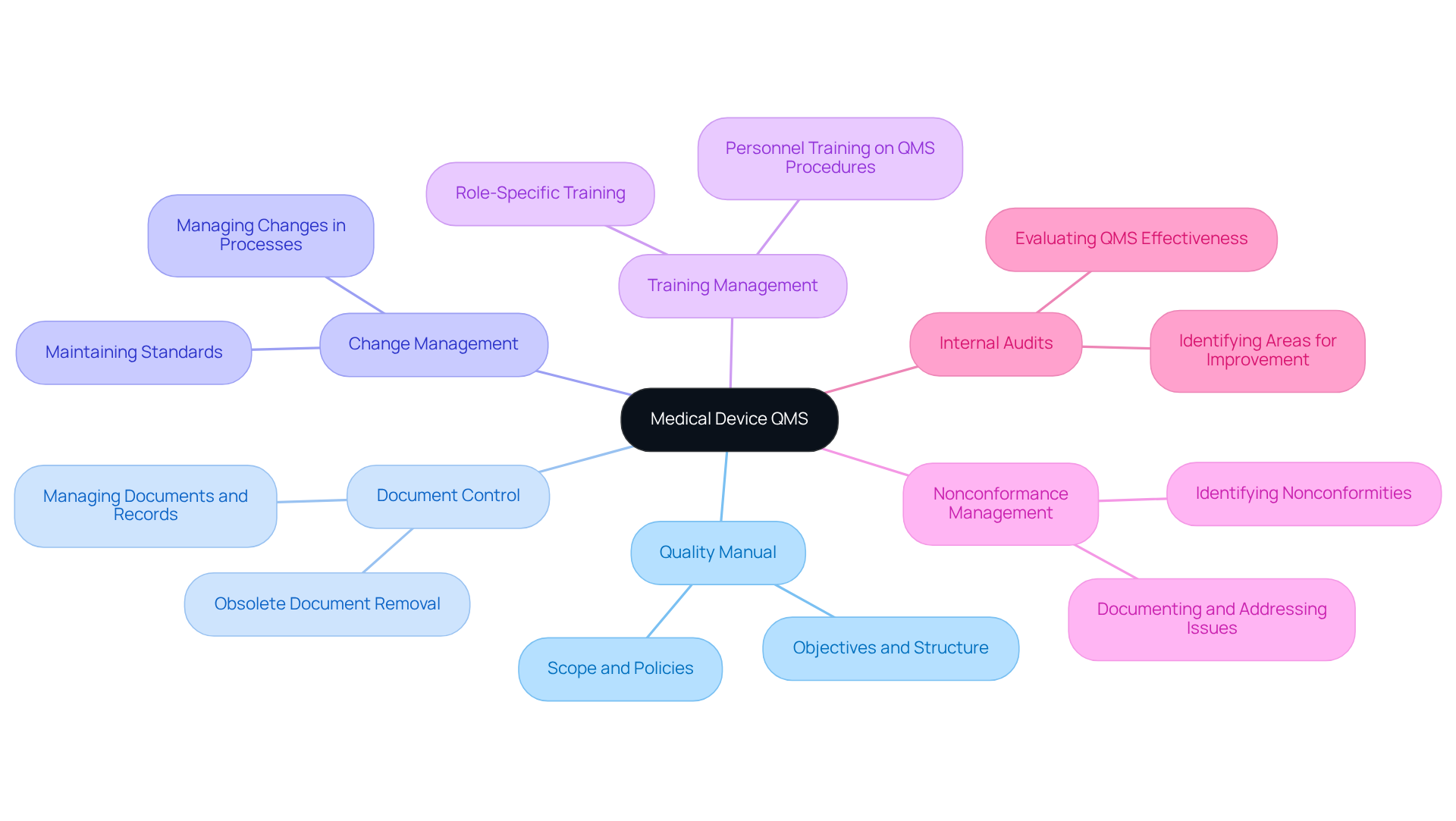
A medical device qms application must encompass several essential elements to ensure compliance and operational efficiency. These components include:
By clearly defining these components, organizations can establish a robust medical device qms application that supports compliance with regulatory standards such as ISO 13485 and FDA regulations.
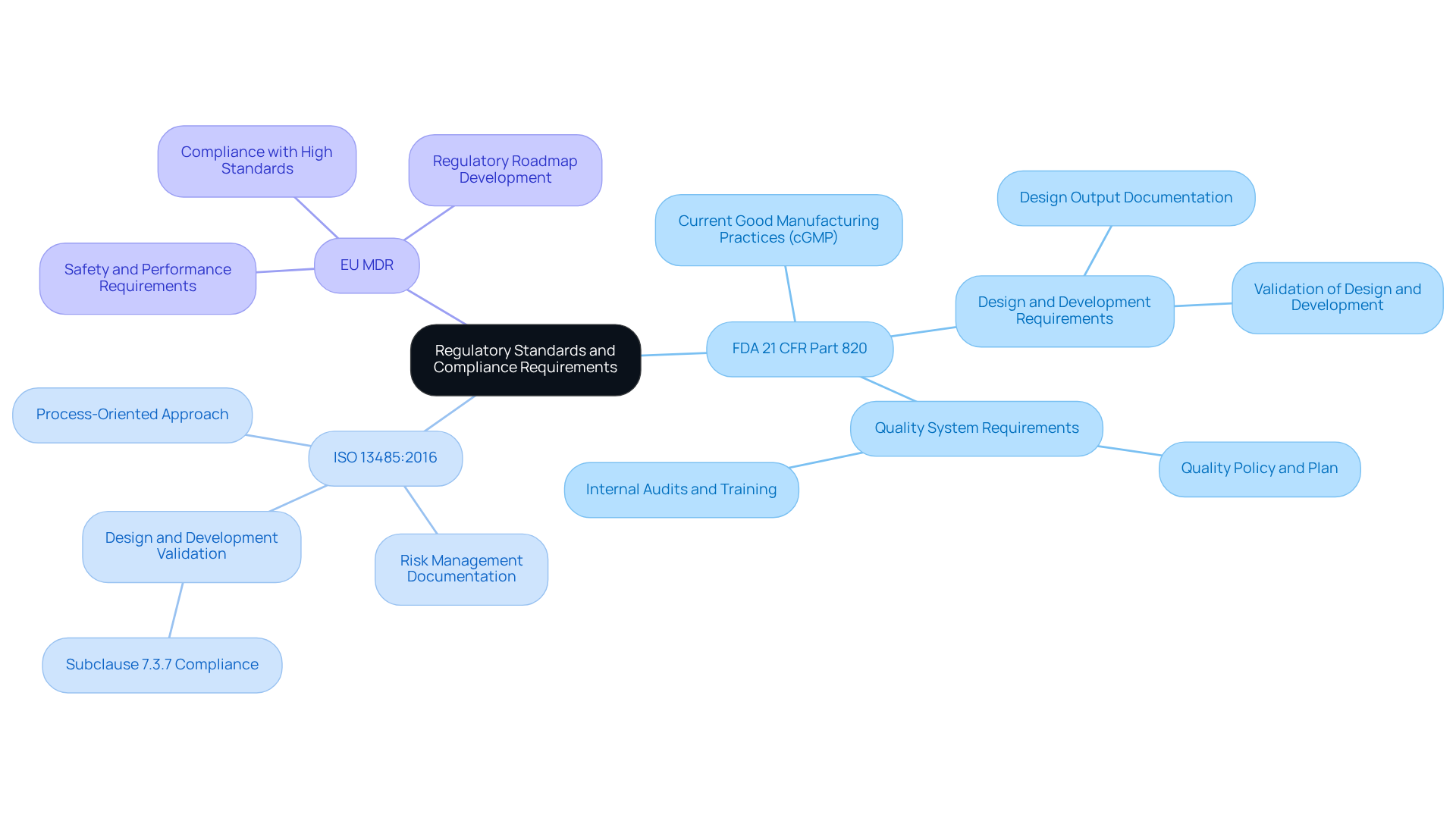
Implementing an effective medical device qms application for medical products requires a comprehensive understanding of relevant regulatory standards and compliance requirements. The primary regulations include:
To align with these standards, organizations should conduct a thorough gap analysis to identify discrepancies between current practices and regulatory expectations. Developing a regulatory roadmap to address identified gaps is crucial. Furthermore, regular training and updates on evolving regulatory changes are essential for maintaining compliance and ensuring that all personnel are informed and prepared to meet industry standards.
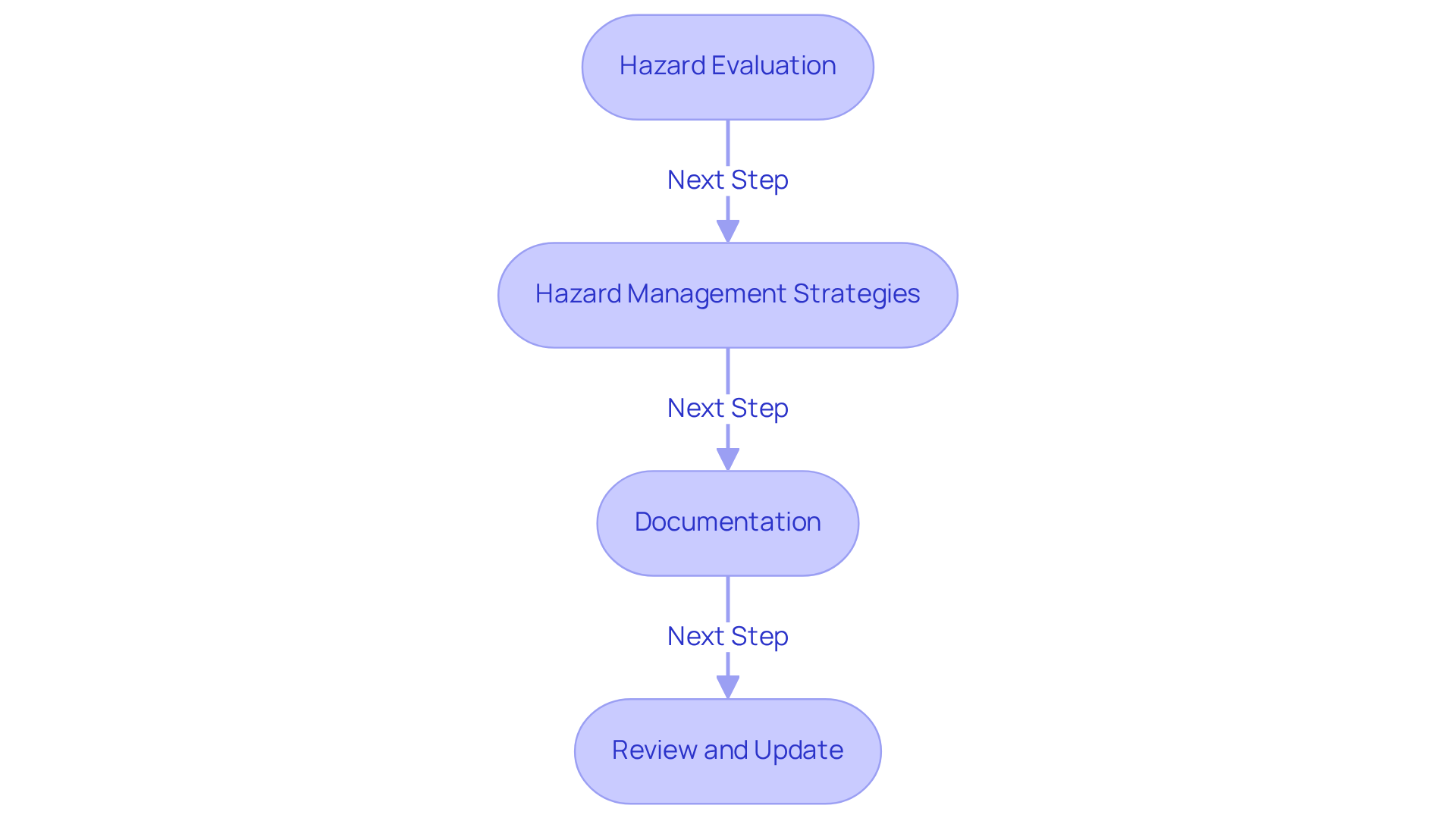
Effective management strategies are crucial for ensuring the safety and effectiveness of medical equipment. Key practices include:
Hazard Evaluation: Conduct comprehensive evaluations to identify potential dangers associated with the device throughout its lifecycle. Utilize tools such as Failure Modes and Effects Analysis (FMEA) and Hazard Analysis, which have proven effective in identifying a significant number of failure modes - over 9,564 in recent studies - underscoring their role in managing uncertainties. At Voler Systems, our prototype approach facilitates early detection of issues during the design phase, enabling prompt adjustments that enhance safety and compliance.
Hazard Management Strategies: Develop and implement hazard management strategies to mitigate identified threats. This may involve design modifications, labeling changes, or additional testing. The ISO 14971 standard, recognized by the FDA, emphasizes the importance of prioritizing inherently safe designs and protective measures as primary control options. At Voler Systems, we ensure that our design adjustments align with this standard, thereby enhancing the safety of our medical devices.
Documentation: Maintain thorough records of hazard management activities, including assessments, control measures, and their effectiveness. This documentation serves as a vital communication tool during regulatory inspections, ensuring transparency and compliance. Voler Systems integrates robust documentation practices into our Quality Management System (QMS), aiding our clients in the medical device QMS application to meet regulatory requirements.
Review and Update: Regularly evaluate and revise management plans to reflect changes in regulations, technology, and user feedback. Ongoing assessment is essential, as new challenges may emerge from the implementation of control measures, necessitating an adaptable management process. Our team at Voler Systems is dedicated to ensuring that our clients' management strategies evolve in accordance with industry standards and technological advancements.
By incorporating these strategies into the medical device QMS application, organizations can enhance their ability to manage risks effectively and ensure compliance with regulatory standards, ultimately leading to safer medical products.
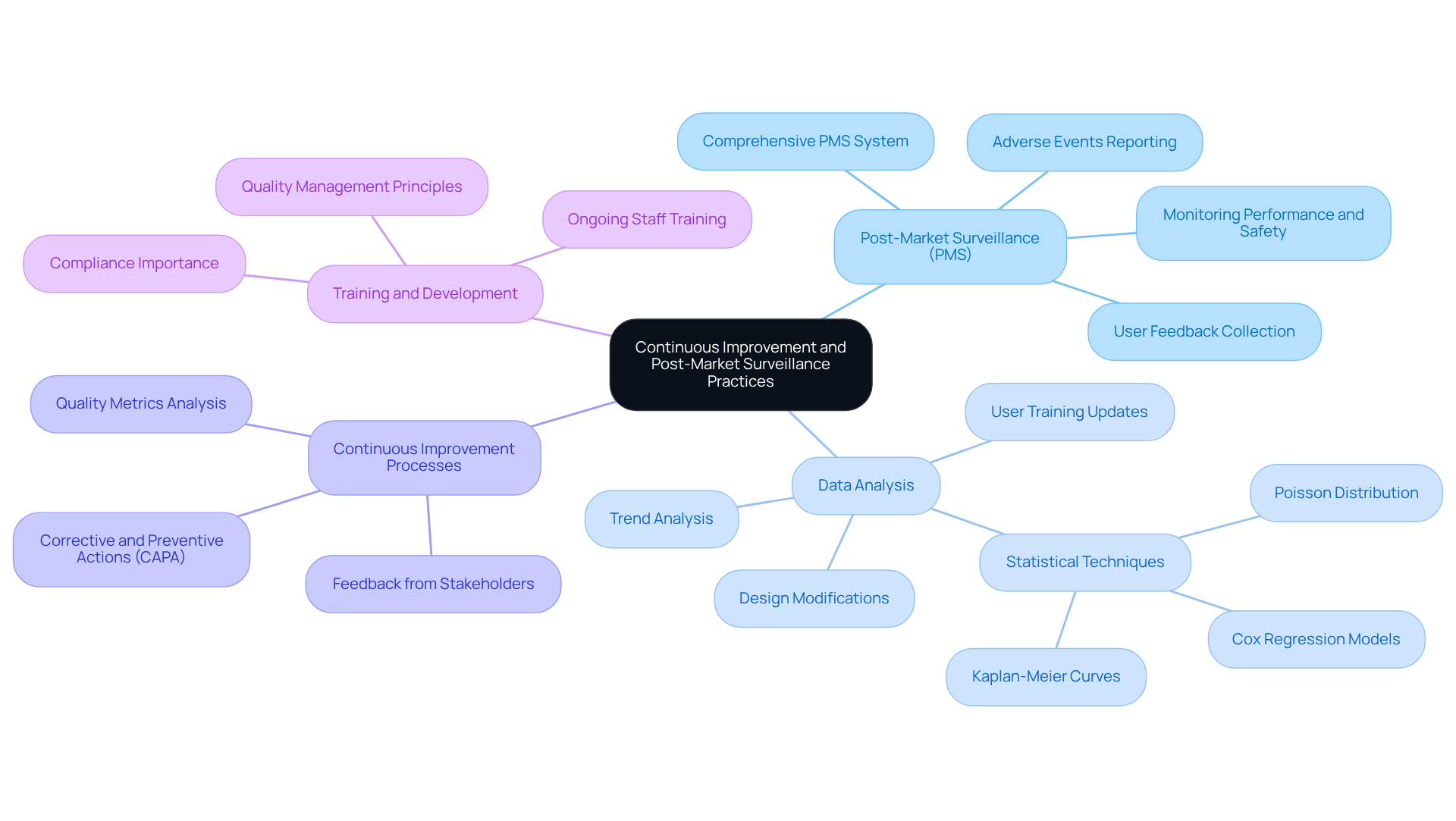
To maintain compliance and ensure product quality, organizations must implement robust practices for continuous improvement and post-market surveillance (PMS). Key components include:
Post-Market Surveillance (PMS): Establish a comprehensive PMS system to monitor the performance and safety of medical instruments after launch. This entails systematically gathering information on equipment malfunctions, user feedback, and adverse events, which is essential for recognizing potential risks and ensuring patient safety.
Data Analysis: Regularly analyze PMS data to uncover trends and areas needing improvement. Utilize statistical techniques, including the Poisson distribution and Kaplan-Meier curves, to assess the occurrence of adverse events and the duration of performance. This analysis informs necessary design modifications, updates to labeling, and additional user training.
Continuous Improvement Processes: Cultivate a culture of continuous improvement by actively seeking feedback from all stakeholders, including employees, customers, and regulatory bodies. Implement corrective and preventive actions (CAPA) based on insights gained from audits, PMS findings, and other quality metrics to enhance product safety and efficacy.
Training and Development: Provide ongoing training for staff on quality management principles and the significance of continuous improvement in maintaining compliance and elevating product quality. This ensures that all team members are equipped to contribute to the organization's commitment to excellence.
By embedding these practices into the medical device QMS application, organizations can not only comply with regulatory standards but also continuously enhance the safety and effectiveness of their medical devices, ultimately fostering innovation and patient trust.
Establishing a comprehensive medical device Quality Management System (QMS) is crucial for ensuring compliance and achieving operational excellence in the medical device industry. By concentrating on essential components such as quality manuals, document control, change management, and effective training, organizations can develop a robust framework that not only satisfies regulatory requirements but also enhances product quality and safety.
This article underscores key practices that are fundamental to a successful medical device QMS application. These include:
Each of these elements is vital for maintaining compliance and fostering an environment of safety and innovation within the organization. Furthermore, the emphasis on post-market surveillance and data analysis highlights the importance of ongoing evaluation and refinement of practices to ensure that medical devices consistently meet high standards of safety and efficacy.
Ultimately, a commitment to mastering these best practices not only supports compliance with current regulations but also positions organizations to proactively address emerging challenges in the medical device landscape. By prioritizing a culture of continuous improvement and robust risk management, stakeholders can enhance patient trust and drive innovation, ensuring that medical devices are not only compliant but also genuinely beneficial to those who rely on them.
What is a medical device Quality Management System (QMS)?
A medical device QMS is a structured system that encompasses essential elements to ensure compliance and operational efficiency in the management of medical devices.
What are the key components of a medical device QMS?
The key components include a Quality Manual, Document Control, Change Management, Training Management, Nonconformance Management, and Internal Audits.
What is the purpose of the Quality Manual in a medical device QMS?
The Quality Manual delineates the scope of the QMS, including policies, objectives, and the organizational structure.
Why is Document Control important in a medical device QMS?
Document Control is important to manage documents and records effectively, ensuring that only current versions are used and obsolete documents are systematically removed.
What is Change Management in the context of a medical device QMS?
Change Management refers to the system implemented to manage changes in processes, products, or documentation to maintain standards and quality.
How does Training Management contribute to a medical device QMS?
Training Management ensures that all personnel receive adequate training on QMS procedures and their specific roles within the system.
What is Nonconformance Management in a medical device QMS?
Nonconformance Management involves developing processes for identifying, documenting, and addressing nonconformities to prevent recurrence.
Why are Internal Audits necessary in a medical device QMS?
Internal Audits are necessary to evaluate the effectiveness of the QMS and identify areas for improvement.
How do these components support compliance with regulatory standards?
By clearly defining these components, organizations can establish a robust medical device QMS that supports compliance with regulatory standards such as ISO 13485 and FDA regulations.
