Introduction
Feasibility testing is a pivotal phase in the development of medical devices, serving as a safeguard against potentially costly missteps. By rigorously evaluating a concept's viability, developers can identify potential technical, clinical, and regulatory challenges before committing significant resources. Despite its critical role, many teams encounter difficulties in executing effective feasibility tests.
What strategies can be implemented to address these challenges and ensure a thorough evaluation process?
Understand the Importance of Feasibility Testing for Medical Devices
A represents a pivotal phase in the , acting as . This process is essential for identifying potential technical, clinical, and regulatory challenges at an early stage, allowing developers to make informed decisions prior to committing substantial resources. Through viability assessments, teams can scrutinize the design, functionality, and market potential of the apparatus, ensuring compliance with safety and efficacy standards. This proactive approach not only mitigates risks but also significantly enhances the likelihood of and market entry.
Key aspects of encompass:
- : Assessing the device's manufacturability and production efficiency.
- : Assessing the instrument's effectiveness and safety in practical applications.
- : .
Recent advancements in viability examination have introduced innovative methodologies, including , which improve the accuracy of assessments and streamline the evaluation process. Current trends highlight a growing emphasis on integrating tools into clinical workflows, ensuring their practicality and effectiveness in real healthcare environments. Successful viability assessments underscore the importance of early-stage evaluations in refining designs and enhancing functionality.
Ultimately, a is crucial for , , thereby facilitating successful commercialization.
, then explore each branch to see the important aspects and details that contribute to understanding its significance in medical device development. Start at the center with the main idea of feasibility test, then explore each branch to see the important aspects and details that contribute to understanding its significance in medical device development.](https://images.tely.ai/telyai/gnkmcplc-start-at-the-center-with-the-main-idea-of-feasibility-testing-then-explore-each-branch-to-see-the-important-aspects-and-details-that-contribute-to-understanding-its-significance-in-medical-device-development.webp)
Follow Step-by-Step Procedures for Conducting Feasibility Tests
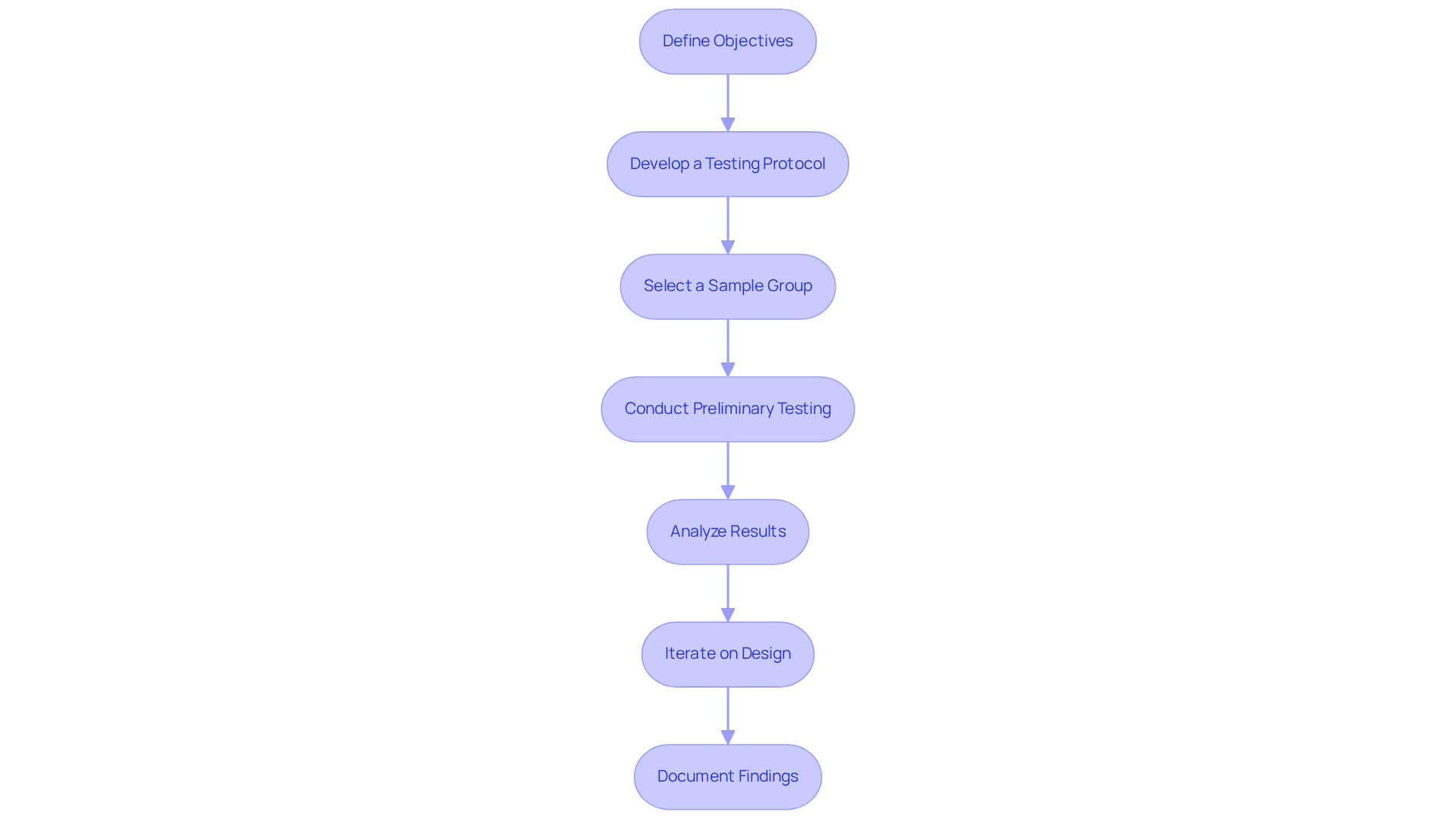
Carrying out viability assessments involves several essential stages that ensure a comprehensive evaluation of the medical apparatus concept. Here’s a step-by-step guide:
- Define Objectives: Clearly articulate the goals of the , specifying the insights you aim to gain regarding the . Establishing clear objectives is crucial for directing the entire evaluation process.
- : Create a detailed plan outlining the methods and criteria for testing. This should encompass the types of tests to be conducted, metrics for success, and a realistic timeline. Best practices suggest that protocols should be adaptable to accommodate findings as they emerge.
- Select a : Identify a representative sample of users or conditions under which the equipment will be tested. This selection is vital for gathering relevant data that reflects real-world usage, thereby enhancing the validity of the study.
- Conduct Preliminary Testing: Execute initial tests to gather data on the device's performance. This may involve bench evaluations, simulations, or early clinical trials. Engaging in initial assessments allows for early recognition of potential issues and user concerns.
- : Review the data collected during testing to identify any issues or areas for improvement. This analysis should focus on both technical performance and user feedback, ensuring that all elements of the product are thoroughly assessed.
- Iterate on Design: Based on the findings, make necessary adjustments to the device design. This iterative approach is essential for enhancing the product before progressing to more extensive evaluation stages, as it facilitates ongoing improvement based on real-world insights.
- : Maintain comprehensive records of the testing procedure, results, and any alterations made. This documentation is vital for and future reference, ensuring compliance with industry standards and facilitating smoother transitions to subsequent development stages. By leveraging ' expertise in , including the development of user manuals and templates, developers can enhance their understanding of regulatory requirements and streamline their processes. Furthermore, consider creating a reimbursement strategy to inform payors about the product's impact on outcomes and ease market entry.
By adhering to these procedures, developers can ensure a thorough that establishes the foundation for successful medical product development. Emphasizing the importance of iterative design, thorough documentation, and addressing potential consequences of skipping early stages will enhance the likelihood of meeting regulatory requirements and achieving market success.
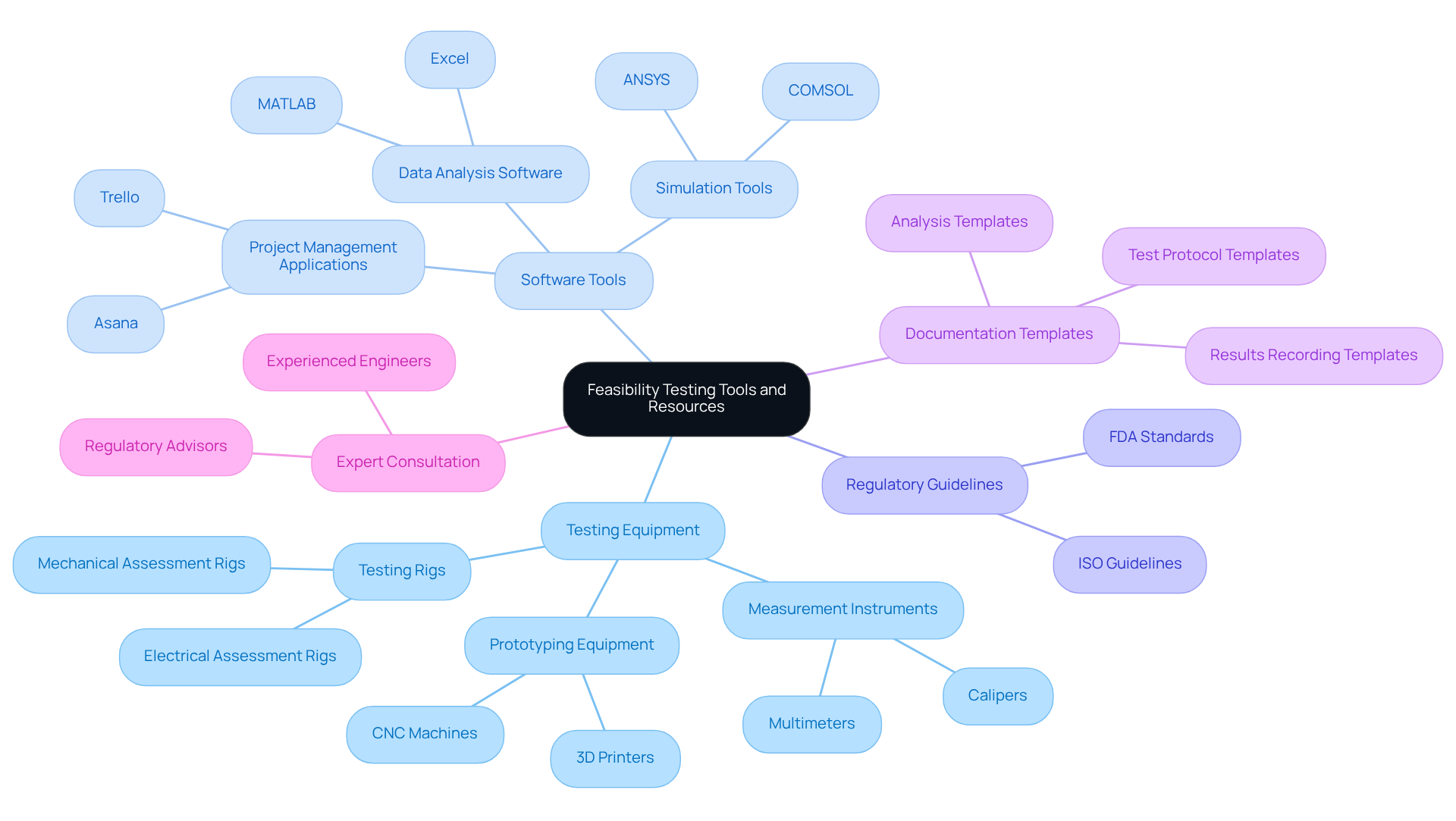
To conduct an effective , it is crucial to gather the appropriate tools and resources. Below is a comprehensive list of what you will require:
-
: Depending on the device, essential tools may include:
- Measurement instruments such as calipers and multimeters for precise evaluations.
- like 3D printers and CNC machines to create functional models.
- Testing rigs designed for mechanical and electrical assessments to ensure performance under various conditions.
-
Software Tools: Utilize software for:
- , employing platforms like MATLAB and Excel to interpret results effectively.
- to model device behavior under different scenarios.
- Project management, leveraging applications like Trello and Asana to streamline workflows and track progress.
-
Regulatory Guidelines: Familiarize yourself with relevant , including those from the FDA and ISO, to ensure compliance throughout the evaluation process.
-
Documentation Templates: Prepare standardized templates for recording test protocols, results, and analyses, which will aid in streamlining documentation and maintaining consistency.
-
: Collaborate with regulatory advisors or experienced engineers who can provide valuable insights and guidance during the evaluation phase.
Equipping yourself with these tools and resources will facilitate a smoother and more effective feasibility test, ultimately leading to improved outcomes in your .
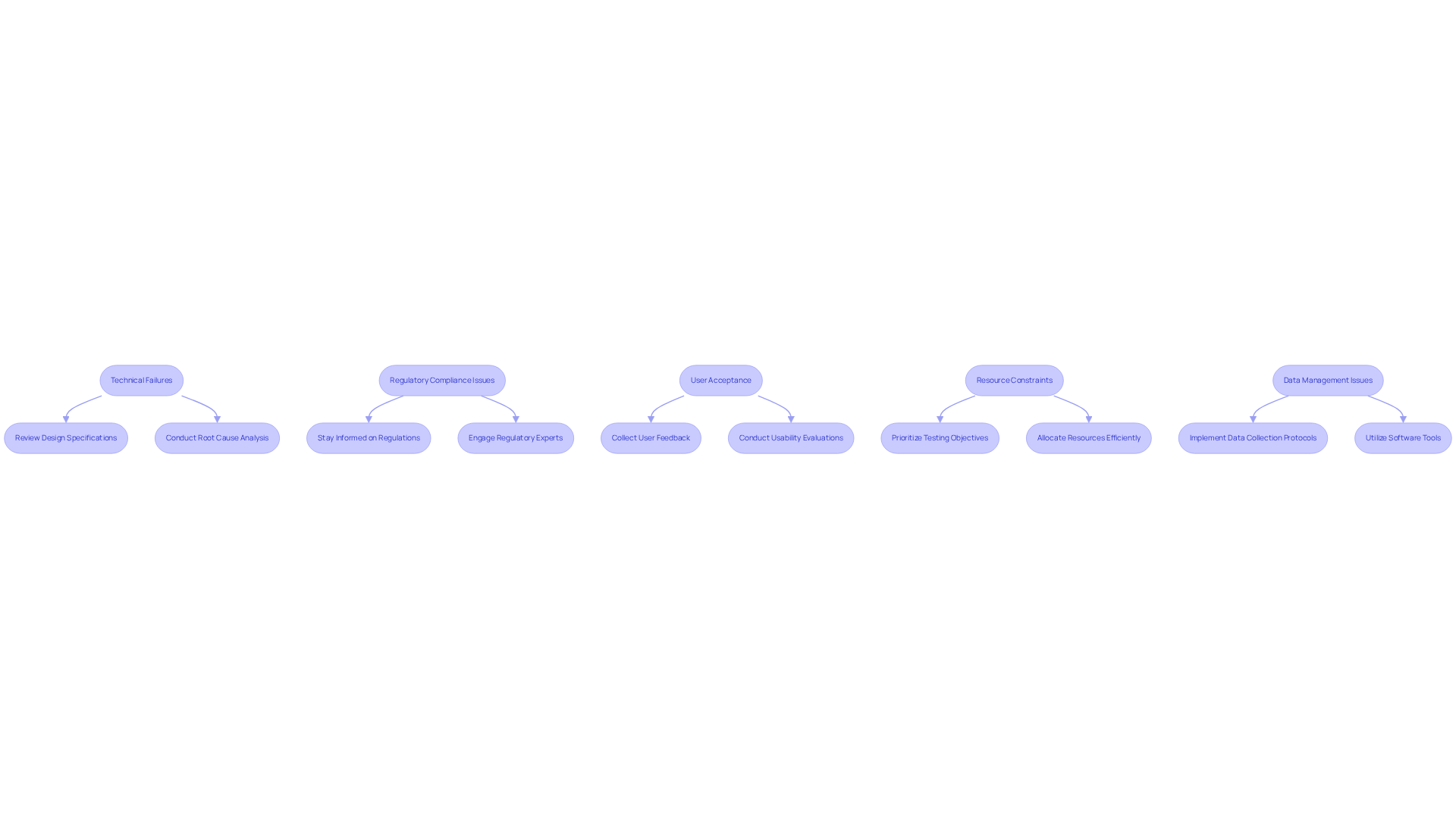
Identify Challenges and Troubleshoot Common Issues in Testing
Feasibility evaluation can present various obstacles that may hinder the process. Below are common issues along with strategies for troubleshooting:
-
: Devices may not perform as expected during testing. To troubleshoot this:
- Review design specifications to ensure all components are functioning correctly.
- Conduct a root cause analysis to identify underlying issues. Notably, nearly 32% of failed the initial acceptance check in 2022, often due to technical discrepancies.
-
: Failing to meet regulatory standards can impede progress. To address this:
- Stay informed about relevant regulations and guidelines, particularly the evolving , which can extend decision times up to 18 months.
- Engage with regulatory experts early in the process to ensure compliance, as expert regulatory support is essential for navigating complexities and minimizing delays.
-
: If users find the device difficult to use, market success may be compromised. To mitigate this:
- Collect user feedback during testing and iterate on design based on their input. can uncover critical pain points that impact acceptance.
- Conduct usability evaluations to effectively identify and address these issues.
-
: Limited time or budget can affect the quality of evaluations. To manage this:
- Prioritize testing objectives and focus on critical aspects first, ensuring essential functionalities are validated before broadening the scope.
- Allocate resources efficiently and consider outsourcing specific tasks if necessary to maintain quality without exceeding budget constraints.
-
: Inadequate data collection can lead to inconclusive results. To enhance data management:
- Implement robust data collection protocols and ensure all team members are trained on them. In fact, 67% of resulted in requests for additional information during the review process, often due to data inadequacies.
- Utilize software tools to streamline data analysis and reporting, thereby enhancing the clarity and accessibility of findings.
By proactively identifying and addressing these challenges, you can enhance the effectiveness of your feasibility test and increase the likelihood of successful .
Conclusion
Feasibility testing is a cornerstone in the development of medical devices, providing a critical early assessment of a concept's viability. This essential process enables developers to identify potential challenges related to technical performance, clinical application, and regulatory compliance before committing significant resources. By prioritizing feasibility testing, teams can enhance their chances of successful product development and ensure that new devices meet safety and efficacy standards.
Key steps in conducting effective feasibility tests include:
- Defining clear objectives
- Developing a comprehensive testing protocol
- Selecting an appropriate sample group
- Iterating on design based on preliminary results
Additionally, gathering the right tools and resources, such as testing equipment and software for data analysis, is crucial for thorough evaluation. Addressing common challenges like technical failures, regulatory compliance issues, and user acceptance can further streamline the testing process and improve outcomes.
Ultimately, embracing a structured approach to feasibility testing not only mitigates risks but also paves the way for successful commercialization of medical devices. Stakeholders in the medical device industry are encouraged to prioritize feasibility assessments as a means of fostering innovation and ensuring that new products effectively address real-world healthcare needs. The insights gained from these evaluations can lead to safer, more effective devices that enhance patient care and contribute to the advancement of medical technology.
Frequently Asked Questions
What is the purpose of a feasibility test in medical device development?
A feasibility test serves as a preliminary assessment to gauge the viability of a concept, identifying potential technical, clinical, and regulatory challenges early in the development process.
Why is conducting a feasibility test important before committing resources?
Conducting a feasibility test allows developers to make informed decisions and mitigate risks, significantly enhancing the likelihood of successful product development and market entry.
What key aspects are evaluated during feasibility testing?
Key aspects include technical feasibility (manufacturability and production efficiency), the effectiveness and safety of the device in practical applications, and navigating the regulatory landscape for compliance with relevant standards.
How have recent advancements improved feasibility testing?
Recent advancements have introduced innovative methodologies, such as AI-driven analytics, which enhance the accuracy of assessments and streamline the evaluation process.
What current trends are observed in feasibility testing for medical devices?
There is a growing emphasis on integrating tools into clinical workflows to ensure their practicality and effectiveness in real healthcare environments.
How does feasibility testing contribute to product commercialization?
Feasibility testing validates concepts, reduces development costs, and ensures that medical products are safe and effective for end-users, facilitating successful commercialization.
List of Sources
- Understand the Importance of Feasibility Testing for Medical Devices
- Early Feasibility Study Challenges for Medical Devices (https://clinicaltrialsarena.com/comment/early-feasibility-study-challenges-medical-devices)
- FastWave completes enrollment in feasibility study for laser IVL, earns 9th patent (https://massdevice.com/fastwave-completes-enrollment-sola-feasibility-study)
- The Importance of Early Feasibility Studies in Medical Device Lifecycles (https://gcmiatl.org/the-importance-of-early-feasibility-studies-in-medical-device-lifecycles)
- Medical Device Concept & Product Development | Innoven (https://innovenn.com/medical-device-development-consulting/medical-device-concept-development-feasibility)
- Medical Device Industry Facts, Trends and Statistics 2025 (https://arterexmedical.com/medical-device-industry-statistics)
- Follow Step-by-Step Procedures for Conducting Feasibility Tests
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- Medical Device Concept & Product Development | Innoven (https://innovenn.com/medical-device-development-consulting/medical-device-concept-development-feasibility)
- The Applied Science (https://theappliedscience.com/journey-of-medical-device)
- pmc.ncbi.nlm.nih.gov (https://pmc.ncbi.nlm.nih.gov/articles/PMC8849521)
- jmir.org (https://jmir.org/2025/1/e77982)
- Gather Essential Tools and Resources for Feasibility Testing
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- exponent.com (https://exponent.com/article/using-statistical-science-optimize-quality-across-medical-device-life-cycle)
- AI-Driven Tools Boost U.S. Medical Device Development (https://businesswire.com/news/home/20251023023929/en/AI-Driven-Tools-Boost-U.S.-Medical-Device-Development)
- Custom Healthcare Tech Success Stories: SaMD, AI & EMR Integration (https://invene.com/case-studies)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Identify Challenges and Troubleshoot Common Issues in Testing
- 6 Important Quotes from Medtech Leaders (https://mddionline.com/business/6-important-quotes-from-medtech-leaders)
- Medical Device Industry Facts, Trends and Statistics 2025 (https://arterexmedical.com/medical-device-industry-statistics)
- 5 Medical Device Regulatory Approval Statistics You Need to Know - Arrotek | Medical Device Innovation (https://arrotek.com/5-medical-device-regulatory-approval-statistics-you-need-to-know)
- Case study finds regulatory challenges are affecting MedTech innovation in the UK | King's College London (https://kcl.ac.uk/news/case-study-finds-regulatory-challenges-are-affecting-medtech-innovation-in-the-uk)
- jmir.org (https://jmir.org/2025/1/e77982)

, then explore each branch to see the important aspects and details that contribute to understanding its significance in medical device development. Start at the center with the main idea of feasibility test, then explore each branch to see the important aspects and details that contribute to understanding its significance in medical device development.](https://images.tely.ai/telyai/gnkmcplc-start-at-the-center-with-the-main-idea-of-feasibility-testing-then-explore-each-branch-to-see-the-important-aspects-and-details-that-contribute-to-understanding-its-significance-in-medical-device-development.webp)



