Why Medical Device Manufacturers Need Industrial Product Design Consultants
Introduction Manufacturers face significant challenges in navigating complex regulatory...

A Product Requirements Document (PRD) is fundamental to successful product development, especially within the highly regulated medical device sector. It clearly delineates essential features, functionalities, and constraints, thereby fostering collaboration among stakeholders and aligning teams around a unified vision.
However, as Agile methodologies evolve, organizations face a critical challenge: how can they adapt their PRDs to remain flexible and responsive to user needs while ensuring compliance and effectiveness?
This exploration will illuminate the vital role PRDs play in navigating the complexities of product development.
A (PRD) is that serves as a formal blueprint outlining the essential features, functionalities, and constraints of a product. It acts as a fundamental guide for the development team, clearly articulating and the goals and requirements that must be met. Typically, outlines details such as the , target audience, and specific functionalities, fostering a shared understanding among all stakeholders regarding project objectives.
In the context of medical devices, particularly , understanding is crucial for compliance with regulatory standards, ensuring safety, and validating the effectiveness of the product being developed. For example, a PRD for a wearable health device may specify requirements for real-time monitoring capabilities, user interface design, and data security measures, all of which are essential for .
Furthermore, the PRD must clearly delineate system and environmental requirements, usability needs, and any assumptions or constraints that could impact the development process. Given the complexities involved in medical device creation, a significant number of firms - over 70% - utilize to streamline their , mitigate risks, and enhance collaboration among teams.
At Voler Systems, our expertise in , particularly in and , positions us to assist in developing precise specifications that adhere to industry standards and enhance performance.
In the realm of creation, (PRD) plays a pivotal role in aligning all stakeholders around a unified and a . Serving as a singular source of truth, these documents guide teams through the complexities of the , significantly reducing the likelihood of misunderstandings. By clearly articulating objectives and requirements, teams can understand , which empowers them to prioritize features effectively, allocate resources wisely, and adhere to . Notably, projects that utilize well-organized PRDs are 40% more likely to be completed on schedule and within budget.
Furthermore, in the highly regulated sector, knowing is essential for . This not only streamlines the regulatory approval process but also enhances the overall quality of the product. Effective incorporate frameworks for , ensuring that healthcare solutions are both technically sound and clinically effective. As a result, requirement documents serve as vital tools for managers and engineers, .
The evolution of (PRD) has been significant, particularly with the rise of . Traditionally, requirement documents were extensive texts rooted in the Waterfall method, detailing every aspect of a product before its development. However, the transition to Agile has fundamentally altered this approach. Modern product requirement documents, which are known for their flexibility and iterative nature, allow for continuous updates and modifications throughout the . This adaptability is especially critical in the healthcare sector, where is essential.
Statistics reveal that 86% of organizations now employ , which has notably influenced PRD creation. for , prioritizing essential user data and insights over exhaustive specifications. For instance, in the development of , Agile modifications have resulted in and , ensuring that products not only meet stringent industry standards but also closely align with user needs. This evolution underscores a broader trend in management, highlighting the and responsiveness in understanding and crafting .
An effective Product Requirements Document (PRD) for includes several :
Integrating these elements into a raises the question of what is , as it not only promotes alignment and clarity throughout the lifecycle but also increases the likelihood of successful development. Statistics indicate that products developed with detailed s exhibit significantly higher success rates, highlighting what is in emphasizing the importance of meticulous documentation in the healthcare sector.
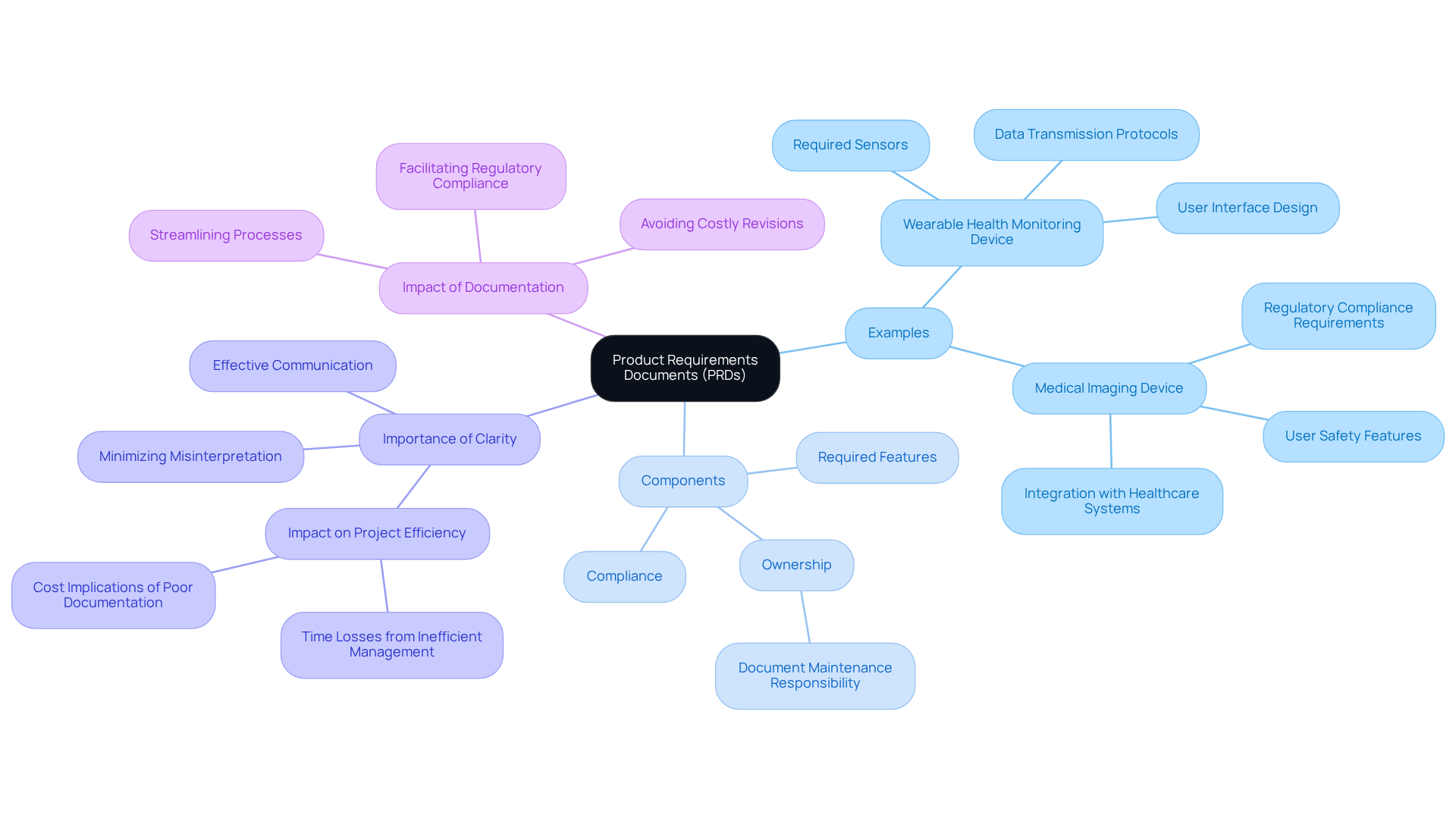
Real-world examples of what is (PRDs) are prevalent across various industries, particularly in technology and healthcare. For instance, a PRD for a wearable , such as the innovative calf-worn device designed for motion and circumference monitoring in knee replacement rehabilitation, may include sections detailing the required sensors, data transmission protocols, and user interface design. This device exemplifies the application of PRDs and underscores the importance of adhering to , an area where Voler Systems excels in supporting startups within the medical technology sector.
Another pertinent example is a PRD for a new , which would delineate , , and integration with existing healthcare systems. These instances illustrate what a is by showcasing the variability in intricacy and specificity, tailored to meet the unique demands of each product and its target market, while fulfilling the essential role of guiding progress and ensuring .
A critical component of PRDs is the designation of an individual responsible for maintaining the document's accuracy and relevance throughout the creation process. This ownership ensures that the PRD remains current with any changes or iterations that arise during the project lifecycle. Furthermore, the iterative nature of is vital; as projects evolve, so too must the requirements, reflecting new insights and stakeholder feedback.
Consistency in terminology within requirements documents is crucial for minimizing misinterpretation and ensuring clarity among all stakeholders involved. This practice facilitates effective communication and understanding across various teams, from engineering to regulatory affairs.
Moreover, the potential time losses due to inefficient document management highlight the practical implications of well-structured PRDs. Research indicates that inadequate documentation methods can lead to significant delays, with projects potentially wasting hundreds of hours that could otherwise be allocated to productive tasks. By implementing a comprehensive PRD, teams can streamline their processes and avoid costly revisions later in the project cycle.
Ultimately, the variability in what is and detail reflects the specific needs of each product and its target market. However, the fundamental purpose remains unchanged: to provide a clear roadmap for development, ensuring that all parties involved comprehend the expected functionalities and performance criteria, particularly in the context of designed with compliance in mind.
A Product Requirements Document (PRD) is a crucial blueprint for product development, especially in the medical device sector. By clearly defining the features, functionalities, and constraints of a product, PRDs promote alignment among stakeholders and guide teams through the complexities of the development process. Their importance is paramount, as they not only enhance collaboration but also ensure compliance with regulatory standards, ultimately leading to successful product outcomes.
This article highlights key insights regarding the structure and significance of PRDs. It covers essential components such as executive summaries and user personas, while also addressing the evolution of PRDs within Agile methodologies. Effective documentation is vital for meeting both user needs and industry standards. Supporting statistics reinforce the idea that well-organized PRDs significantly increase the likelihood of projects being completed on time and within budget.
Given the complexities involved in product development, particularly in healthcare, the imperative is clear: prioritize the creation and maintenance of comprehensive Product Requirements Documents. By doing so, organizations can streamline their processes, enhance communication, and ultimately drive innovation, ensuring that products meet the highest standards of safety and efficacy. Embracing the principles of effective PRD development is not merely advantageous; it is essential for success in an increasingly competitive landscape.
What is a Product Requirements Document (PRD)?
A Product Requirements Document (PRD) is a formal blueprint that outlines the essential features, functionalities, and constraints of a product, serving as a guide for the development team and articulating the goals and requirements that must be met.
Why is a PRD important in the context of medical devices?
In the context of medical devices, especially wearable health technologies, a PRD is crucial for compliance with regulatory standards, ensuring safety, and validating the effectiveness of the product being developed.
What details are typically included in a PRD?
A PRD typically includes the product's purpose, target audience, specific functionalities, system and environmental requirements, usability needs, and any assumptions or constraints that may impact the development process.
How does using a PRD benefit product development?
Utilizing a well-organized PRD can significantly reduce misunderstandings among stakeholders, empower teams to prioritize features, allocate resources effectively, and adhere to project timelines. Projects with organized PRDs are 40% more likely to be completed on schedule and within budget.
How does a PRD contribute to regulatory compliance in medical device development?
A PRD helps demonstrate compliance with safety and efficacy standards, streamlining the regulatory approval process and enhancing the overall quality of the product. It incorporates frameworks for clinical validation, ensuring that healthcare solutions are both technically sound and clinically effective.
What role does Voler Systems play in relation to PRDs?
Voler Systems specializes in advanced electronic device design, particularly in AI-powered engineering and ultra-low energy solutions, and assists medical device manufacturers in developing precise specifications that adhere to industry standards and enhance performance.
