Introduction
Product risk analysis is essential in the medical device industry, forming the foundation for patient safety and regulatory compliance. As manufacturers face evolving standards and heightened scrutiny, the demand for effective methodologies and structured processes has reached a critical point. Despite this, many organizations continue to depend on outdated practices that do not adequately address the complexities of contemporary risk management. Companies must adapt their risk analysis strategies not only to fulfill regulatory requirements but also to improve the safety and efficacy of their medical devices.
Define Product Risk Analysis in Medical Devices
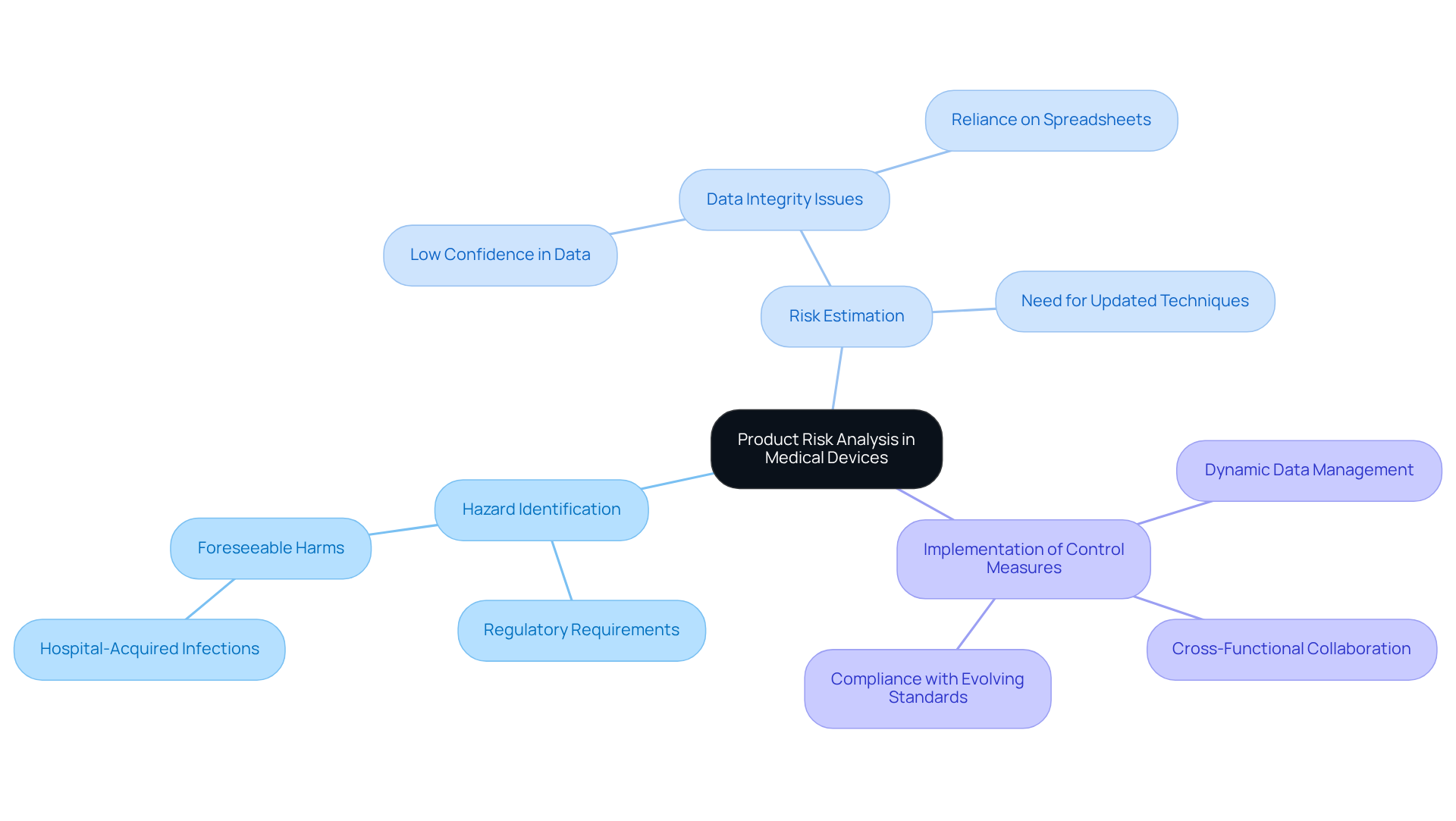
is a systematic process designed to identify, evaluate, and mitigate risks throughout the . This process is essential for ensuring that medical devices are both for patient use. It involves various methodologies, including:
- Hazard identification
- Risk estimation
- Implementation of control measures
All aimed at minimizing potential harm to users. , require manufacturers to conduct thorough evaluations to comply with and ensure product reliability.
Recent surveys indicate that fewer than 25% of leaders in are satisfied with their current management practices, and only one-third express confidence in the reliability of their assessment data. This dissatisfaction highlights the urgent need for updated techniques to evaluate uncertainties, as that fail to capture the complexities of management.
Successful examples of product uncertainty evaluation in medical equipment manufacturing demonstrate the benefits of integrating dynamic data management and real-time assessments. For instance, companies that have adopted advanced management platforms developed by report improved collaboration and data integrity, which are vital for maintaining compliance with evolving regulatory standards. As the industry faces increased scrutiny from regulatory bodies, prioritizing a robust process is not merely advantageous; it is essential for ensuring the safety and efficacy of medical instruments.
Utilize Effective Methodologies for Risk Assessment
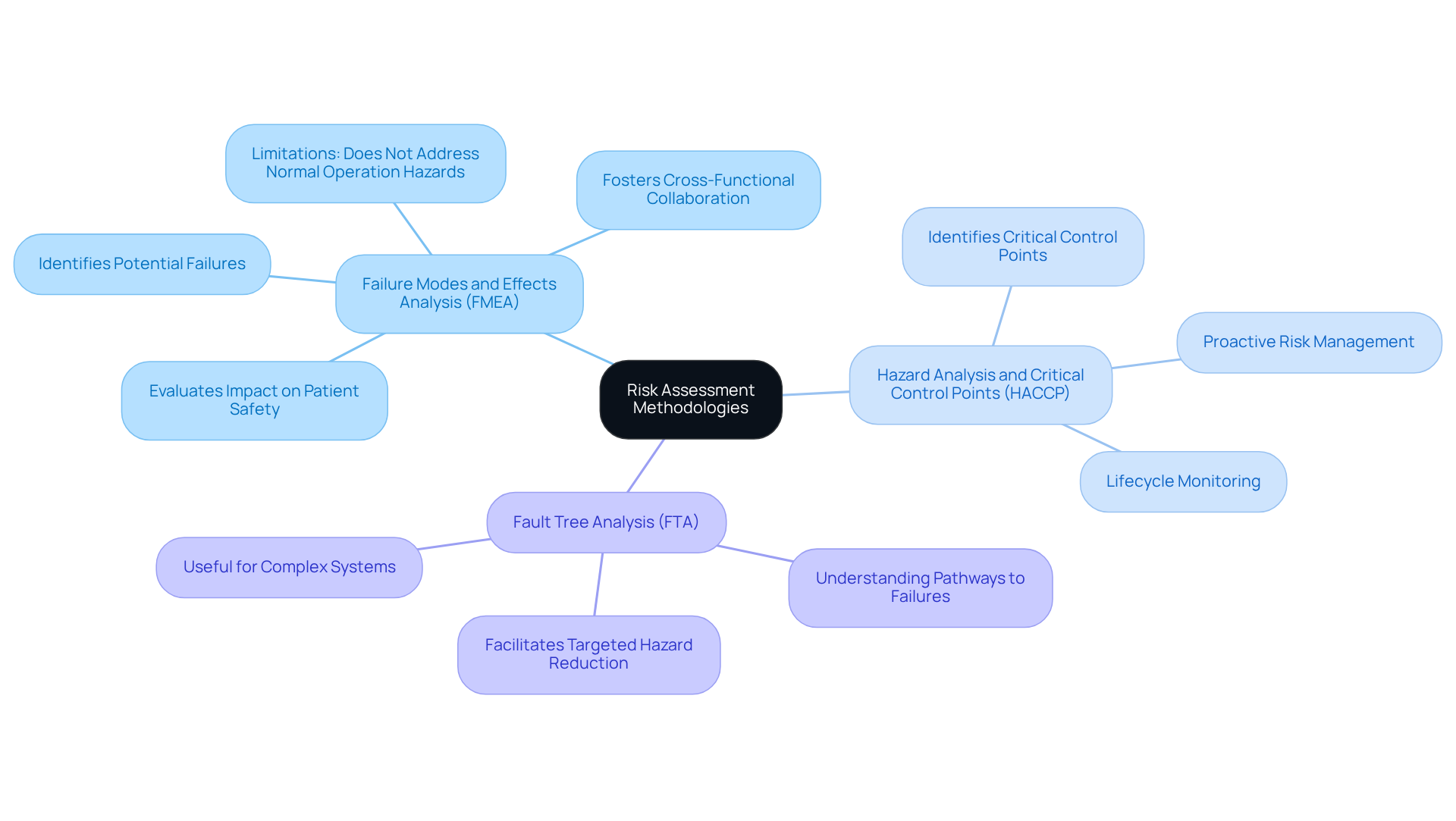
To conduct a , it is essential to utilize effective methodologies tailored to the specific under review. Key methodologies include:
- : This structured approach identifies potential failure modes within a device, evaluating their impact on and device performance. FMEA is widely recognized in the industry for its ability to foster , allowing teams to prioritize and mitigate failure modes effectively. However, it is important to note that FMEA primarily addresses technical failures and may not account for hazards that occur during normal operation, such as user errors or environmental factors. Consequently, it should not serve as the sole basis for a strategy to manage uncertainties.
- : Originally developed for food safety, HACCP can be adapted for the medical device sector to identify critical points in the device lifecycle where risks can be controlled. This proactive approach underscores the importance of monitoring and managing challenges throughout the , ensuring that potential threats are addressed before they compromise .
- : This deductive method aids in understanding the pathways that may lead to system failures, facilitating targeted hazard reduction strategies. FTA is particularly beneficial for complex systems where multiple failure scenarios may interact, providing a comprehensive view of potential hazards.
By employing these methodologies, manufacturers can perform a product to systematically evaluate hazards and implement effective measures to enhance equipment safety. The integration of FMEA, HACCP, and FTA not only bolsters compliance with but also cultivates a culture of safety and quality within the organization.
Implement a Structured Risk Analysis Process
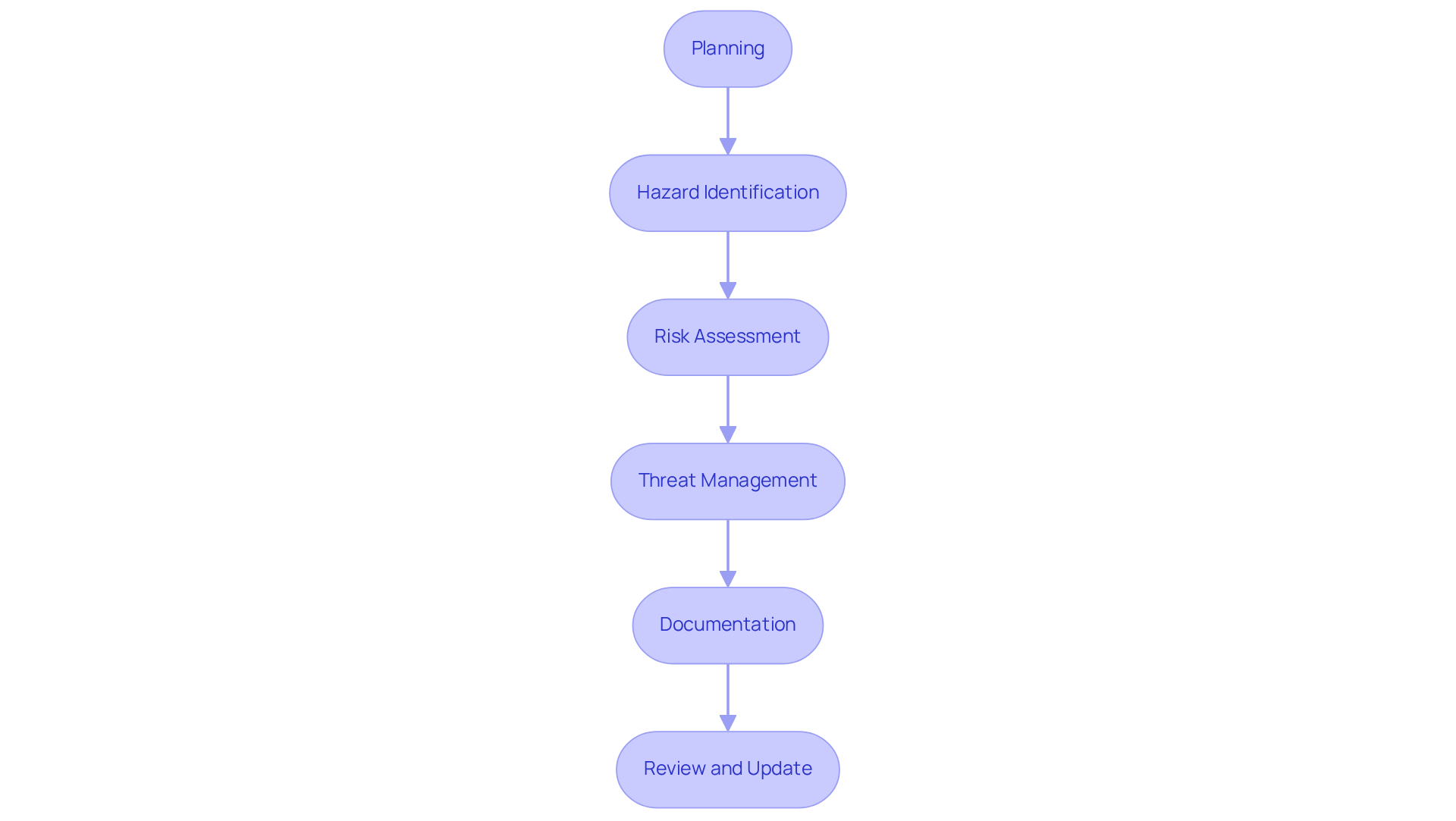
A structured risk analysis process for medical devices encompasses several essential steps:
- Planning: Clearly define the scope of the threat assessment, focusing on the item's intended use and the target user group. ' proficiency in can aid startups in building a strong foundation for their threat evaluation.
- : Identify potential hazards associated with the device through comprehensive literature reviews, consultations with experts, and analysis of historical data. Leveraging ' experience in innovative medical technology development can enhance this identification process.
- Risk Assessment: Evaluate the likelihood and severity of identified threats using both quantitative and qualitative methods, ensuring a robust evaluation. This step is crucial for aligning with regulatory standards, which can help navigate effectively.
- Threat Management: Develop plans to reduce identified hazards, which may involve design alterations, user education, and updates to labeling. It is essential to address foreseeable misuse and incorporate postmarket surveillance data to continuously monitor device performance and safety. ' can simplify hardware development and design modifications, ensuring that control measures are effectively implemented.
- Documentation: Maintain meticulous records of the , detailing methodologies employed, findings, and decisions made to ensure transparency and compliance. Precise information is crucial for efficient hazard management, as indicated by Matthew Baretich, P.E., Ph.D., who stresses that if your data is set up appropriately, failure rates can be easily determined. can assist in creating comprehensive documentation that meets regulatory requirements.
- Review and Update: Regularly revisit and refresh the assessment as new information arises or as the equipment undergoes modifications, ensuring ongoing compliance with evolving regulatory standards. This dynamic approach illustrates the mindset change in management practices, as emphasized by Billy Delfs, who observes that . ' role in compliant and reliable electronic equipment design supports this ongoing process.
By following these steps, manufacturers can implement a comprehensive strategy for that complies with regulatory standards and improves product protection, ultimately offering a competitive edge in the market.
Establish Continuous Monitoring and Improvement Practices
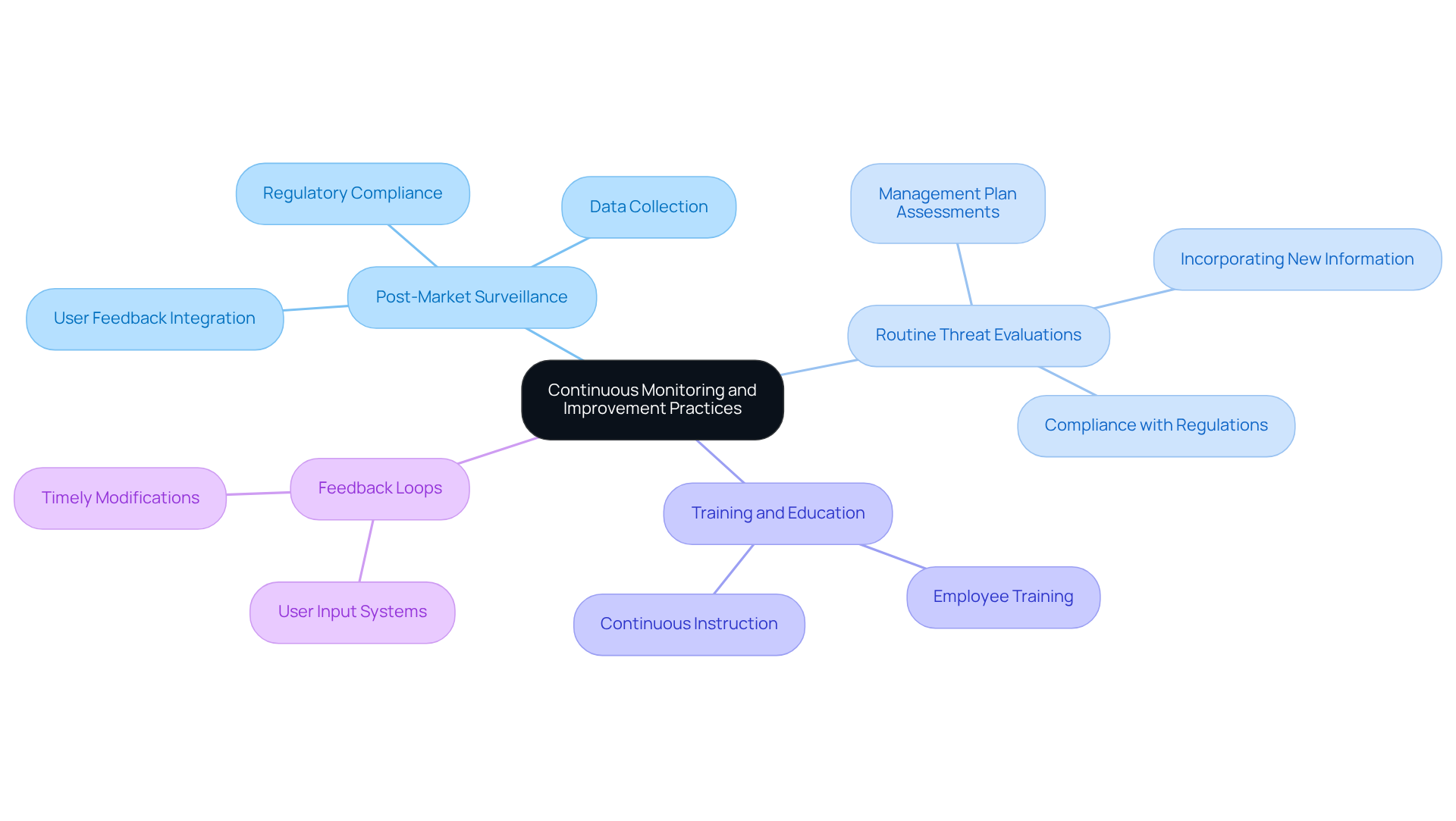
Ongoing oversight and enhancement methods are essential for ensuring the security and efficiency of medical instruments. Key practices include:
- : Establish robust systems to monitor device performance in real-world settings, systematically collecting data on and user feedback. This proactive approach enhances , fosters innovation, and builds market trust. For instance, has successfully assisted a large medical device company with legacy test equipment, leading to improved compliance and reliability in their monitoring processes.
- : Conduct regular assessments of the management plan to incorporate new information, technological developments, and . This ensures that management strategies remain relevant and effective in addressing emerging challenges. Compliance with regulations such as 21 CFR Part 806 and 21 CFR Part 803 is crucial for maintaining proper reporting of device corrections and .
- : Provide continuous instruction for personnel involved in managing uncertainties to ensure they are informed about the latest practices and regulatory requirements. Equipping employees with knowledge is vital for effective threat mitigation.
- : Implement systems for integrating input from users and healthcare professionals into the management process. This facilitates timely modifications to risk management, ensuring that instruments meet the evolving needs of patients and healthcare providers. Engaging with regulatory authorities is also a best practice that can enhance compliance and foster collaboration.
By cultivating a culture of ongoing advancement and addressing common challenges such as data gathering issues, manufacturers can significantly enhance safety and ensure adherence to evolving regulatory standards. Effective , as demonstrated by ' contributions, can lead to timely corrective actions, ultimately improving device performance and patient outcomes.
Conclusion
In the development and management of medical devices, product risk analysis stands as a critical component, ensuring safety and effectiveness for patient use. This systematic process not only identifies and evaluates potential risks but also implements strategies to mitigate them throughout the product lifecycle. Given the stringent compliance requirements set by regulatory bodies, the significance of a robust risk analysis cannot be overstated.
The article delineates various methodologies essential for effective risk assessment, including:
- FMEA
- HACCP
- FTA
Each contributing uniquely to the identification and management of hazards. Furthermore, a structured risk analysis process encompassing:
- Planning
- Hazard identification
- Risk assessment
- Threat management
- Documentation
- Regular review
is crucial for maintaining compliance with evolving standards. Continuous monitoring practices, such as post-market surveillance and routine evaluations, further enhance safety and efficacy, ensuring that medical devices adhere to the highest quality standards.
Ultimately, integrating dynamic data management and real-time assessments into the risk analysis process fosters a culture of safety and equips manufacturers to navigate the complexities of regulatory compliance. By prioritizing these best practices, medical device companies can significantly improve patient outcomes and build trust in their products, thereby reinforcing the vital role of diligent risk analysis within the industry.
Frequently Asked Questions
What is product risk analysis in medical devices?
Product risk analysis in medical devices is a systematic process aimed at identifying, evaluating, and mitigating risks throughout the product lifecycle to ensure the safety and effectiveness of medical devices for patient use.
What methodologies are involved in product risk analysis?
The methodologies involved include hazard identification, risk estimation, and the implementation of control measures, all aimed at minimizing potential harm to users.
Why is product risk analysis important for medical device manufacturers?
It is important because regulatory authorities, such as the FDA and ISO, require thorough evaluations to comply with safety standards and ensure product reliability.
What are some concerns expressed by leaders in medical equipment development regarding risk management practices?
Recent surveys indicate that fewer than 25% of leaders are satisfied with their current management practices, and only one-third express confidence in the reliability of their assessment data, highlighting the need for updated techniques.
What issues arise from traditional methods of product risk analysis?
Traditional methods often rely on static spreadsheets that fail to capture the complexities of management, leading to dissatisfaction among industry leaders.
How can integrating dynamic data management improve product risk analysis?
Integrating dynamic data management and real-time assessments can enhance collaboration and data integrity, which are crucial for maintaining compliance with evolving regulatory standards.
Why is prioritizing product risk analysis essential for the medical device industry?
Prioritizing a robust product risk analysis process is essential due to increased scrutiny from regulatory bodies, ensuring the safety and efficacy of medical instruments.
List of Sources
- Define Product Risk Analysis in Medical Devices
- FDA's Risk Management Expectations for Medical Devices: What Manufacturers Need to Know (https://gardner.law/news/fda-risk-management-expectations-medical-devices)
- Rising Risk: Why Many Medical Device Companies Are In Danger of Risk Analysis Missteps (https://blog.cognition.us/why-many-medical-device-companies-are-in-danger-of-risk-analysis-missteps)
- zewskicorp.com (https://zewskicorp.com/blog/risk-analysis-in-medical-device-design-why-it-matters)
- Utilize Effective Methodologies for Risk Assessment
- FMEA: Why You're Missing Critical Risks in Medical Device Lifecycle Management (https://propharmagroup.com/thought-leadership/fmea-missing-critical-risks-medical-device-lifecycle-management)
- ISO 14971: FMEA, FTA and risk analysis for devices (https://csdmed.mc/en/news/medical-devices-regulation/risk-management-fmea-fta-132)
- FDA's Risk Management Expectations for Medical Devices: What Manufacturers Need to Know (https://gardner.law/news/fda-risk-management-expectations-medical-devices)
- euro-symbiose.com (https://euro-symbiose.com/actuality-en/fmea-and-iso-14971-two-complementary-approaches-to-managing-risks-associated-with-medical-devices)
- FDA Updates Risk Assessment for Medical Devices in US | Emily Harari posted on the topic | LinkedIn (https://linkedin.com/posts/emilyharari_cdrh-learn-activity-7418385394595184640-Y2Zo)
- Implement a Structured Risk Analysis Process
- FDA's Risk Management Expectations for Medical Devices: What Manufacturers Need to Know (https://gardner.law/news/fda-risk-management-expectations-medical-devices)
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
- Medical Device Risk Scoring: Measuring Risk and Preparing for its Eventuality - TechNation (https://1technation.com/medical-device-risk-scoring-measuring-risk-and-preparing-for-its-eventuality)
- FDA Updates Risk Assessment for Medical Devices in US | Emily Harari posted on the topic | LinkedIn (https://linkedin.com/posts/emilyharari_cdrh-learn-activity-7418385394595184640-Y2Zo)
- Establish Continuous Monitoring and Improvement Practices
- Post Market Surveillance: What You Need to Know to Ensure Patient… (https://nsf.org/knowledge-library/post-market-surveillance-what-you-need-to-know-to-ensure-patient-safety)
- Checking your browser - reCAPTCHA (https://pmc.ncbi.nlm.nih.gov/articles/PMC12624750)
- Industry groups urge FDA to use existing controls to monitor AI-assisted devices (https://raps.org/news-and-articles/news-articles/2026/1/industry-groups-urge-fda-to-use-existing-controls)
- Medical Device Post-Market Surveillance: Complete FDA Compliance Guide 2025 (https://complizen.ai/post/medical-device-post-market-surveillance-fda)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)





