What is a Prototype Design Engineer? Role, Skills, and Importance
Discover the critical role and skills of a prototype design engineer in product...

The integration of 3D printing technology into the medical device sector is not merely a trend; it signifies a fundamental shift towards more personalized and efficient healthcare solutions. This article explores best practices for developing 3D printed prototypes, highlighting critical aspects such as:
These elements can significantly enhance product quality and expedite time to market. However, navigating the complexities of these practices raises an important question: How can manufacturers effectively balance innovation with the stringent requirements of safety and efficacy in medical device development?
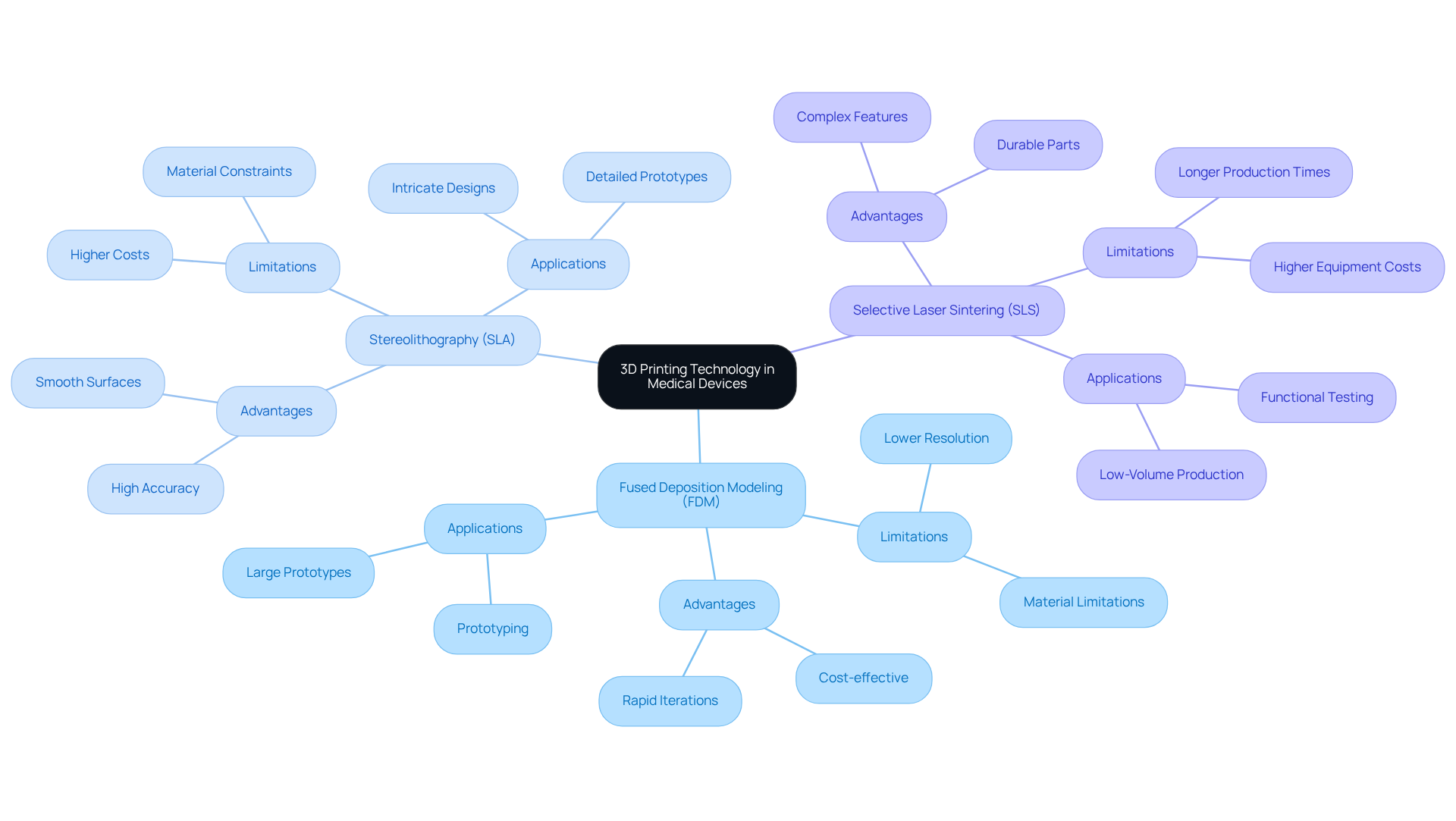
3D printing, also known as additive manufacturing, is revolutionizing the healthcare equipment sector by facilitating the creation of complex shapes and customized solutions. At , our emphasizes the optimization of hardware development and the implementation of modifications crucial for . A thorough understanding of various - (FDM), Stereolithography (SLA), and (SLS) - is vital for selecting the most appropriate approach for your prototype. Each method offers distinct advantages and limitations that influence material compatibility, resolution, and production speed.
For instance, SLA is renowned for its exceptional accuracy and smooth surfaces, making it ideal for intricate designs that require high detail. Conversely, FDM provides a more accessible and cost-effective solution for larger prototypes, enabling rapid iterations and adjustments. SLS is particularly effective in producing durable parts with complex features, making it suitable for and low-volume production.
Familiarity with these technologies equips engineers to optimize designs for both functionality and manufacturability, ultimately improving patient outcomes and expediting time-to-market. Effective applications of these techniques include the development of and patient-specific implants, underscoring the transformative impact of 3D printing in healthcare product prototyping. As the industry continues to advance, leveraging these sophisticated manufacturing techniques will be essential to meet the growing demand for personalized health solutions.
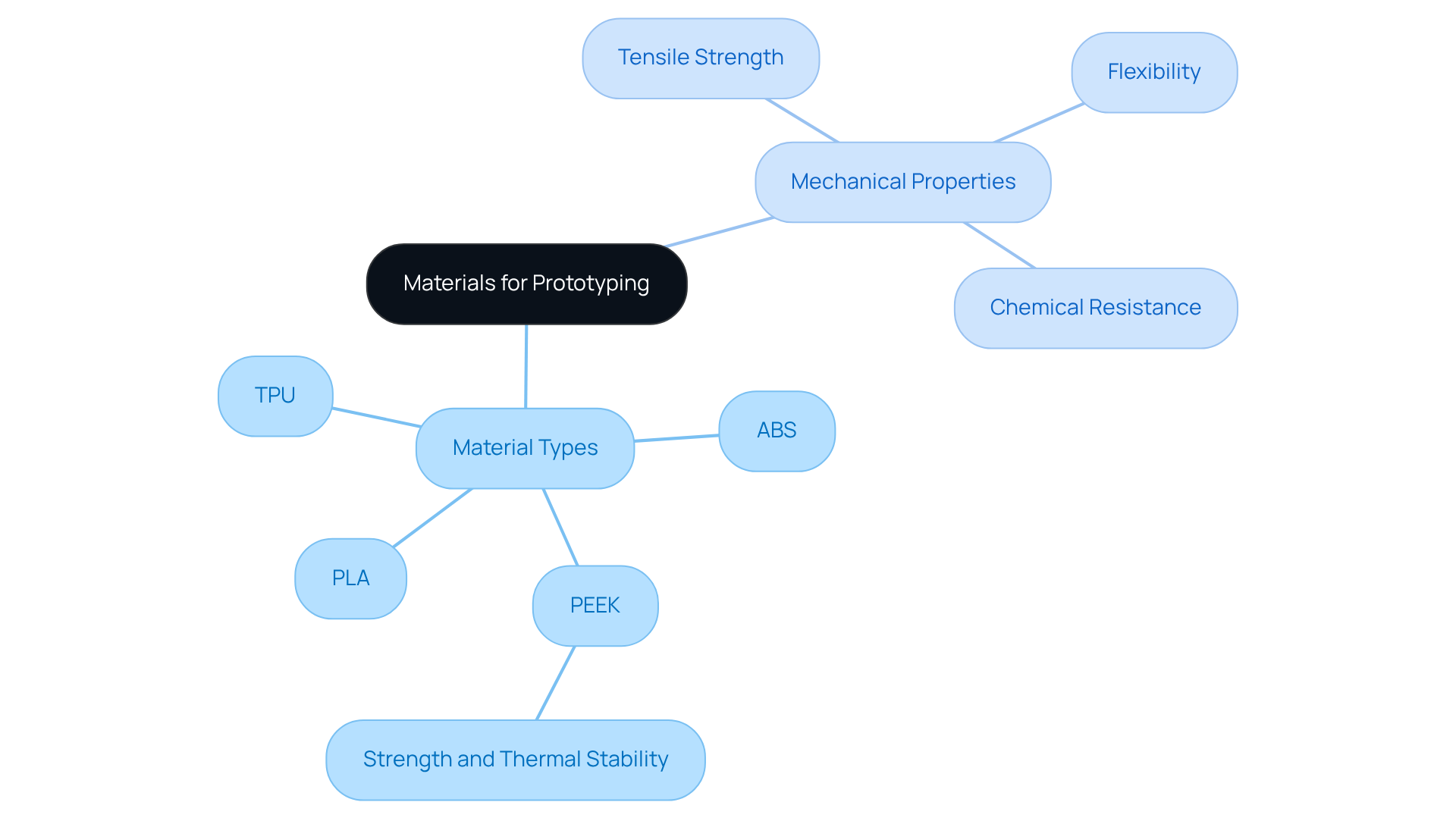
Selecting appropriate materials for is essential. These . Commonly used materials include:
For example, PEEK is particularly valued for its strength and thermal stability, making it suitable for load-bearing applications.
Moreover, understanding the -such as tensile strength, flexibility, and chemical resistance-is vital for ensuring that prototypes can endure real-world conditions. Collaborating with material suppliers and utilizing resources like the FDA's guidelines on material selection can streamline this process, ensuring that prototypes are not only functional but also compliant with .
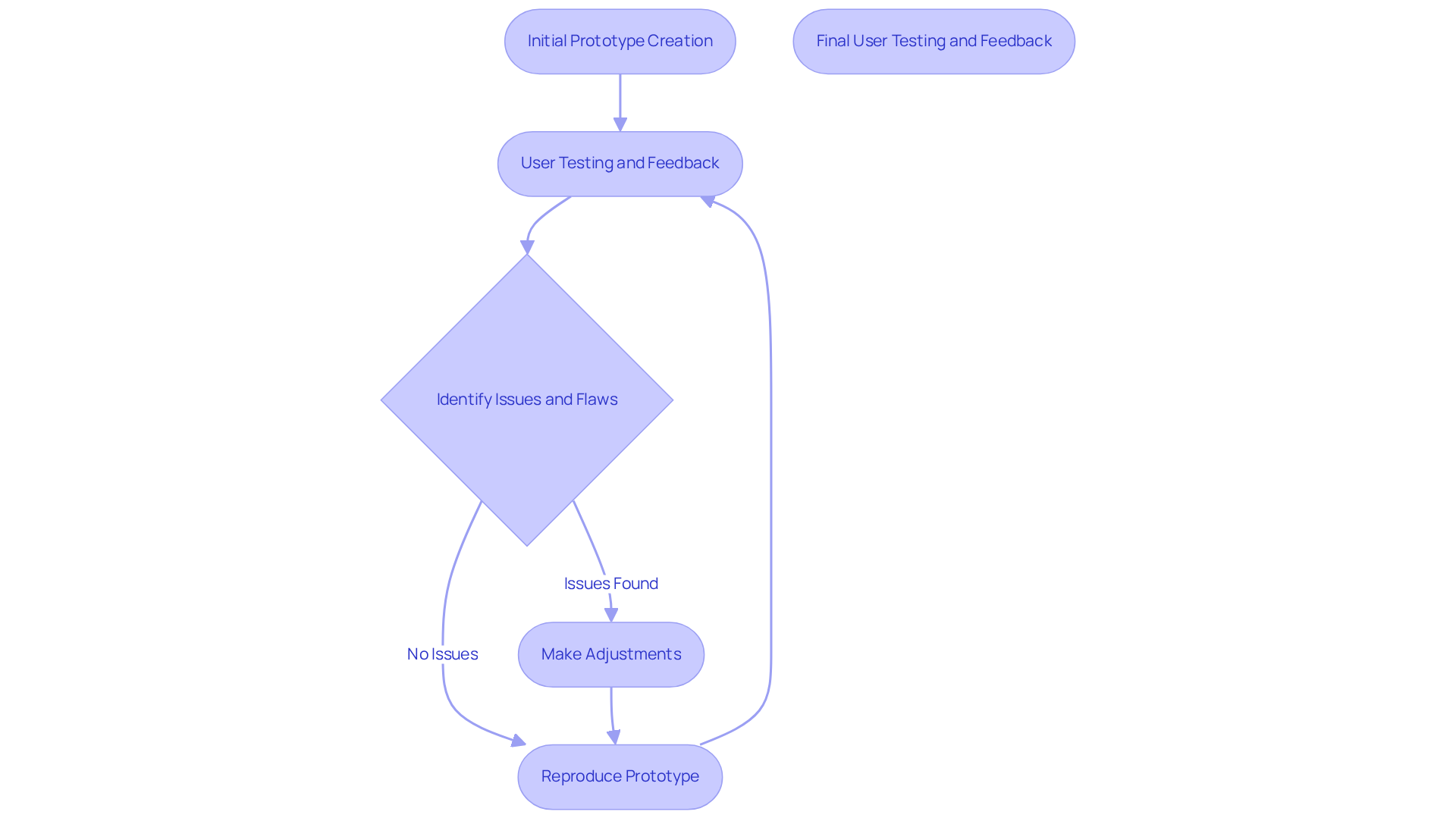
Iterative modeling is a critical process in the development of , characterized by the creation of multiple versions that undergo rigorous testing and user feedback. This approach allows teams to identify structural flaws and usability issues early, facilitating timely adjustments prior to final production. For example, enable teams to quickly produce and evaluate , gathering vital data on user interactions and device performance. This feedback loop is crucial for refining designs to meet user needs and comply with regulatory standards. By integrating during this phase, prototypes can be improved to ensure that the final product is both intuitive and safe for end-users.
Case studies illustrate that companies utilizing have significantly reduced development time and costs while simultaneously enhancing product quality. For instance, the ONIRY diagnostic instrument benefited from a strategic development approach that prioritized healthcare precision and user comfort, ultimately resulting in a clinically reliable tool that could be effectively scaled. Similarly, the innovative calf-worn device designed for motion and circumference monitoring in knee replacement rehabilitation exemplifies how thoughtful design can promote adherence to standards while enhancing user experience. User testimonials regarding these devices further validate the effectiveness of ' methodology, underscoring and compliance.
Investing in can mitigate project risks, with intelligent design strategies potentially reducing overall expenses by 20-30%. Engaging regulators early in the process ensures compliance with stringent industry standards, further enhancing the efficiency of the development phase. ' expertise in and the creation of positions them as a leader in addressing these challenges, ensuring that products not only meet regulatory requirements but also exceed user expectations.
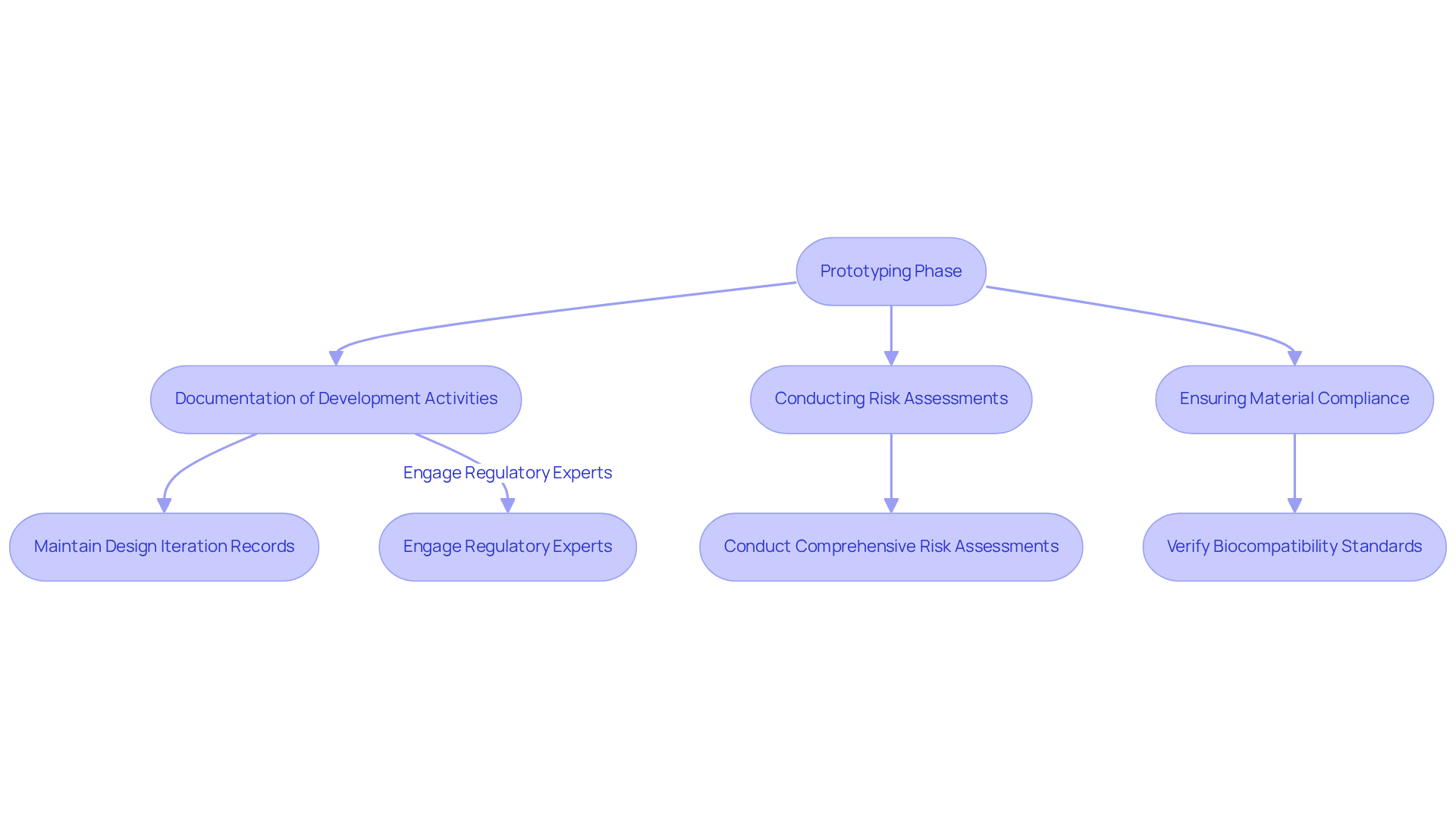
is essential during the initial phase of medical device development. Adhering to guidelines from the , particularly , is vital for ensuring that prototypes meet safety and efficacy requirements. This process involves:
Engaging regulatory experts early in the prototyping process can help identify potential compliance issues and streamline the approval pathway. For instance, maintaining detailed records of design iterations and testing outcomes can significantly facilitate the submission process for or .
By prioritizing from the outset, companies can effectively mitigate risks and enhance their chances of successful market entry.
The integration of 3D printing technology in medical device prototyping marks a significant advancement in the healthcare sector, facilitating the creation of customized and efficient solutions tailored to patient needs. By comprehensively understanding the various 3D printing methods and their specific advantages, engineers can optimize designs that enhance functionality and improve patient outcomes.
Key practices such as selecting appropriate materials, implementing iterative prototyping, and ensuring regulatory compliance are critical for successful medical device development. The use of biocompatible and durable materials, a feedback-driven design process, and adherence to industry regulations can streamline the prototyping phase and mitigate associated risks. These practices not only enhance product quality but also ensure that devices meet established safety and efficacy standards.
As the landscape of medical device development continues to evolve, embracing these best practices will be essential for innovators. By prioritizing advanced technologies and methodologies, the medical industry can better address the growing demand for personalized health solutions, ultimately leading to improved patient care and outcomes. The future of healthcare equipment rests with those who effectively leverage 3D printing, ensuring that the next generation of medical devices is both innovative and compliant with the highest standards.
What is 3D printing technology in the context of medical devices?
3D printing, or additive manufacturing, is transforming the healthcare equipment sector by enabling the creation of complex shapes and customized solutions for medical devices.
What are the key 3D printing methods used in medical device manufacturing?
The key 3D printing methods include Fused Deposition Modeling (FDM), Stereolithography (SLA), and Selective Laser Sintering (SLS).
What are the advantages of Stereolithography (SLA)?
SLA is known for its exceptional accuracy and smooth surfaces, making it ideal for intricate designs that require high detail.
What are the benefits of Fused Deposition Modeling (FDM)?
FDM is a more accessible and cost-effective solution for larger prototypes, allowing for rapid iterations and adjustments.
When is Selective Laser Sintering (SLS) most effective?
SLS is particularly effective for producing durable parts with complex features, making it suitable for functional testing and low-volume production.
How does understanding 3D printing methods benefit engineers in medical device development?
Familiarity with these technologies helps engineers optimize designs for functionality and manufacturability, improving patient outcomes and expediting time-to-market.
What are some effective applications of 3D printing in healthcare?
Effective applications include the development of customized surgical instruments and patient-specific implants, highlighting the transformative impact of 3D printing in healthcare product prototyping.
Why is leveraging 3D printing techniques important for the healthcare industry?
Leveraging these advanced manufacturing techniques is essential to meet the growing demand for personalized health solutions as the industry continues to advance.
