Introduction
Navigating the complex landscape of medical device manufacturing presents a distinct set of challenges that can significantly influence product development timelines and success rates. As the industry contends with stringent regulatory requirements, technological advancements, and shifting market demands, manufacturers must not only meet compliance standards but also ensure cost-effectiveness and high-quality outputs.
How can companies effectively balance these competing priorities while accelerating the development of innovative healthcare solutions? This article explores best practices for mastering industrial manufacturing product design in the medical device sector, providing insights into overcoming obstacles and optimizing processes for success.
Understand Industry-Specific Challenges in Medical Device Development
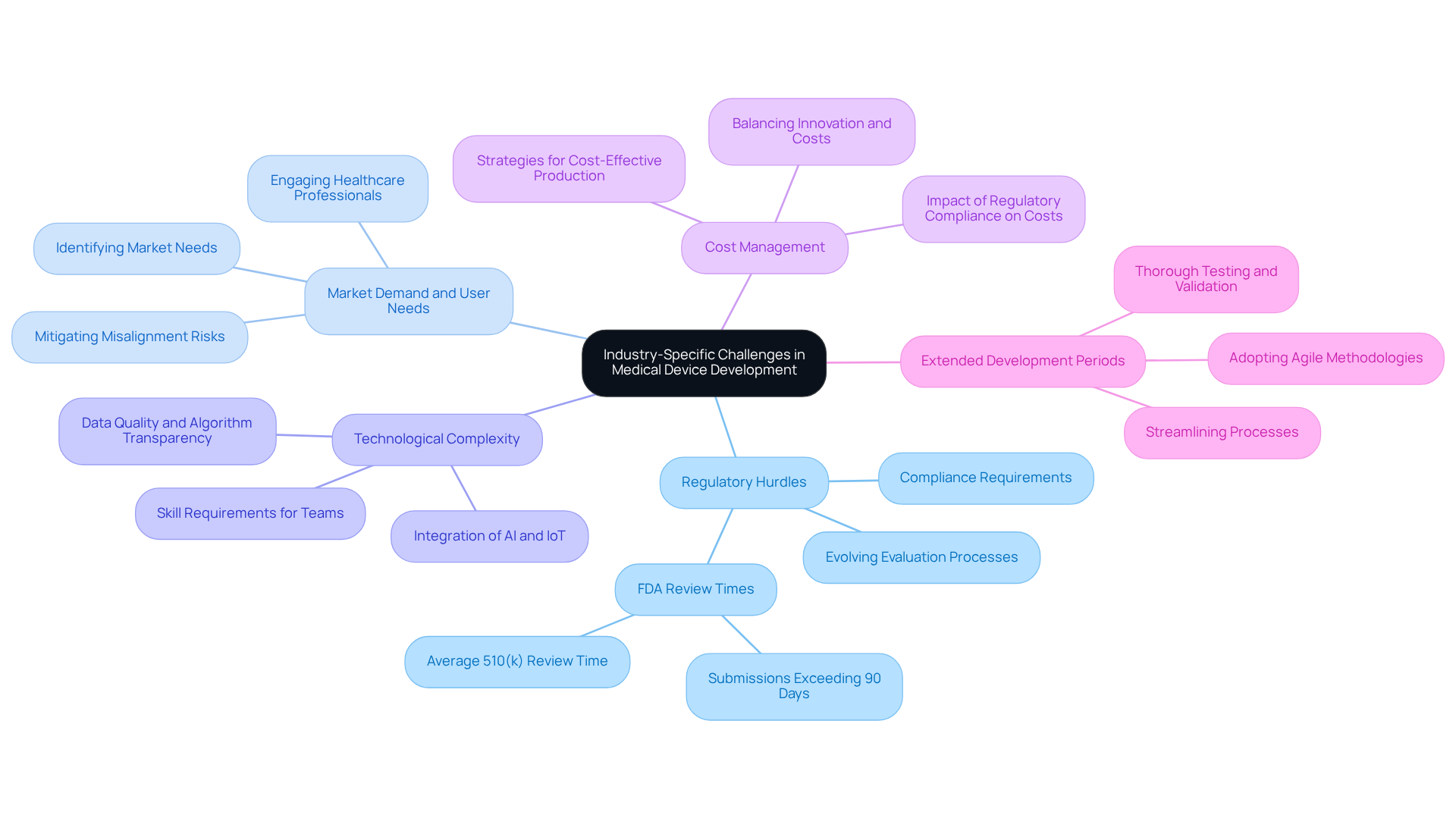
The medical equipment industry faces numerous challenges that significantly impact and success rates. Among these, are a primary concern:
- : Compliance with stringent regulations from bodies such as the FDA and EMA is essential. The FDA's evolving evaluation processes for software-driven and AI-enabled devices add complexity, often necessitating extensive evidence that can delay timelines. In 2025, FDA 510(k) review times averaged between 140 to 175 days, with many submissions exceeding the 90-day target. This regulatory pressure requires careful planning and adherence to to avoid costly delays.
- : Accurately identifying and validating market needs can be time-consuming. Engaging healthcare professionals and potential users early in the design process is crucial to mitigate risks associated with misaligned product features and market expectations.
- : The integration of advanced technologies, such as AI and IoT, introduces additional layers of complexity in device design. Manufacturers must ensure their teams possess the necessary skills and knowledge to navigate these innovations effectively. As AI becomes increasingly integrated into healthcare applications, the associated risks related to data quality and algorithm transparency must also be managed. excels in , leveraging FPGA and AI to create intelligent solutions that adapt to complex applications, ensuring seamless integration with sensors and actuators.
- : Balancing innovation with cost-effectiveness remains a constant challenge. Companies must adopt strategies that facilitate high-quality production without exceeding budget constraints, particularly in a landscape where regulatory compliance can drive costs higher. addresses this by enhancing system design and power management, especially in wireless health instruments, to prolong battery life and ensure dependable performance.
- Extended Development Periods: The creation of often spans multiple years, primarily due to the requirement for thorough testing and validation. Streamlining processes and adopting agile methodologies can help reduce these timelines, allowing for quicker market entry and responsiveness to changing demands. ' approach includes , which enhances efficiency and accelerates the development process.
By acknowledging and addressing these challenges, manufacturers can strengthen their teams and processes to effectively manage the intricacies of healthcare product development, with as a collaborator in innovative design and engineering solutions.
Implement Robust Regulatory Compliance Strategies
To effectively navigate the complex , manufacturers should adopt the following practices:
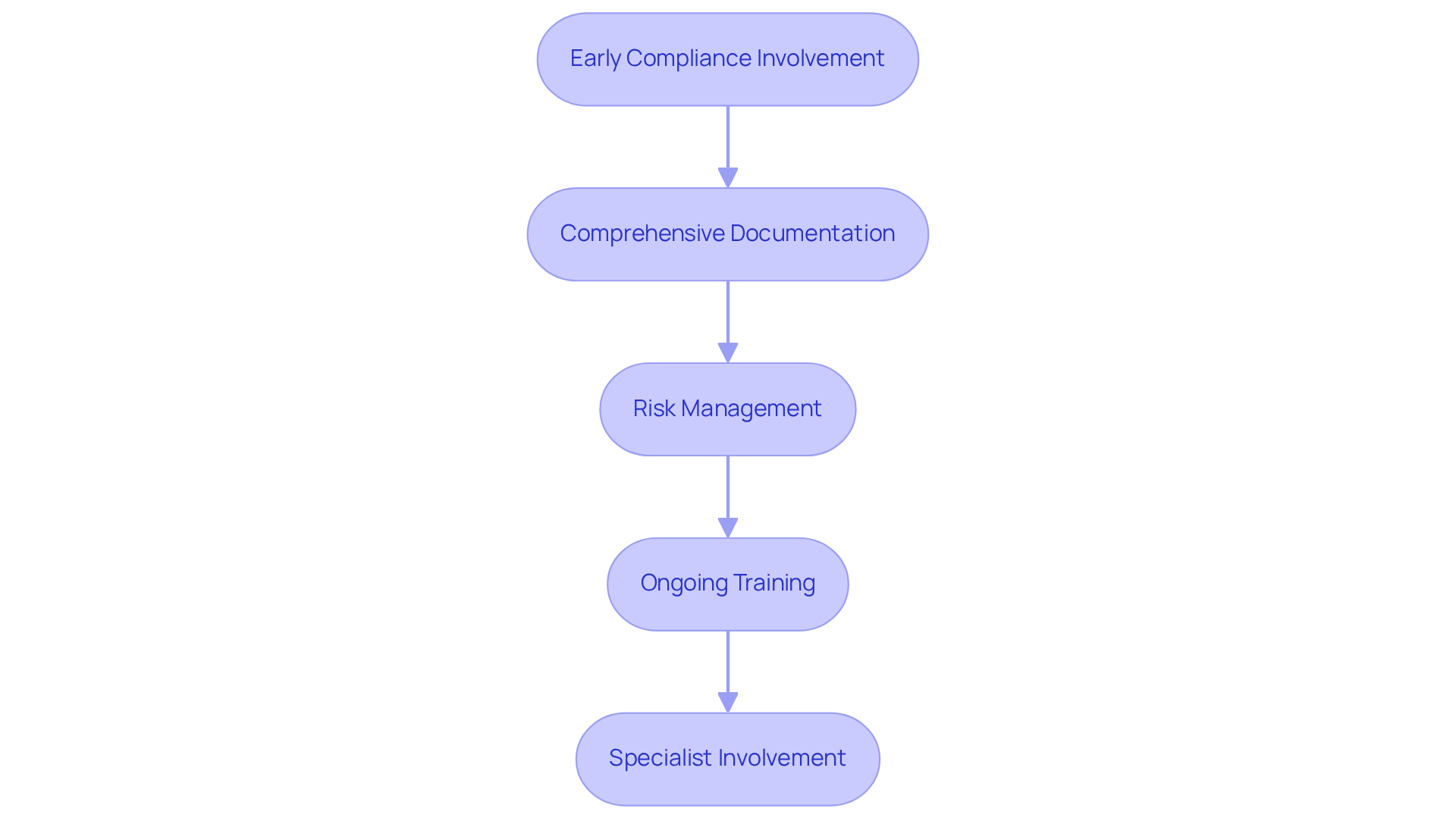
- Early Compliance Involvement: Engaging is essential. This proactive approach allows for the identification of relevant regulations and standards, significantly decreasing the chances of expensive redesigns later in development.
- Comprehensive Documentation: It is crucial to maintain thorough documentation throughout the development lifecycle. This encompasses design history files, risk management plans, and validation reports, all of which are vital for . , ensuring that all necessary documentation is adequately prepared.
- Risk Management: Implementing a robust in accordance with is imperative. This process includes identifying potential hazards, assessing associated risks, and establishing effective controls to mitigate them, thereby ensuring product safety and compliance.
- Ongoing Training: Regular training sessions for all team members regarding current are necessary. Keeping the team updated on policy changes cultivates a culture of compliance and readiness.
- Specialist Involvement: Employing specialists in compliance for healthcare devices can provide critical insights. Their expertise can streamline the approval process, assisting manufacturers in navigating complex regulations more efficiently. .
By adhering to these strategies and leveraging the support of Voler Systems, manufacturers can enhance their , reduce the risk of regulatory setbacks, and accelerate the introduction of safer products to the market.
Optimize Cost Management and Resource Allocation
To effectively manage costs and allocate resources in , consider the following strategies:
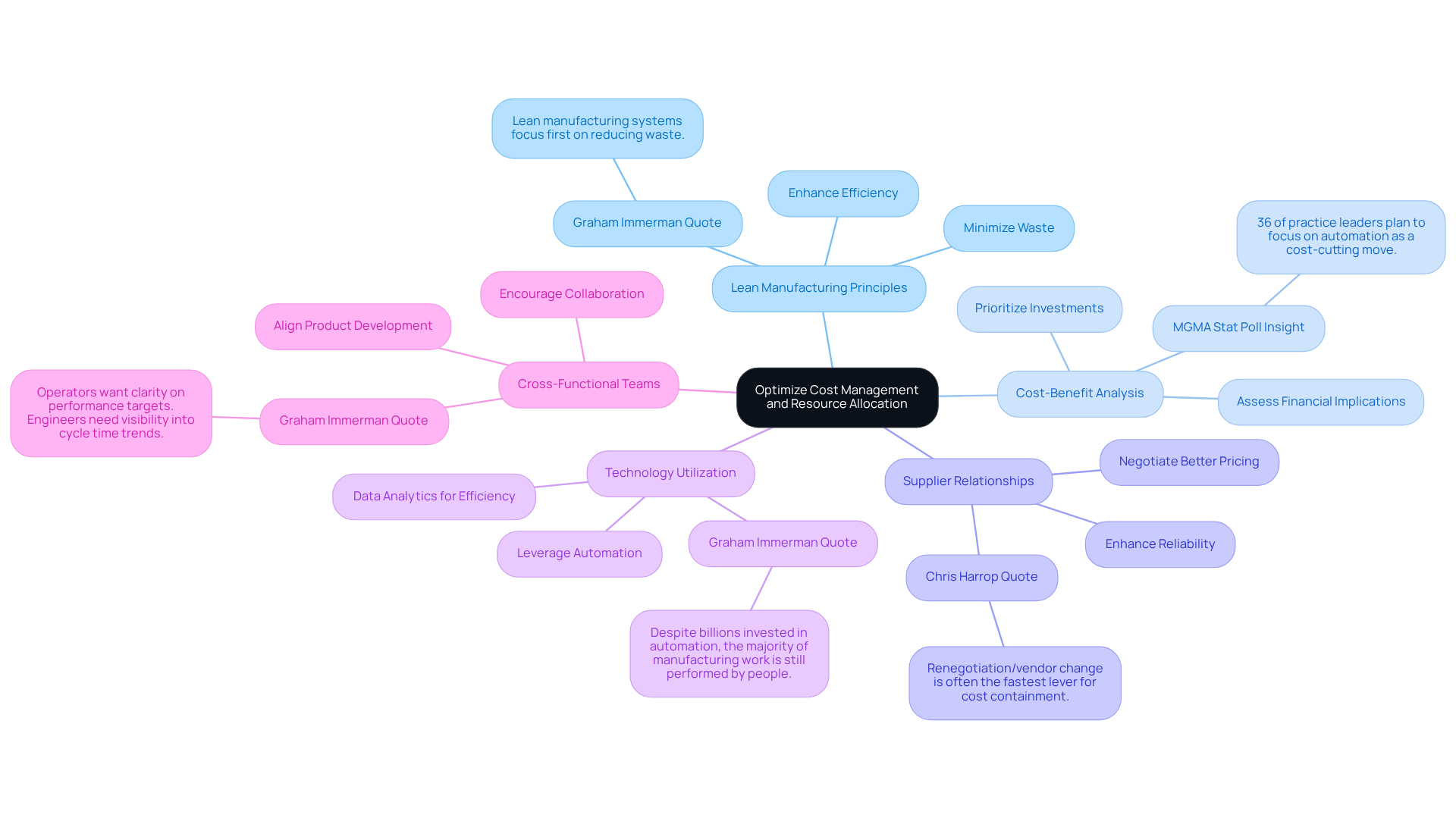
- : Implement lean manufacturing techniques to minimize waste and enhance efficiency. This involves streamlining processes, reducing excess inventory, and optimizing production workflows, which can significantly lower operational costs. As Graham Immerman notes, "Lean manufacturing systems focus first on reducing waste."
- : Regularly conduct cost-benefit analyses to assess the financial implications of design choices and production methods. This practice helps that yield the highest returns, ensuring effective resource allocation. A January 2026 MGMA Stat poll indicated that 36% of practice leaders plan to focus on automation as a cost-cutting move, underscoring the importance of informed decision-making in resource allocation.
- : Cultivate strong relationships with suppliers to negotiate better pricing and terms. Long-term collaborations can lead to significant cost reductions and enhanced reliability within the supply chain, which is essential in the competitive healthcare equipment market. Chris Harrop emphasizes that "renegotiation/vendor change is often the fastest lever for cost containment."
- : Leverage , such as automation and data analytics, to boost production efficiency and reduce labor costs. Investing in the right technology can yield substantial long-term savings and enhance overall productivity. The integration of machine monitoring systems can help eliminate waste and improve operational efficiency.
- : Encourage collaboration among engineering, marketing, and finance teams to align all aspects of product development with objectives. This holistic approach fosters informed decision-making and can lead to innovative solutions that balance quality and cost. Various case studies indicate that successful implementations of lean principles often involve cross-functional collaboration.
By applying these strategies, manufacturers can enhance their , ensuring competitiveness while providing high-quality healthcare products.
Establish Comprehensive Quality Assurance and Testing Protocols
To uphold the highest quality standards in medical device manufacturing, companies should implement the following , leveraging ' expertise:
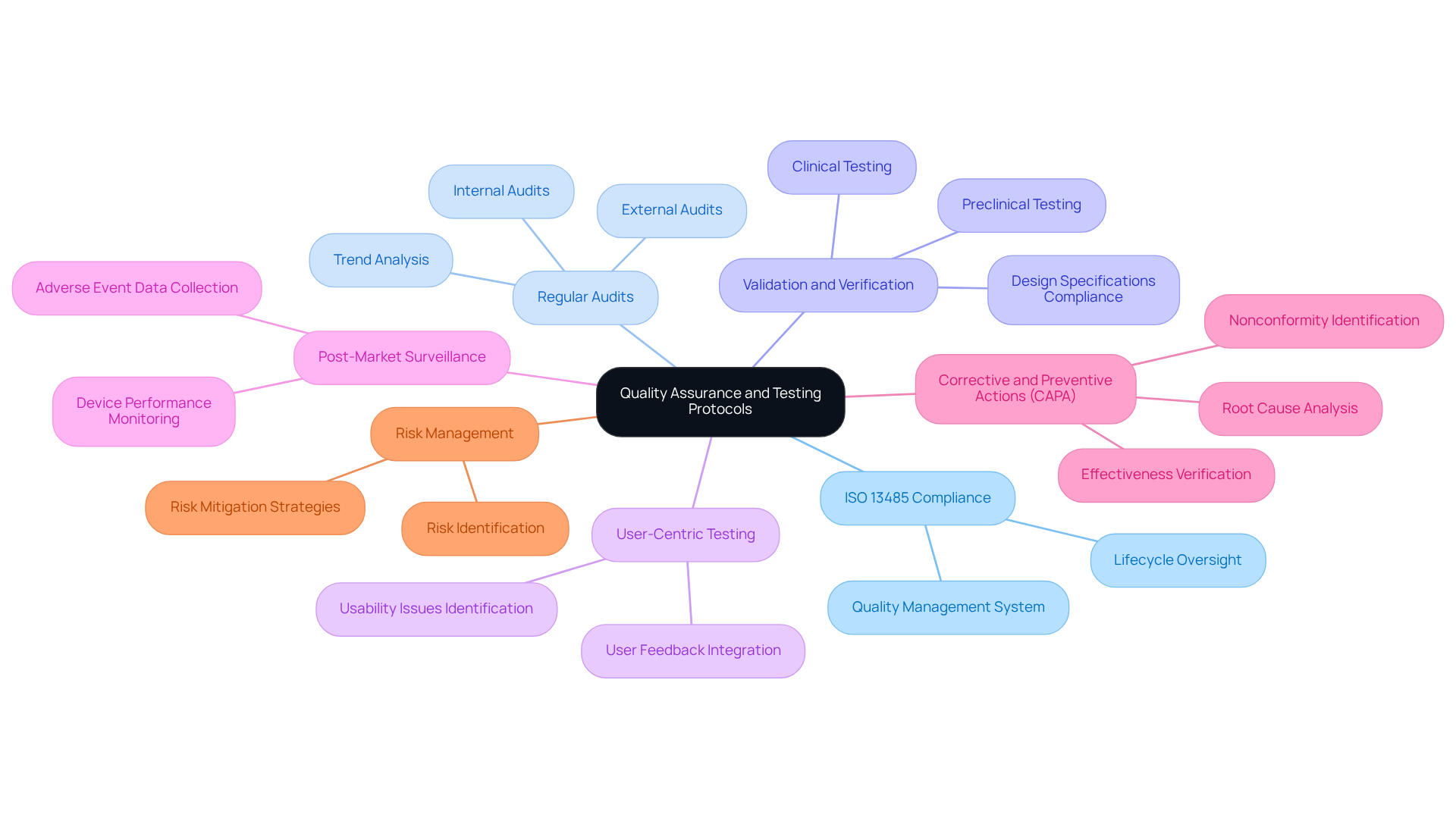
- : Establish a quality management system that adheres to ISO 13485 standards. This framework guarantees a systematic method to oversee quality throughout the item lifecycle, conforming to compliance expectations and boosting reliability.
- : Conduct both internal and external audits at defined intervals to evaluate compliance with quality standards and identify areas for improvement. are essential for maintaining accountability and fostering a culture of continuous improvement. Studies indicate that organizations with robust audit practices experience fewer nonconformities. Additionally, organizations should collect objective evidence of conformity and analyze trends to drive ongoing enhancements in their quality management systems.
- : Implement stringent to confirm that items meet design specifications and regulatory requirements. This includes comprehensive preclinical and clinical testing, which is vital for ensuring safety and efficacy before market release. Streamlining these processes can be supported by ' extensive experience in successful project implementations, as demonstrated in various case studies.
- : Integrate user feedback into the testing process to uncover usability issues and enhance the overall user experience. Involving end-users results in more intuitive designs and aligns offerings with real-world applications, ultimately enhancing patient satisfaction.
- : Develop a system to continuously monitor device performance and collect data on any adverse events. This ongoing evaluation is crucial for maintaining safety and efficacy, allowing manufacturers to respond proactively to any emerging issues.
- Corrective and Preventive Actions (CAPA): Establish a robust CAPA system to identify nonconformities, investigate root causes, and implement corrective actions. This proactive strategy is crucial for ensuring compliance and preventing recurrence of issues, thereby enhancing overall quality.
- Risk Management: Emphasize risk management throughout the item lifecycle, as highlighted by the . This involves integrating risk management practices into all stages of development and monitoring to ensure that potential risks are identified and mitigated effectively.
By establishing these comprehensive , including documentation compliance support, manufacturers can significantly enhance product quality, ensure regulatory compliance, and ultimately improve patient outcomes, all while benefiting from ' commitment to quality and timely delivery.
Conclusion
Mastering the complexities of industrial manufacturing product design for medical devices is crucial for success in a highly regulated and competitive landscape. Understanding the unique challenges of medical device development - ranging from regulatory compliance to cost management - is essential for manufacturers seeking to innovate and meet market demands. By implementing strategic practices, organizations can effectively navigate these complexities.
Key insights include:
- The necessity of engaging compliance experts early in the design process
- Maintaining comprehensive documentation
- Fostering cross-functional collaboration to optimize resource allocation
Furthermore, employing robust quality assurance protocols ensures that products not only meet regulatory standards but also enhance patient outcomes. Utilizing technologies and methodologies such as lean manufacturing and AI-enabled design can further streamline operations and drive efficiency.
In conclusion, embracing these best practices in medical device design transcends mere compliance; it fosters innovation and ensures that products can adapt to the evolving needs of healthcare professionals and patients alike. Manufacturers are encouraged to adopt a proactive approach, continuously refining their strategies and processes to maintain a competitive edge in this dynamic industry. By doing so, they not only mitigate risks but also position themselves as leaders in delivering safe, effective, and high-quality medical devices.
Frequently Asked Questions
What are the primary challenges in medical device development?
The primary challenges include regulatory hurdles, market demand and user needs, technological complexity, cost management, and extended development periods.
How do regulatory hurdles affect medical device development?
Compliance with strict regulations from bodies like the FDA and EMA is crucial. The FDA's evolving evaluation processes, especially for software-driven and AI-enabled devices, can lead to delays, with average review times in 2025 ranging from 140 to 175 days.
Why is understanding market demand important in medical device development?
Accurately identifying and validating market needs is essential to avoid misaligned product features and expectations. Engaging healthcare professionals early in the design process helps mitigate these risks.
What role does technological complexity play in medical device development?
The integration of advanced technologies, such as AI and IoT, adds complexity to device design. Manufacturers need skilled teams to manage these innovations and address risks related to data quality and algorithm transparency.
How can companies manage costs in medical device development?
Companies must balance innovation with cost-effectiveness by adopting strategies that ensure high-quality production without exceeding budget constraints, particularly as regulatory compliance can increase costs.
What are the implications of extended development periods in healthcare instrument creation?
The development of healthcare instruments can take multiple years due to the need for thorough testing and validation. Streamlining processes and using agile methodologies can help reduce these timelines.
How does Voler Systems contribute to overcoming these challenges?
Voler Systems specializes in embedded systems design and employs AI-enabled design to enhance efficiency and accelerate the development process, addressing challenges in system design, power management, and overall product development.
List of Sources
- Understand Industry-Specific Challenges in Medical Device Development
- The medical device industry outlook for 2026 | Medical Economics (https://medicaleconomics.com/view/the-medical-device-industry-outlook-for-2026)
- Medical Device Industry Facts (https://advamed.org/medical-device-industry-facts)
- Medical Device Industry Facts, Trends and Statistics 2025 (https://arterexmedical.com/medical-device-industry-statistics)
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- Factors Influencing Clearance Time for Medical Devices (https://mddionline.com/medical-device-regulations/factors-influencing-fda-clearance-time-for-medical-devices-evolution-of-a-critical-regulatory-pathway)
- Implement Robust Regulatory Compliance Strategies
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- 5 Medical Device Regulatory Approval Statistics You Need to Know - Arrotek | Medical Device Innovation (https://arrotek.com/5-medical-device-regulatory-approval-statistics-you-need-to-know)
- Compliance Trends: What 2026 Will Demand of Healthcare Organizations (https://medtrainer.com/blog/compliance-trends-2026)
- Statistical Analysis for Medical Devices | MCRA (https://mcra.com/services/statistical-analysis-medical-devices)
- 7 quotes from 2023 to guide you on the medtech market (https://tiinatyni.com/blogi/7-quotes-from-2023-to-guide-you-on-the-medtech-market)
- Optimize Cost Management and Resource Allocation
- Automation, process fixes top list of cost-cutting moves for medical practices in 2026 (https://mgma.com/mgma-stat/top-cost-cutting-moves-for-medical-practices-in-2026)
- Lean Manufacturing: Principles and Systems (https://machinemetrics.com/blog/lean-manufacturing-machine-monitoring)
- Lean Six Sigma Case Study Shimadzu (https://msicertified.com/blog/lean-six-sigma-case-study-shimadzu)
- Cutting medical device manufacturing costs without cutting corners (https://todaysmedicaldevelopments.com/article/cutting-medical-device-manufacturing-costs-without-cutting-corners)
- Lean Manufacturing Quotes to Drive Efficient Industrial Operations (https://mapcon.com/blog/2014/07/11-lean-manufacturing-quotes-to-inspire-you)
- Establish Comprehensive Quality Assurance and Testing Protocols
- Measurement, Analysis, And Improvement In Medical Devices | Registrar Corp (https://registrarcorp.com/blog/medical-devices/iso-13485/measurement-analysis-improvement-in-medical-devices)
- QMSR: What the U.S. FDA’s New Inspection Manual Really Means for Device Manufacturers (https://emergobyul.com/news/qmsr-what-us-fdas-new-inspection-manual-really-means-device-manufacturers)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- Assurance of Medical Device Quality with Quality Management System: An Analysis of Good Manufacturing Practice Implementation in Taiwan - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC4449896)





