Introduction
Machine-to-Machine (M2M) communication is transforming the healthcare landscape by allowing devices to autonomously exchange critical information. This innovation paves the way for advancements such as remote patient monitoring and real-time health analytics. This guide outlines the essential steps for implementing M2M communication in medical devices, emphasizing significant benefits like enhanced patient care and improved operational efficiency. However, as healthcare providers adopt this technology, they encounter challenges, including:
- Connectivity issues
- Regulatory compliance
How can they effectively navigate these hurdles to fully leverage the potential of M2M communication?
Understand Machine-to-Machine (M2M) Communication
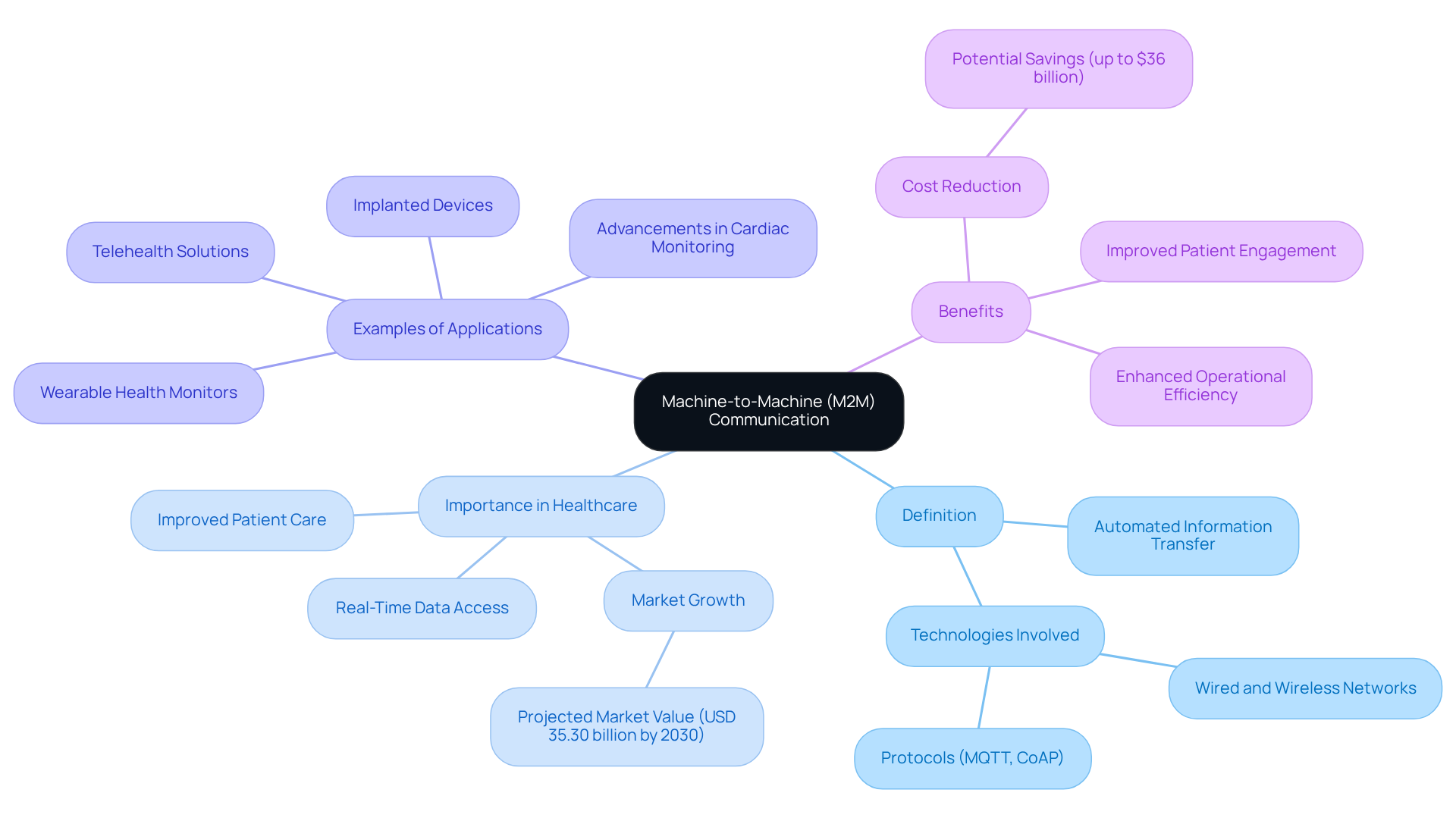
Machine-to-Machine (M2M) interaction, which is designed for machine to machine or m2m communication, refers to the automated transfer of information between devices without human involvement. In the medical equipment sector, it facilitates seamless information exchange that is essential for applications such as and real-time health data analysis.
- Definition: encompasses various technologies that enable devices to connect directly, utilizing both wired and wireless networks. Protocols like MQTT and CoAP are specifically designed for efficient information transmission, ensuring that critical data is communicated promptly.
- The importance in healthcare is that it significantly improves by allowing systems to share vital information, such as heart rate or glucose levels, directly with healthcare providers. This can lead to timely interventions, ultimately enhancing patient outcomes. For example, the global M2M medical market is projected to reach USD 35.30 billion by 2030, highlighting the increasing reliance on these technologies to optimize .
- Examples of common applications of M2M in medical devices, such as wearable health monitors, implanted devices, and telehealth solutions that enable remote consultations and monitoring, illustrate how these technologies empower patients to independently measure and transmit data directly to their healthcare providers, improving the quality of care. Notably, advancements in cardiac monitoring are evolving from tethered systems to fully wireless solutions, demonstrating the innovative capabilities of companies like .
- Benefits: The advantages of are numerous, including enhanced operational efficiency, reduced costs, and improved patient engagement through continuous monitoring and feedback. The integration of M2M technology in healthcare has the potential to save global healthcare systems up to $36 billion, underscoring its financial and operational benefits. As Muni Kumar Meravath, a Healthcare Market Research Analyst, observes, "One of the key factors driving the market is the growing need for (RPM) to help manage chronic conditions like diabetes, heart disease, and respiratory issues."
In conclusion, is a transformative force in the medical equipment landscape, driving innovations that lead to improved and outcomes.
Identify M2M Implementation Requirements and Components
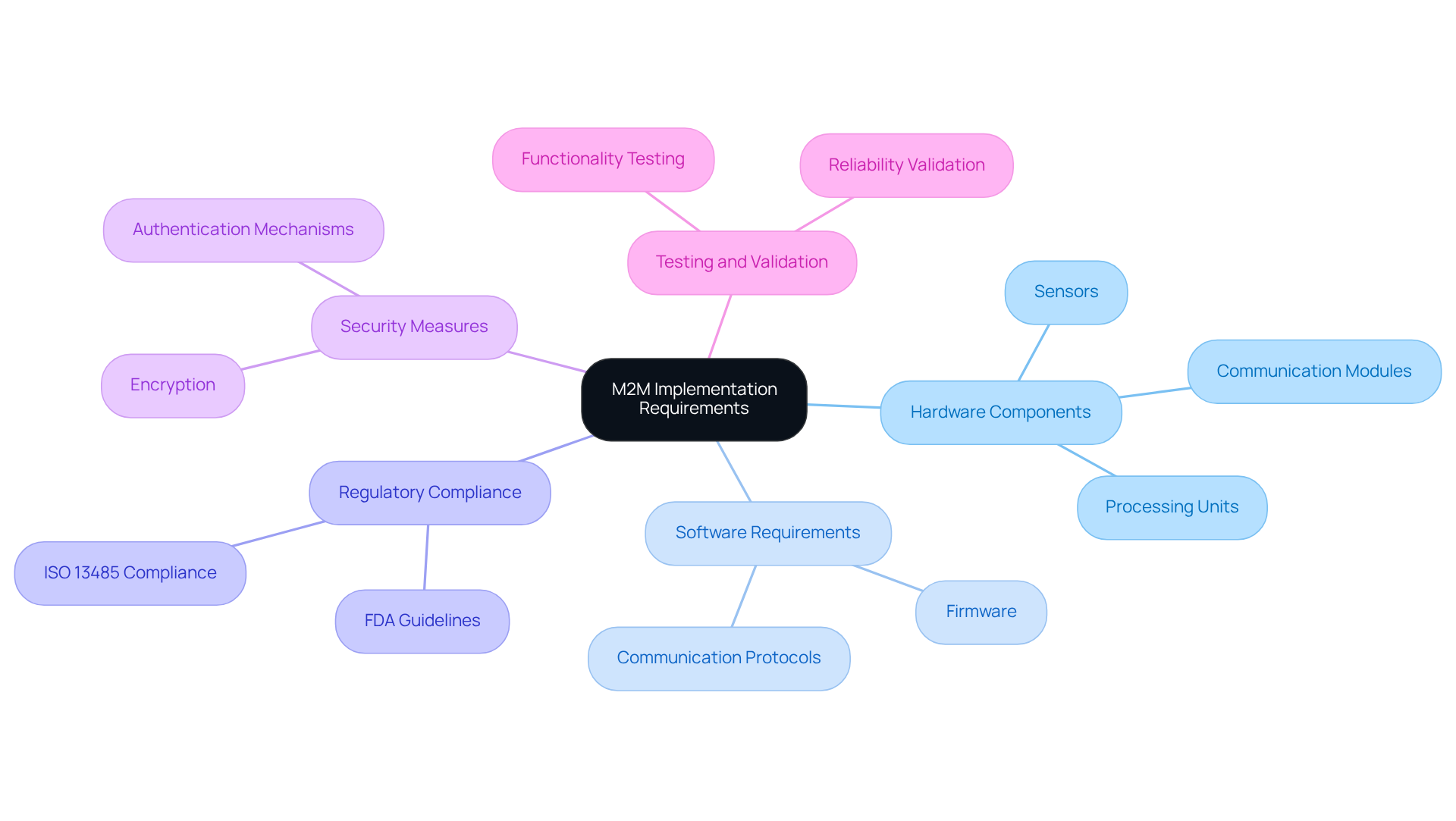
To effectively implement in , it is crucial to identify several key requirements and components:
-
Hardware Components:
- Sensors: Vital for gathering patient data, such as heart rate, blood pressure, and other health metrics, these enable .
- : Essential for information transfer, these include cellular, Wi-Fi, or Bluetooth modules that facilitate connectivity between devices and healthcare systems.
- Processing Units: Microcontrollers or processors are necessary for handling information processing and communication tasks, ensuring the efficient functioning of the equipment.
-
Software Requirements:
- Firmware: Custom firmware is required to oversee data collection and transmission, tailored to the specific functionalities of the medical device.
- : Implementing protocols such as MQTT or HTTP is critical for efficient information exchange, ensuring that messages are transmitted reliably and promptly.
-
: Ensuring that the device adheres to industry standards and regulations, including , is essential to guarantee safety and effectiveness.
-
Security Measures: Robust must be established to protect patient data during transmission. This includes implementing encryption and authentication mechanisms to safeguard sensitive information.
-
: Thorough testing is necessary to validate the functionality and reliability of the M2M connection system before deployment. This step is crucial to ensure that the apparatus functions as intended and fulfills all regulatory requirements.
Integrate M2M Communication into Your Devices
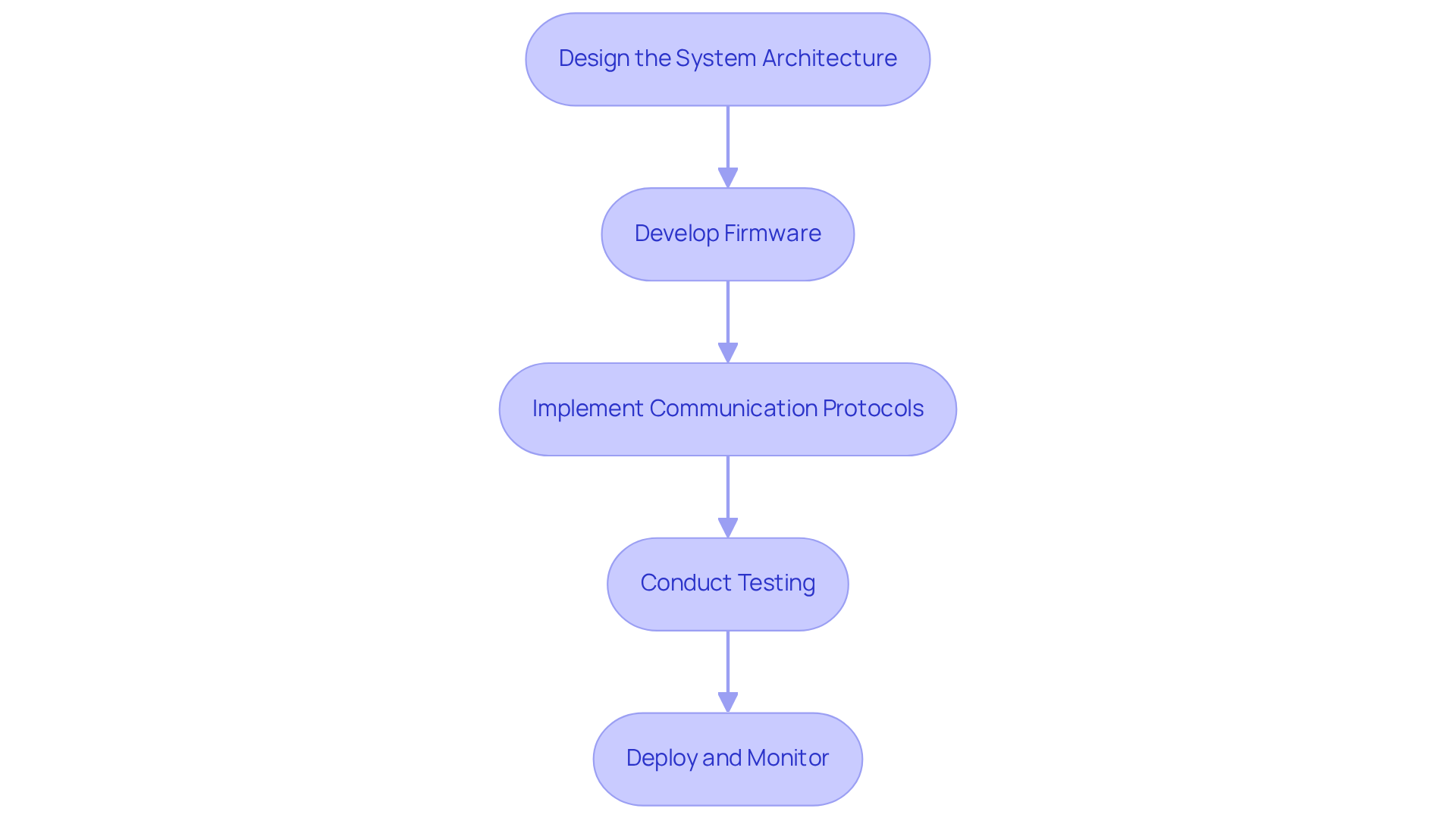
Integrating into involves several essential steps:
-
Design the :
- Begin by outlining the interaction between sensors, communication modules, and processing units. A flow diagram can be advantageous to visualize information paths and ensure smooth integration.
-
Develop Firmware:
- Create and rigorously test the firmware that will operate on the device. This firmware must efficiently gather information from sensors and enable its transmission through the chosen .
-
Implement :
- Integrate robust into the firmware, ensuring that data is transmitted securely and efficiently. Adopting standards like IEEE 11073 can improve interoperability among equipment.
-
Conduct Testing:
- Execute thorough unit testing on individual components and comprehensive system testing on the entire device. This step is crucial to confirm that as intended and meets .
-
Deploy and Monitor:
- After successful testing, deploy the device in a controlled environment. Ongoing observation of its performance is essential, enabling modifications to enhance and overall functionality of the apparatus. Regular security assessments should also be conducted to safeguard sensitive patient data.
By adhering to these steps, manufacturers can guarantee that their medical equipment is designed for or effectively, thereby improving and operational efficiency.
Troubleshoot Common M2M Implementation Challenges
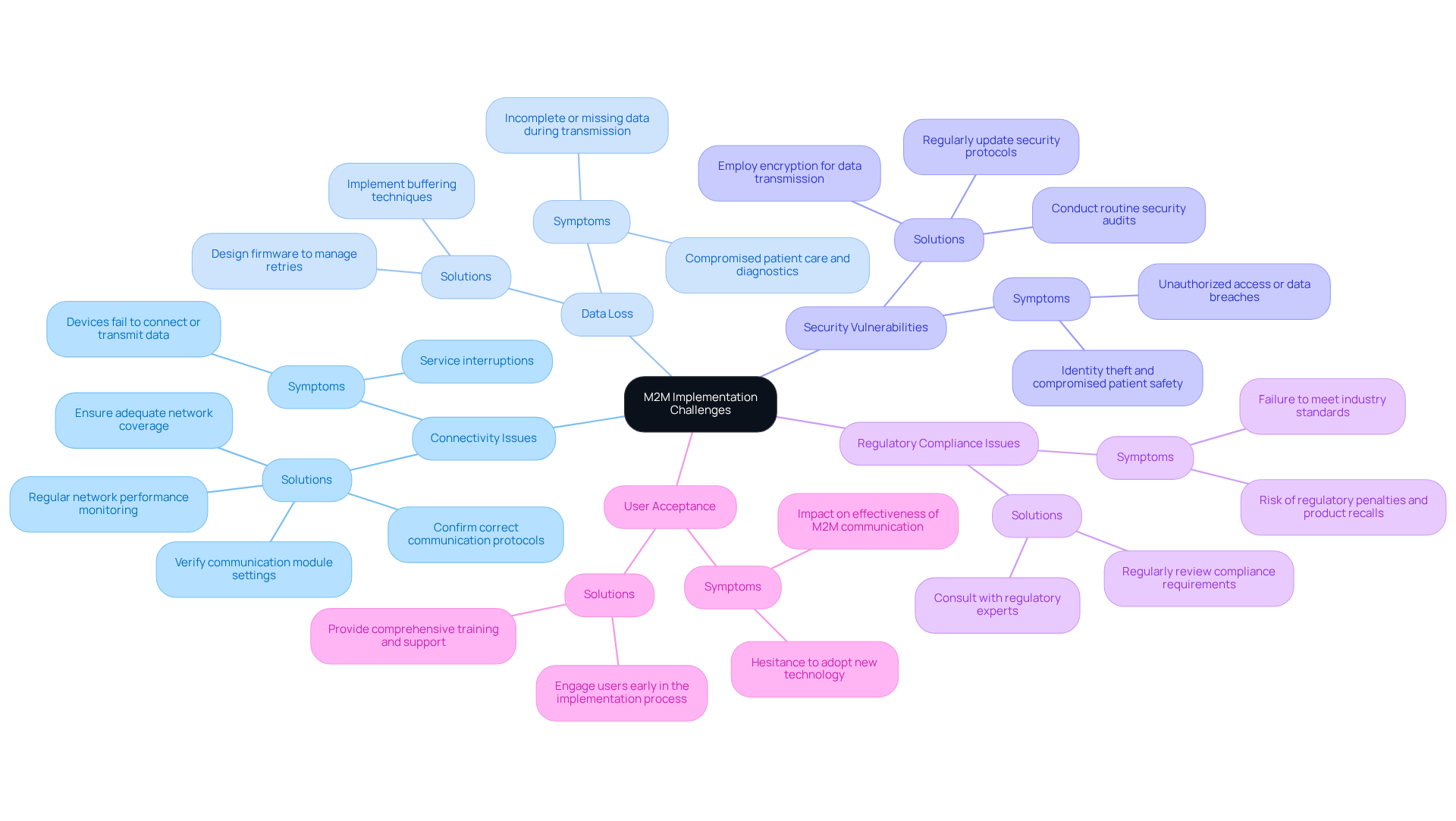
Implementing M2M interaction in medical devices presents several challenges that must be effectively addressed, especially since it is designed for machine to machine or . Below are common issues along with their corresponding solutions:
-
:
- Symptoms: Devices may fail to connect or transmit data, resulting in service interruptions.
- Solutions: Verify the communication module settings, ensure adequate network coverage, and confirm that the correct communication protocols are in place. Regular monitoring of network performance can help identify potential connectivity problems early.
-
:
- Symptoms: Incomplete or missing data during transmission can compromise patient care and diagnostics.
- Solutions: Implement buffering techniques to temporarily store information during transmission failures. Additionally, ensure that the firmware is designed to manage retries for unsuccessful transmissions, which can significantly reduce the occurrence of .
-
:
- Symptoms: Unauthorized access or data breaches can lead to severe consequences, including identity theft and compromised patient safety.
- Solutions: Regularly update security protocols and employ encryption for data transmission. Conducting routine security audits can help identify and mitigate vulnerabilities before they are exploited.
-
:
- Symptoms: Devices may fail to meet industry standards, risking regulatory penalties and product recalls.
- Solutions: Regularly review compliance requirements and consult with regulatory experts to ensure adherence to guidelines set by the FDA and HIPAA. This proactive approach can help avoid .
-
:
- Symptoms: Users may be hesitant to adopt new technology, impacting the effectiveness of .
- Solutions: Provide comprehensive training and support to users, emphasizing the benefits of for enhancing patient care and operational efficiency. Engaging users early in the implementation process can foster acceptance and ease the transition to new systems.
Conclusion
Implementing Machine-to-Machine (M2M) communication in medical devices marks a significant advancement in healthcare technology, facilitating seamless data exchange that enhances patient care. This integration not only enables real-time monitoring and efficient communication between devices but also empowers healthcare providers to make timely decisions based on accurate data.
This guide has thoroughly explored the key components of M2M implementation, including:
- Essential hardware and software requirements
- The importance of regulatory compliance
- The necessity for robust security measures
Additionally, it has highlighted various applications of M2M technology, ranging from wearable health monitors to telehealth solutions, demonstrating its potential to transform patient engagement and operational efficiency in healthcare settings.
As the healthcare landscape continues to evolve, embracing M2M communication is crucial for improving patient outcomes and optimizing resource management. Stakeholders in the medical device industry are encouraged to adopt these technologies and best practices to remain at the forefront of innovation. By prioritizing M2M integration, the healthcare sector can pave the way for a future where patient care is not only enhanced but also more efficient and effective.
Frequently Asked Questions
What is Machine-to-Machine (M2M) communication?
M2M communication refers to the automated transfer of information between devices without human involvement, enabling seamless information exchange, particularly in the medical equipment sector.
How does M2M communication benefit healthcare?
M2M communication significantly improves patient care by allowing systems to share vital information, such as heart rate or glucose levels, directly with healthcare providers, facilitating timely interventions and enhancing patient outcomes.
What technologies are involved in M2M interaction?
M2M interaction encompasses various technologies that enable devices to connect directly, utilizing both wired and wireless networks, with protocols like MQTT and CoAP designed for efficient information transmission.
What are some common applications of M2M in medical devices?
Common applications include wearable health monitors, implanted devices, and telehealth solutions that enable remote consultations and monitoring, allowing patients to measure health metrics and transmit data to healthcare providers.
What is the projected market size for M2M in the medical sector by 2030?
The global M2M medical market is projected to reach USD 35.30 billion by 2030, indicating an increasing reliance on these technologies to optimize healthcare delivery.
What are the benefits of M2M interaction in healthcare?
Benefits include enhanced operational efficiency, reduced costs, improved patient engagement through continuous monitoring and feedback, and the potential to save global healthcare systems up to $36 billion.
What is driving the growth of M2M technology in healthcare?
The growing need for remote patient monitoring (RPM) to help manage chronic conditions like diabetes, heart disease, and respiratory issues is a key factor driving the market.
List of Sources
- Understand Machine-to-Machine (M2M) Communication
- Healthcare M2M Market Size, Share, Trends Report 2032 (https://precisionbusinessinsights.com/market-reports/healthcare-m2m-market)
- M2M in healthcare - telemedicine (https://en.whereversim.de/iot-news/m2m-sim-telemedicine)
- Master M2M Anwendungen: Best Practices for Medical Device Success (https://volersystems.com/blog/master-m2m-anwendungen-best-practices-for-medical-device-success)
- Machine-to-Machine (M2M) Communication: Revolutionizing the Future of Connectivity (https://minewingems.com/news/machine-to-machine-m2m-communication-revolutionizing-the-future-of-connectivity)
- Identify M2M Implementation Requirements and Components
- EUDAMED Mandatory by May 2026: Key Steps for Manufacturers (https://celegence.com/eudamed-mandatory-28-may-2026-medical-device-ivd-manufacturers)
- FDA’s New QMSR Rule for 2026: What Medical Device Manufacturers Must Do Now (https://theoris.com/fdas-new-qmsr-rule-for-2026-what-medical-device-manufacturers-must-do-now)
- M2M in healthcare - telemedicine (https://en.whereversim.de/iot-news/m2m-sim-telemedicine)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- Navigate 2026's Medical Device Manufacturing Trends (https://mddionline.com/medical-device-regulations/navigate-2026-s-medical-device-manufacturing-trends)
- Integrate M2M Communication into Your Devices
- Master Machine Communication: Best Practices for Medical Devices (https://volersystems.com/blog/master-machine-communication-best-practices-for-medical-devices?hs_amp=true)
- M2M in healthcare - telemedicine (https://en.whereversim.de/iot-news/m2m-sim-telemedicine)
- Rise of the machines: The future of M2M in healthcare - Embedded Computing Design (https://embeddedcomputing.com/application/healthcare/rise-of-the-machines-the-future-of-m2m-in-healthcare)
- MedTech Product Development Trends in 2026 (https://enlil.com/blog/medtech-product-development-trends-in-2026)
- MedTech Trends Shaping 2026 (https://iqvia.com/blogs/2026/02/medtech-trends-shaping-2026)
- Troubleshoot Common M2M Implementation Challenges
- Medical data leaks from over 1M healthcare IoT devices (https://iottechnews.com/news/medical-data-leaks-over-1m-healthcare-iot-devices)
- M2M Vulnerabilities in Medical Devices - Blue Goat Cyber (https://bluegoatcyber.com/blog/m2m-communication-vulnerabilities-in-medical-devices-a-cybersecurity-concern)
- Top 10 Challenges in the MedTech industry (https://ptc.com/en/blogs/medtech/medical-device-industry-challenges?srsltid=AfmBOorYPeCUNWLmRnD-aUlh7QBIdQX2xvjkEQVDsOc96GleTLcDm9wf)
- Healthcare bodies exposed to cyber-security risks - IoT M2M Council (https://iotm2mcouncil.org/iot-library/news/connected-health-news/healthcare-bodies-exposed-to-cyber-security-risks)
- Five Challenges in Connected Medical Device Development: And How to Overcome Them (https://s3connectedhealth.com/blog/five-challenges-in-connected-medical-device-development-and-how-to-overcome-them)





