4 Steps to Choose the Right Medical Device Development Company
Discover essential steps to select the right medical device development company for your...
Embedded software is fundamental to modern medical devices, orchestrating essential functions that directly influence patient care and safety. As the healthcare landscape continues to evolve, it is crucial to understand best practices for developing and implementing this software. This understanding is vital for ensuring compliance with stringent regulatory standards while simultaneously enhancing device performance. However, the increasing complexity of these systems, coupled with the rising threat of vulnerabilities, raises a critical question: how can developers effectively navigate the challenges of creating secure, efficient, and compliant embedded software for medical applications?
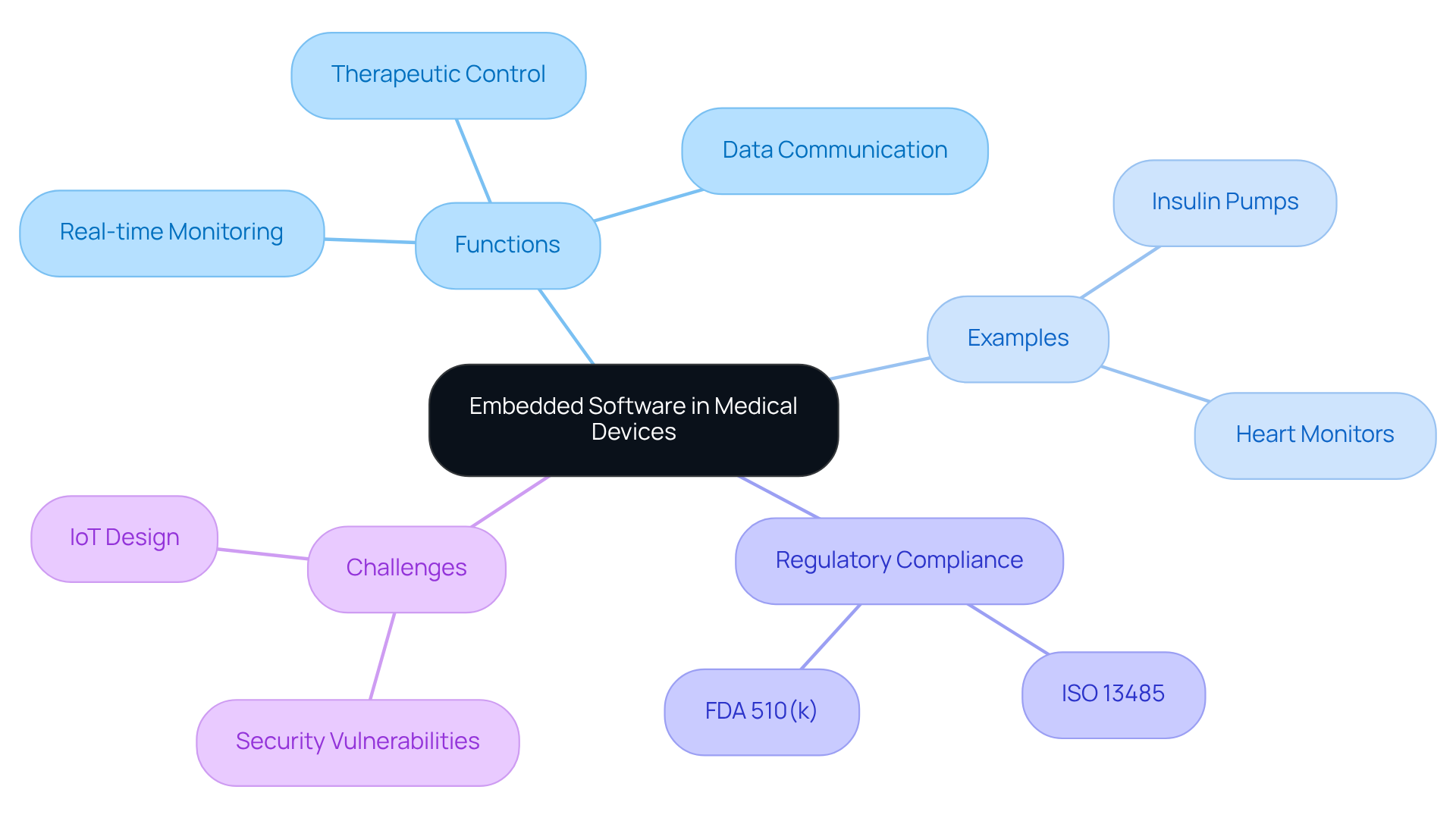
serves as a specialized component within healthcare instruments, enabling them to manage essential functions and processes vital for patient care. This application operates directly within the hardware of the unit, facilitating tasks such as , therapeutic control, and secure data communication. Its role is pivotal in ensuring the reliability, safety, and compliance of medical equipment with stringent regulatory standards, including .
For example, in insulin pumps and heart monitors, , which is critical for patient safety and effective treatment. The incorporation of allows these tools to detect anomalies that may elude human operators, thereby significantly enhancing diagnostic accuracy. As healthcare tools increasingly rely on connectivity, the importance of robust embedded programming cannot be overstated; it not only supports regulatory compliance but also directly influences patient outcomes by enabling proactive healthcare management.
Dr. Emily Carter notes that "real-time monitoring via integrated systems enables proactive instead of reactive healthcare," highlighting the transformative potential of integrated technology. Furthermore, with , the demand for secure and compliant embedded applications is more pressing than ever. addresses challenges in , such as sensor integration and power management, through its comprehensive consulting. By leveraging artificial intelligence in their designs, Voler Systems enhances the functionality and adaptability of healthcare devices, ensuring they meet the highest standards of performance and safety.
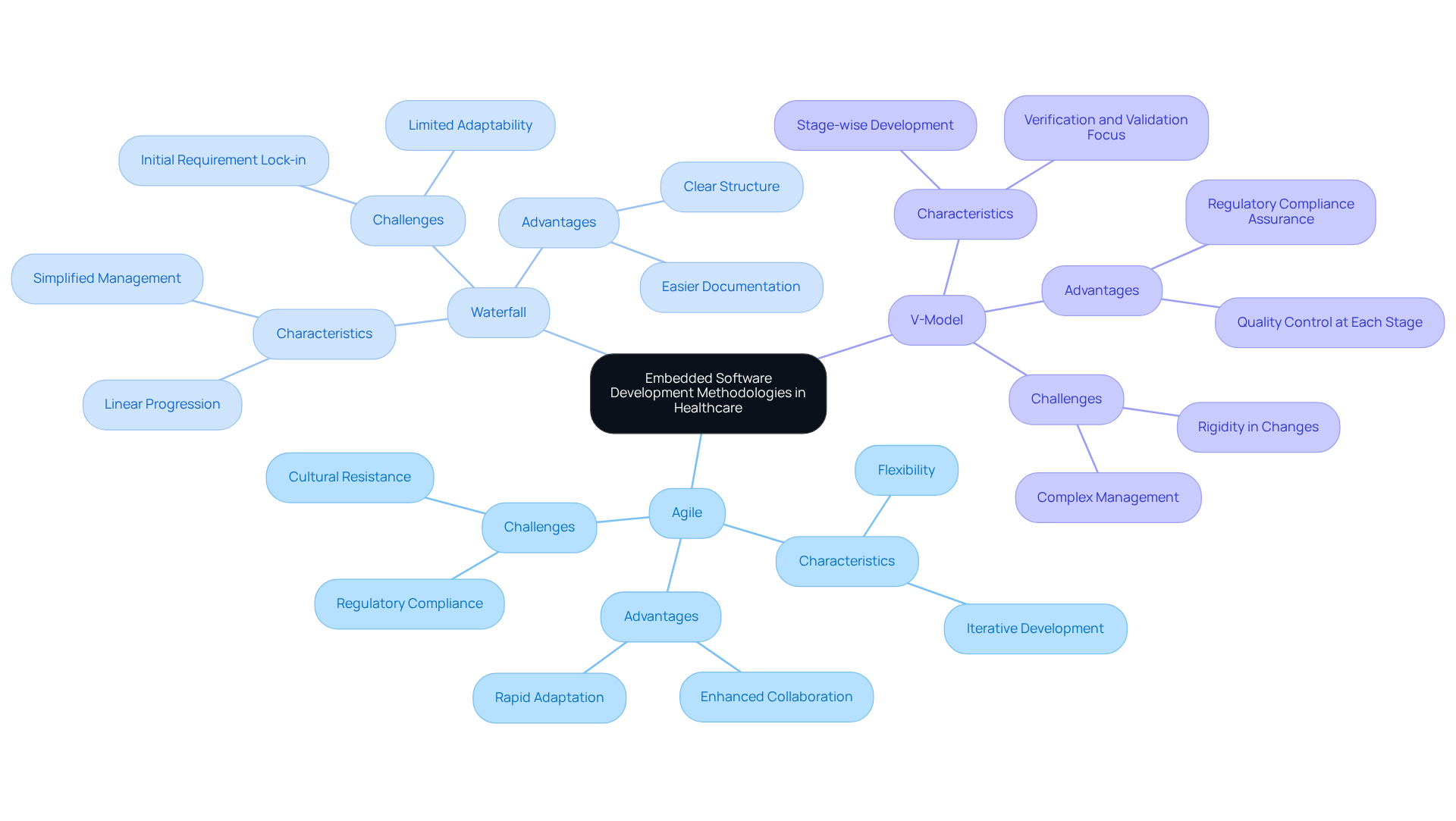
Embedded software development methodologies in healthcare applications exhibit significant variation, each presenting unique advantages and challenges. The is characterized by its iterative development and flexibility, enabling teams to swiftly adapt to changes-an essential trait in the ever-evolving . employs to enhance its , facilitating rapid modifications in hardware development and design for wearable . Conversely, the follows a linear progression, which simplifies management but restricts adaptability to changing requirements. The V-Model further refines this approach by emphasizing verification and validation at each stage, thereby ensuring adherence to stringent regulatory standards.
Selecting the appropriate methodology depends on specific , regulatory obligations, and team dynamics. Startups focused on innovative healthcare products may find Agile more aligned with their needs, while established companies might favor the structured nature of the Waterfall model. Ultimately, the decision should reflect the imperative of ensuring product quality and compliance, which are critical in the healthcare sector.
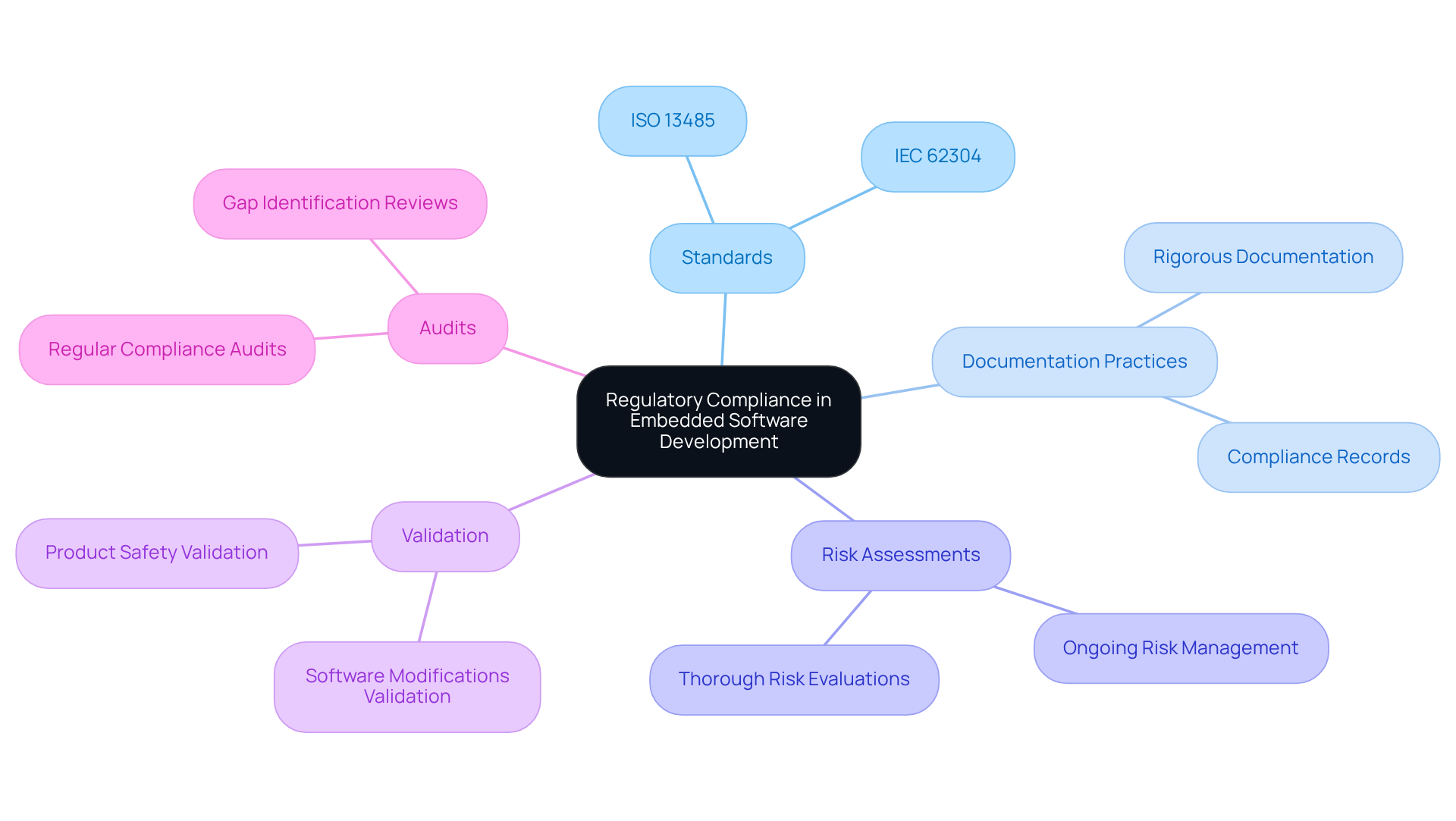
Regulatory adherence plays a vital role in the provision of for healthcare instruments. Standards such as for , ensuring that safety and effectiveness are prioritized throughout the development phase. In parallel, ISO 13485 establishes essential that are crucial for maintaining compliance.
Developers are required to implement rigorous documentation practices, , and validate all software modifications. For example, a company designing a new cardiac monitor must comply with these standards to ensure that their product meets and secures approval from .
Regular audits and reviews of compliance processes are essential for identifying potential gaps and ensuring ongoing adherence to regulations. Notably, , with a significant number of healthcare product firms integrating dev embedded software services into their development procedures. This trend underscores a within the sector.
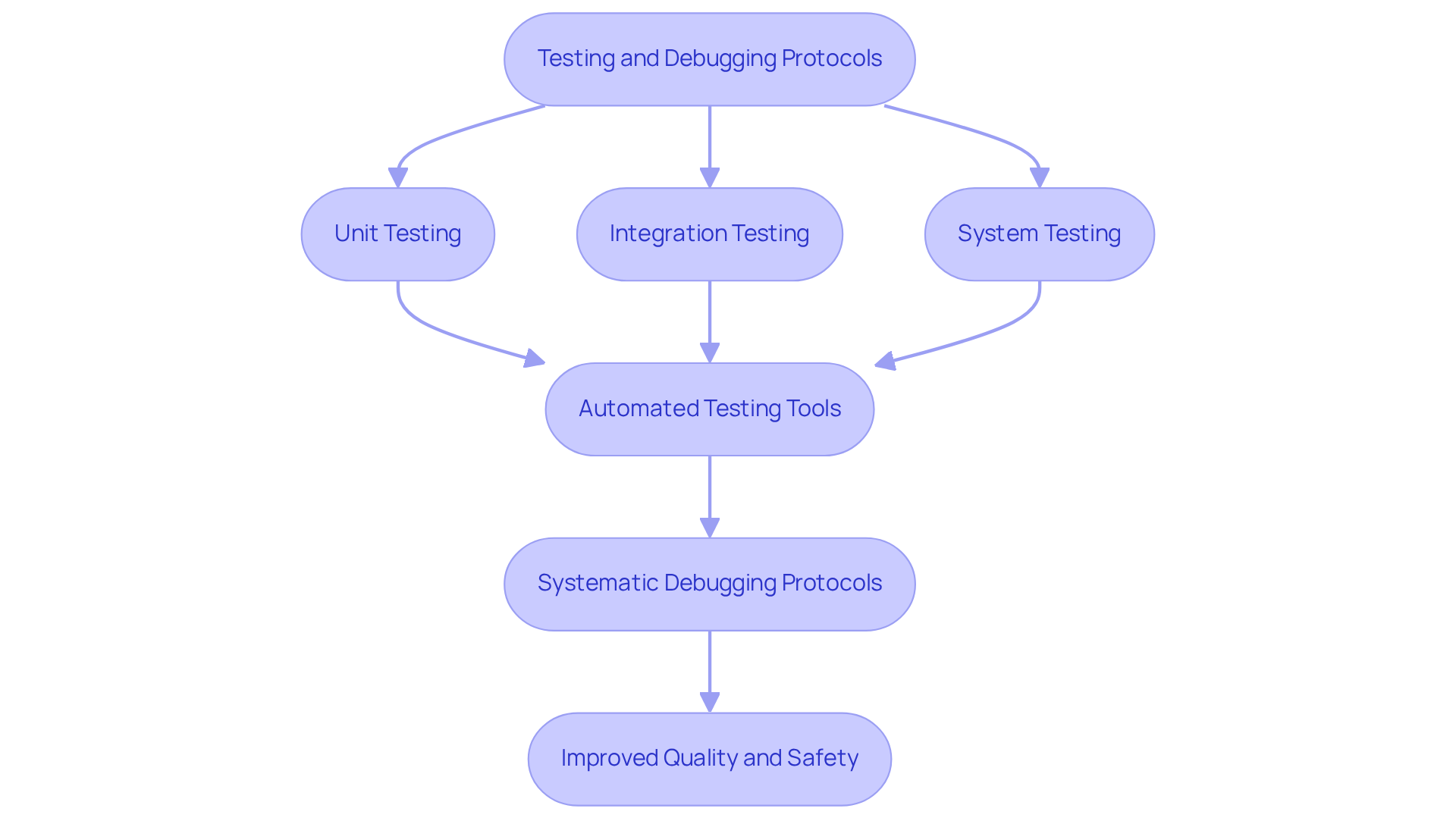
Thorough are essential for creating in . A comprehensive range of - such as unit testing, integration testing, and system testing - ensures that all components function correctly and adhere to safety standards. Automated testing tools significantly enhance efficiency and accuracy, allowing for quicker identification of defects. For instance, manufacturers of health-related equipment can employ to ensure that new updates do not introduce new bugs, thereby preserving product integrity.
Moreover, establishing is crucial for identifying and resolving issues, ensuring that the software performs reliably under all conditions. Regular not only aids in compliance with but also elevates the of the medical device. The adoption of has demonstrated a compound annual growth rate of approximately 15% over the past three years, reflecting its increasing significance in the industry.
Embedded software serves as the backbone of modern medical devices, playing a crucial role in ensuring their functionality, safety, and compliance with regulatory standards. The integration of robust embedded systems not only enhances patient care but also supports healthcare professionals in making informed decisions through real-time data processing and monitoring. As the demand for advanced medical technology grows, the importance of adhering to best practices in embedded software development becomes increasingly evident.
This article outlines key practices for effective embedded software services in medical devices. These practices include:
Each of these elements is essential for delivering high-quality, reliable medical devices that prioritize patient safety and adhere to industry standards.
In conclusion, the landscape of embedded software in healthcare is evolving rapidly, necessitating a commitment to excellence in development practices. Stakeholders in the medical device industry are encouraged to adopt these best practices to foster innovation while ensuring compliance and safety. By prioritizing rigorous testing, regulatory adherence, and responsive development methodologies, the industry can significantly enhance patient outcomes and build trust in medical technologies. Embracing these principles is not merely a regulatory obligation; it is a vital step toward advancing healthcare and improving lives.
What is embedded software in the context of medical devices?
Embedded software is a specialized component within healthcare instruments that enables them to manage essential functions and processes vital for patient care, operating directly within the hardware of the unit.
What are some functions of embedded software in medical devices?
Embedded software facilitates tasks such as real-time health monitoring, therapeutic control, and secure data communication.
Why is embedded software important for medical devices?
It is pivotal for ensuring the reliability, safety, and compliance of medical equipment with stringent regulatory standards, such as ISO 13485 and FDA 510(k).
Can you provide examples of medical devices that use embedded software?
Examples include insulin pumps and heart monitors, where integrated systems process sensor data and execute commands instantaneously for patient safety and effective treatment.
How does embedded software enhance diagnostic accuracy in healthcare?
The incorporation of advanced algorithms allows embedded systems to detect anomalies that may elude human operators, significantly improving diagnostic accuracy.
What is the impact of connectivity on embedded software in healthcare tools?
As healthcare tools increasingly rely on connectivity, robust embedded programming becomes essential for regulatory compliance and directly influences patient outcomes by enabling proactive healthcare management.
What does Dr. Emily Carter say about the role of integrated systems in healthcare?
Dr. Emily Carter notes that "real-time monitoring via integrated systems enables proactive instead of reactive healthcare," emphasizing the transformative potential of integrated technology.
What challenges are associated with IoT design in healthcare?
Challenges include sensor integration and power management, which are addressed by companies like Voler Systems through comprehensive IoT design consulting.
How does Voler Systems enhance healthcare devices?
Voler Systems leverages artificial intelligence in their designs to enhance the functionality and adaptability of healthcare devices, ensuring they meet high standards of performance and safety.
What is the significance of security in embedded applications for medical devices?
With 53% of connected healthcare instruments in hospitals exhibiting recognized critical vulnerabilities, the demand for secure and compliant embedded applications is increasingly important for patient safety and regulatory adherence.
