Understanding Embedded Engineer Meaning in Modern Technology
Introduction The realm of technology is increasingly characterized by the seamless...

Navigating the complexities of medical device development necessitates a strategic blend of regulatory compliance, innovative engineering solutions, and effective collaboration. As the industry evolves, a thorough understanding of the intricate landscape of FDA regulations and ISO standards is essential for ensuring product success. This article examines best practices that can enhance product engineering in the medical sector, highlighting how companies can leverage AI-driven technologies and foster cross-functional teamwork to meet both market demands and regulatory requirements. With the stakes higher than ever, what key strategies can propel medical device manufacturers toward success in an increasingly competitive environment?
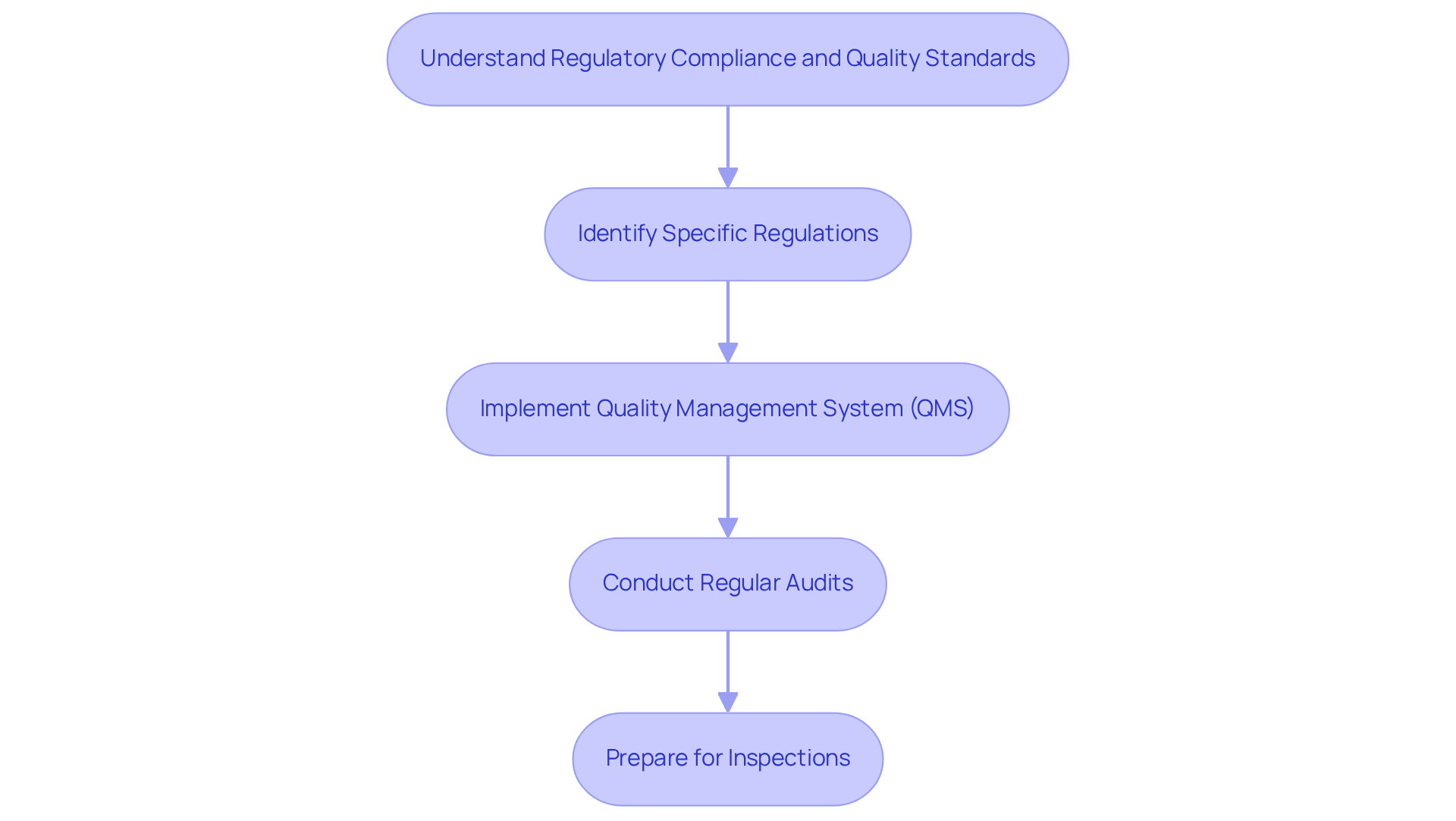
Navigating the healthcare equipment landscape requires a thorough understanding of . This encompasses familiarity with the FDA's regulations, ISO standards, and . Start by identifying the specific regulations pertinent to your equipment type, as these can vary significantly based on classification (Class I, II, or III).
Implementing a robust (QMS) that aligns with is crucial for ensuring consistent quality throughout the product lifecycle. Regular audits and training sessions are essential for maintaining compliance and preparing for inspections. For example, a company that integrated a QMS early in its development process reported a due to fewer compliance-related delays.
As of February 2, 2026, the revised emphasizes the incorporation of ISO 13485:2016, making adherence not just beneficial but essential for success in the health product sector. has effectively assisted numerous manufacturers in navigating these complexities, ensuring their products meet all necessary while upholding high quality standards. A notable case involved a healthcare equipment startup that, with ' guidance, achieved compliance ahead of schedule, significantly enhancing their market entry strategy.
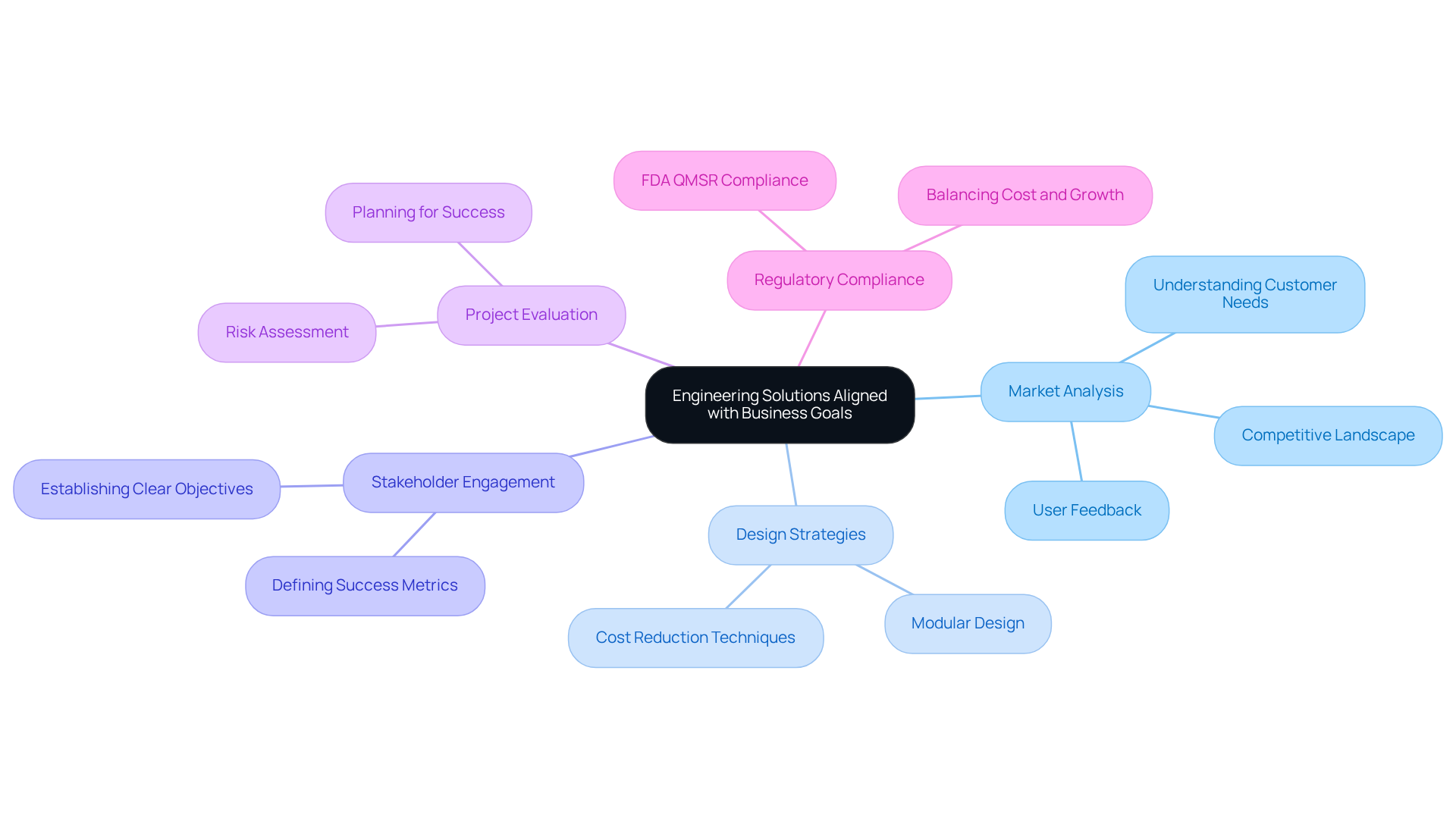
Selecting the appropriate is vital for the success of . A thorough is essential to understand customer needs and the competitive landscape, which should guide the selection of technologies and design strategies that align with business objectives.
For example, implementing can significantly lower production costs by streamlining manufacturing and assembly processes. Additionally, engaging with stakeholders to establish clear objectives and success metrics is crucial. Engineers must learn to:
A notable case study involving a wearable health monitor demonstrated that led to a 25% increase in user satisfaction and a significant reduction in post-launch modifications. This underscores the importance of into the to enhance product effectiveness and market readiness.
Furthermore, with the set to take effect in February 2026, companies must ensure their comply with evolving standards. Incorporating insights from industry experts, such as Namrita Negi, who highlights the necessity of , can further assist manufacturers in making informed decisions that meet both market demands and regulatory requirements.
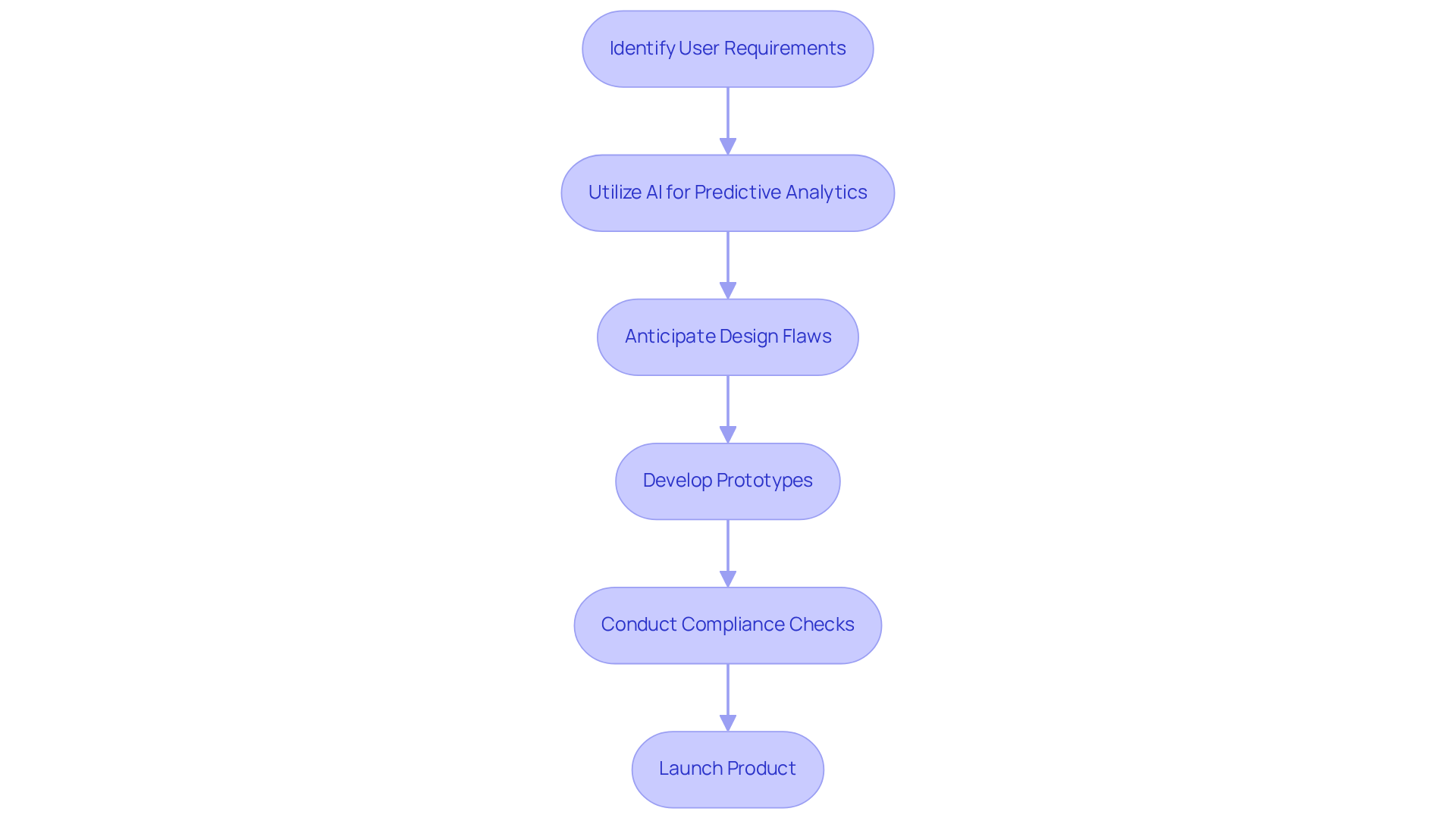
by significantly shortening . By , manufacturers can anticipate potential before physical prototypes are created.
For example, , known for its expertise in and , has developed numerous ultra-low-power designs that leverage AI to enhance functionality based on actual usage patterns. This method ensures that products meet user requirements effectively while accounting for environmental factors such as moisture and temperature that can affect performance.
Furthermore, collaborates closely with clients to develop for wearable technology, streamlining compliance checks and addressing the challenges associated with AI integration in medtech. This proactive approach not only refines the design process but also , ultimately leading to more .
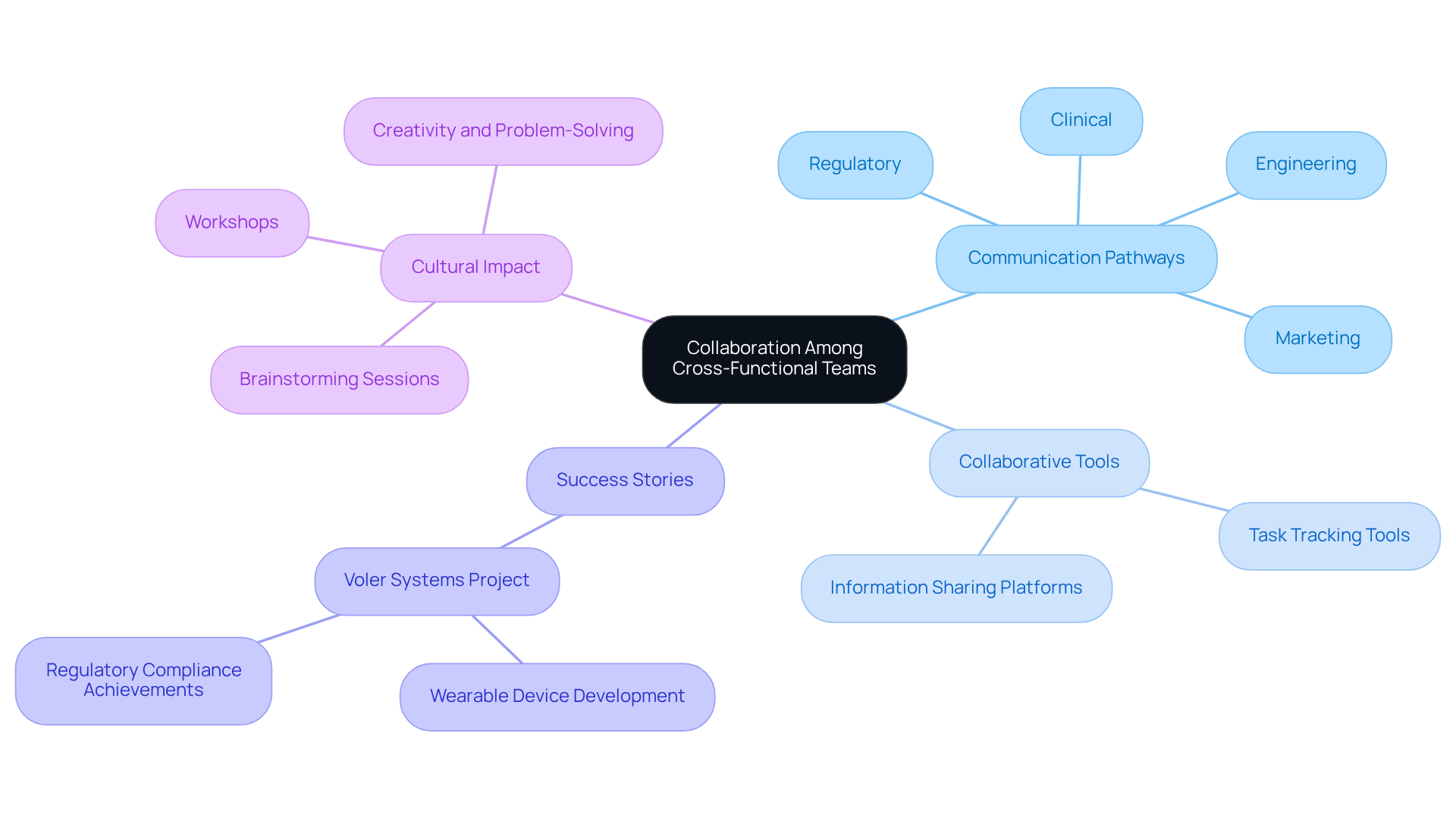
Collaboration among is crucial for the . Establishing regular communication pathways among engineering, regulatory, marketing, and clinical teams ensures alignment on objectives and timelines. Utilizing and platforms that facilitate information sharing and task tracking can significantly enhance efficiency. For instance, a healthcare technology firm that adopted a cross-disciplinary team strategy experienced a 50% reduction in delays, underscoring the impact of diverse perspectives on innovation.
exemplifies this commitment to teamwork, ensuring compliance with emissions and ESD standards through a thorough review process, which is vital for the success of medical equipment. In a notable project, partnered with a client to create a wearable device that not only satisfied regulatory requirements but also enhanced user experience, garnering positive feedback from end-users.
Encouraging team members to participate in brainstorming sessions and workshops cultivates a culture of collaboration and creativity, thereby improving problem-solving capabilities. This approach not only results in more comprehensive and user-centered but also aligns with the increasing trend of , projected to reach nearly $6 trillion by the end of 2026.
Navigating the intricate landscape of medical device development necessitates a strategic approach that encompasses regulatory compliance, quality standards, innovative engineering solutions, and effective collaboration. This article underscores that success in this field relies on a comprehensive understanding of these elements, especially as the industry braces for evolving regulations and intensified competition.
Key insights include:
As the medical device sector continues to evolve, manufacturers are urged to prioritize these essential strategies. By embracing regulatory compliance, leveraging advanced engineering practices, and nurturing collaboration, companies can not only enhance their product offerings but also position themselves for sustained success in a rapidly changing market. Taking proactive measures now will ensure that organizations are well-prepared to address the challenges and opportunities that lie ahead in medical device engineering.
What is the importance of understanding regulatory compliance in healthcare equipment?
Understanding regulatory compliance is crucial for navigating the healthcare equipment landscape, as it involves familiarity with the FDA's regulations, ISO standards, and Good Manufacturing Practices (GMP).
How do regulations vary in healthcare equipment?
Regulations can vary significantly based on the classification of the equipment, which is categorized into Class I, II, or III.
What is a Quality Management System (QMS) and why is it important?
A Quality Management System (QMS) is essential for ensuring consistent quality throughout the product lifecycle, and it should align with ISO 13485 standards.
What are the key components of maintaining compliance in healthcare equipment?
Key components include implementing a robust QMS, conducting regular audits, and holding training sessions to prepare for inspections.
What benefits can arise from integrating a QMS early in the development process?
Companies that integrate a QMS early can experience significant benefits, such as a reported 30% reduction in time to market due to fewer compliance-related delays.
What is the significance of the revised Quality Management System Regulation (QMSR) effective from February 2, 2026?
The revised QMSR emphasizes the incorporation of ISO 13485:2016, making adherence essential for success in the health product sector.
How can companies get assistance with regulatory compliance?
Companies can seek assistance from organizations like Voler Systems, which help manufacturers navigate regulatory complexities and ensure compliance with high-quality standards.
Can you provide an example of a successful compliance strategy?
A notable example involves a healthcare equipment startup that, with guidance from Voler Systems, achieved compliance ahead of schedule, significantly enhancing their market entry strategy.
