4 Best Practices for 3D Printed Prototypes in Medical Devices
Explore best practices for creating effective 3D printed prototypes in medical device...

Transitioning from prototype to product in the medical device industry represents a pivotal phase that can significantly influence the success or failure of a new technology. Given the high stakes and the ever-evolving regulatory landscape, stakeholders must adeptly navigate a complex array of objectives, compliance requirements, and collaborative efforts. This article examines essential best practices that not only streamline the transition process but also enhance product quality and market readiness.
What are the key strategies that can effectively bridge the gap between a promising prototype and a compliant, market-ready medical device?
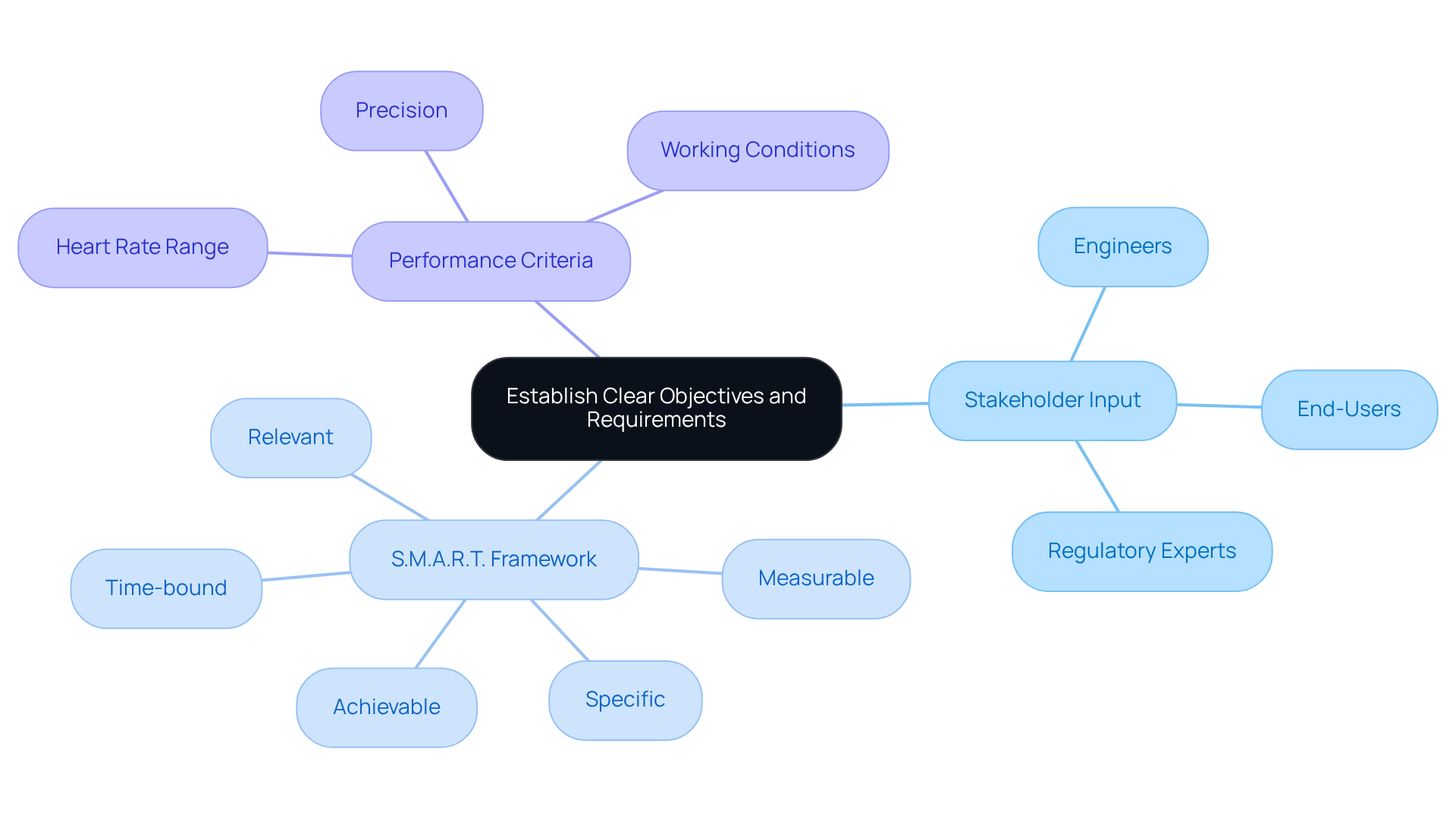
To transition effectively from prototype to product, establishing from the outset is crucial. This process involves gathering input from all stakeholders, including engineers, regulatory experts, and end-users, to define the , , and .
Employing frameworks such as S.M.A.R.T. (Specific, Measurable, Achievable, Relevant, Time-bound) can significantly enhance the formulation of these objectives. For example, a healthcare instrument designed for heart rate tracking should specify:
This clarity not only but also serves as a benchmark for validation and testing, ultimately improving project success rates.
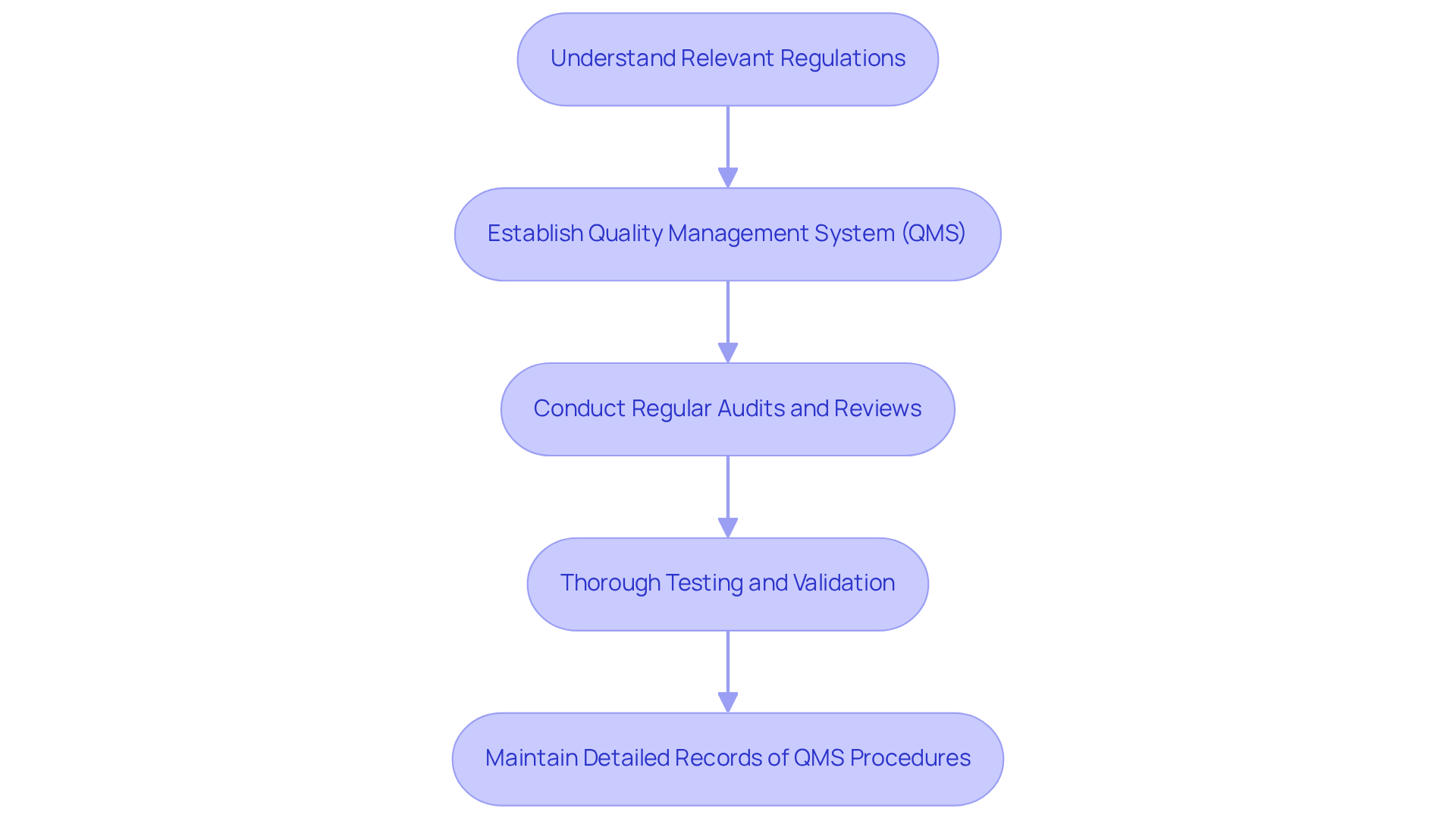
To ensure that meet essential safety and efficacy standards, it is crucial to integrate and quality control into the development framework. This process begins with a thorough understanding of relevant regulations, including those established by the FDA and ISO standards. Establishing a robust aligned with simplifies by creating clear protocols for development control, documentation, and risk management.
Regular audits and reviews are vital to ensure adherence to these protocols. For instance, when transitioning a prototype for a wearable health monitor, thorough testing and validation, along with maintaining detailed records of QMS procedures, are essential to demonstrate compliance with relevant standards. This proactive approach not only facilitates a smoother path to market but also enhances and manufacturer credibility, aligning with the principles of outlined in .
The implementation of a QMS is a continuous endeavor focused on achieving excellence and guaranteeing . This illustrates the essential steps required for successful in healthcare instruments.
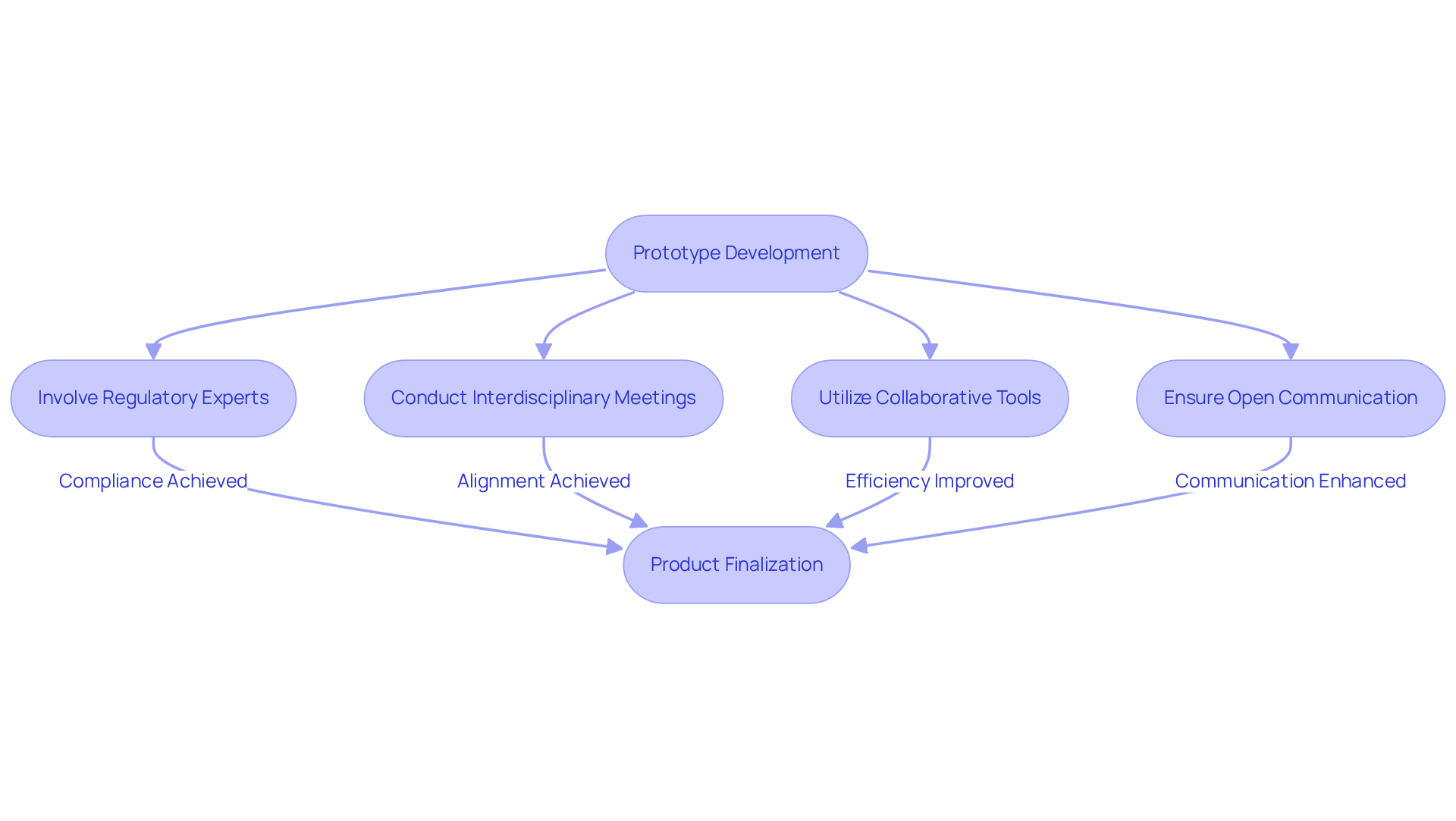
Effective collaboration among diverse teams is essential for successfully in the . Establishing an environment where engineers, regulatory experts, and marketing professionals can freely exchange insights and feedback during the development phase is vital. Regular promote , ensuring that all team members stay aligned with project objectives.
For instance, involving regulatory experts early in the design phase can help before they develop into significant challenges. Furthermore, employing enhances transparency and streamlines workflows, ultimately leading to a more efficient development cycle. This approach not only accelerates timelines but also and market readiness of .
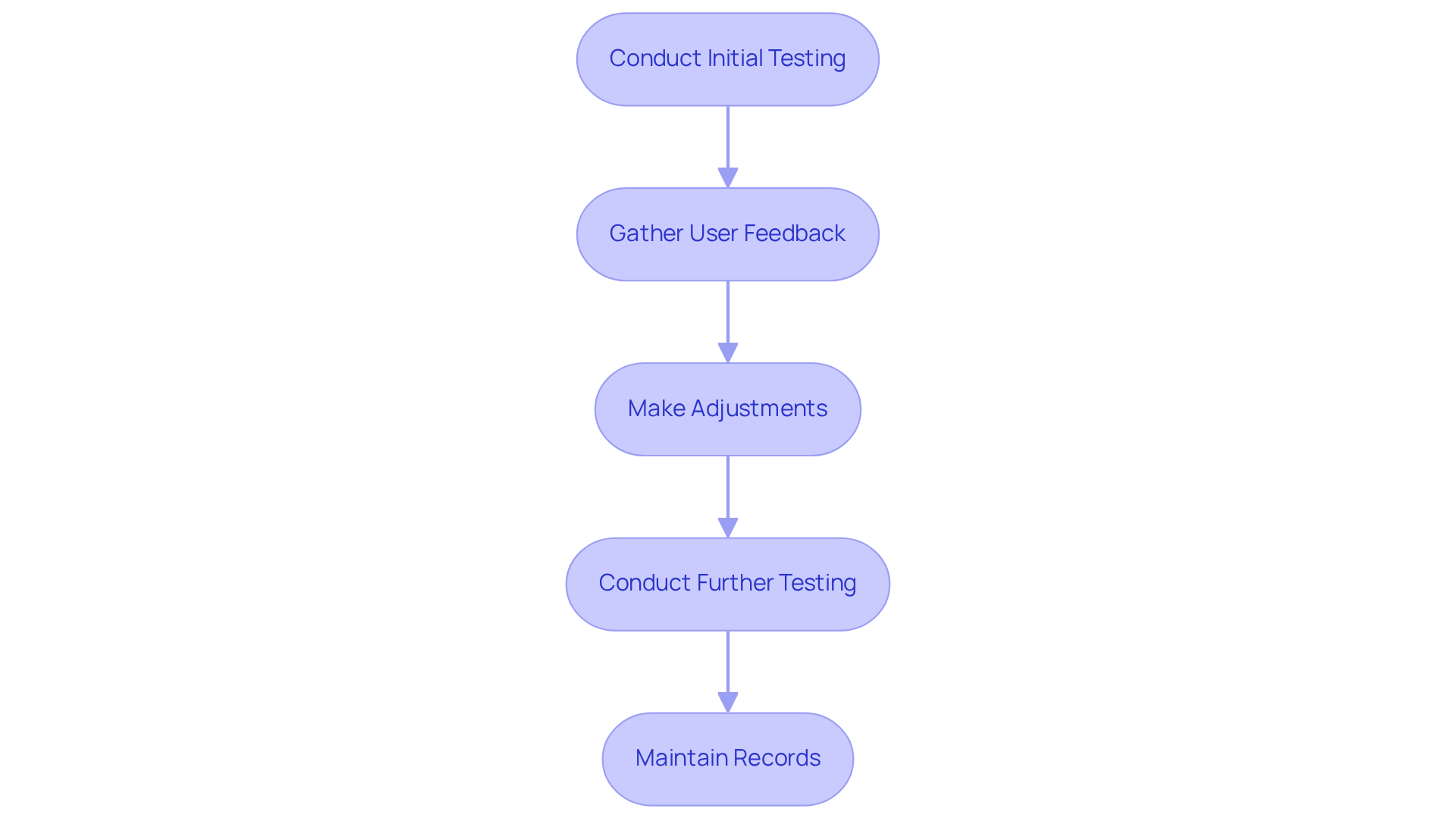
To ensure that meet the highest safety and effectiveness standards, it is essential to implement . This approach involves conducting multiple rounds of testing at various stages, which allows for feedback and adjustments based on real-world performance.
For example, a prototype to product of a new insulin delivery device should undergo usability testing with actual users to gather insights regarding its functionality and ease of use. This real-world feedback is crucial; as , achieving the correct layout necessitates a thorough investigation of the product's functional requirements and users' needs.
By integrating this feedback into subsequent iterations, developers can refine the design to better align with user needs and . Furthermore, maintaining and facilitating future audits, ensuring that the product adheres to both market standards and regulatory obligations.
offers tailored , utilizing established methodologies to support health technology startups in effectively navigating .
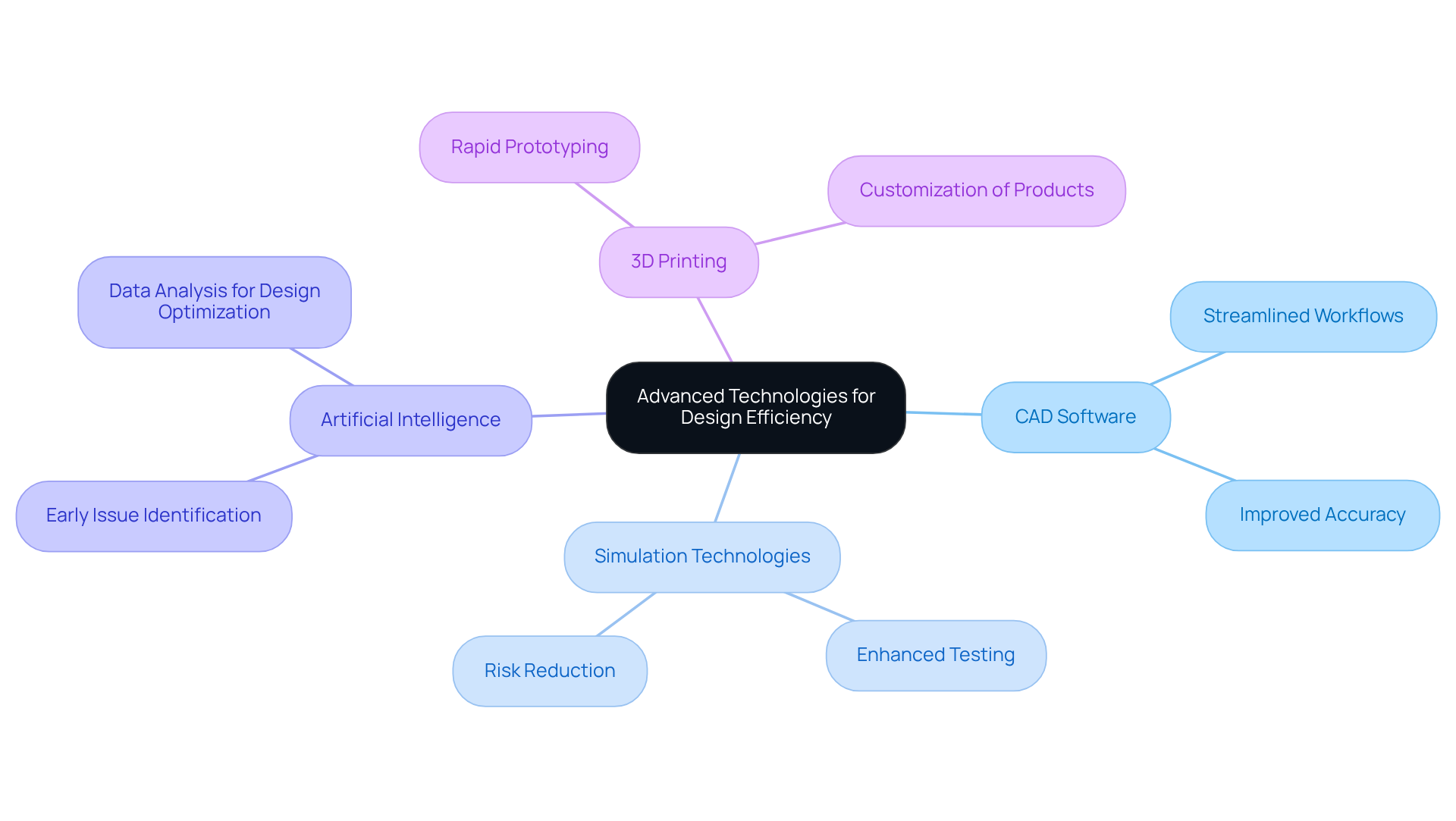
Integrating into the significantly enhances efficiency and innovation. Tools such as computer-aided drafting (CAD) software, simulation technologies, and (AI) streamline workflows and improve accuracy. For instance, AI algorithms can analyze design data to identify potential issues early in development, allowing for quicker adjustments.
Furthermore, the incorporation of accelerates the by enabling , which permits teams to produce and evaluate multiple versions of a product in a fraction of the time required by conventional methods. This approach not only reduces development timelines but also and reliability of .
As the medical device industry encounters increasing , leveraging these advanced tools becomes essential for maintaining competitiveness and ensuring compliance with industry standards.
Transitioning from prototype to product in the medical device industry is a multifaceted process that requires meticulous planning and execution. Establishing clear objectives, ensuring regulatory compliance, fostering collaboration, implementing iterative testing, and leveraging advanced technologies are essential strategies that enable organizations to navigate this complex landscape effectively. Each of these elements plays a pivotal role in transforming innovative ideas into market-ready solutions that prioritize patient safety and efficacy.
The article highlights several key strategies crucial for a successful transition:
Ultimately, the importance of these best practices cannot be overstated. As the medical device industry continues to evolve, embracing these strategies will not only facilitate smoother transitions from prototype to product but also contribute to the overall advancement of healthcare solutions. Stakeholders are encouraged to adopt these best practices to enhance their development workflows and maintain competitiveness in a rapidly changing market.
Why is it important to establish clear objectives and requirements when transitioning from prototype to product?
Establishing clear objectives and requirements is crucial as it involves gathering input from all stakeholders to define the product's intended use, performance criteria, and compliance requirements, which ultimately guides the development process and improves project success rates.
What framework can be used to enhance the formulation of objectives for product development?
The S.M.A.R.T. framework (Specific, Measurable, Achievable, Relevant, Time-bound) can significantly enhance the formulation of objectives by providing a structured approach to defining clear and actionable goals.
What are some examples of specific objectives for a healthcare instrument designed for heart rate tracking?
Specific objectives for such a healthcare instrument might include defining the necessary precision, the range of heart rates it must assess, and the working conditions under which it operates.
How does regulatory compliance factor into the development of medical devices?
Regulatory compliance is essential to ensure that medical devices meet safety and efficacy standards. It involves understanding relevant regulations, such as those from the FDA and ISO standards, and integrating them into the development framework.
What is a Quality Management System (QMS) and why is it important?
A Quality Management System (QMS) is a structured system that outlines protocols for development control, documentation, and risk management. It is important because it simplifies compliance initiatives and ensures adherence to safety and efficacy standards.
What role do audits and reviews play in maintaining regulatory compliance?
Regular audits and reviews are vital for ensuring adherence to QMS protocols, facilitating thorough testing and validation, and maintaining detailed records, which are essential for demonstrating compliance with relevant standards.
How does implementing a QMS contribute to patient safety and manufacturer credibility?
Implementing a QMS is a continuous endeavor that focuses on achieving excellence and guaranteeing patient safety, which enhances manufacturer credibility and aligns with the principles of continuous improvement in healthcare instrument design.
